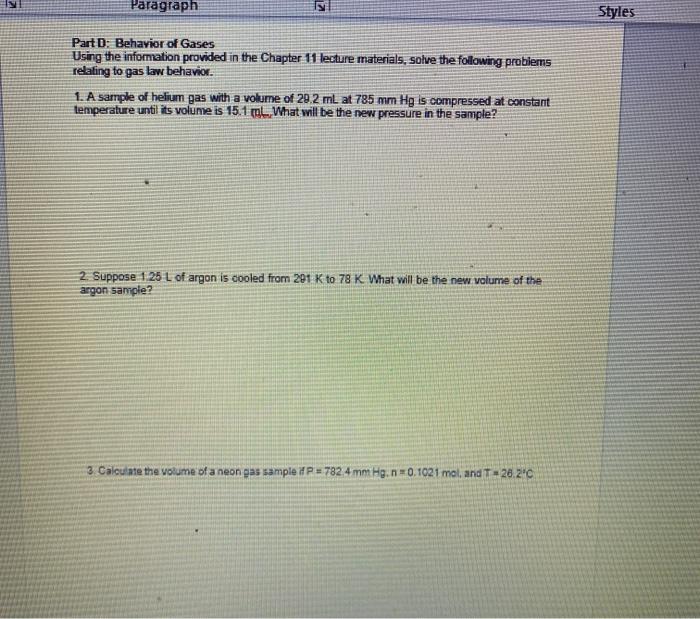
Paragraph Part D: Behavior of Gases Using the information provided in the Chapter 11 lecture materials,...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Paragraph Part D: Behavior of Gases Using the information provided in the Chapter 11 lecture materials, solve the following problems relating to gas law behavior. 1. A sample of helium gas with a volume of 29.2 ml at 785 mm Hg is compressed at constant temperature until its volume is 15.1 ml What will be the new pressure in the sample? 2. Suppose 1.25 L of argon is cooled from 291 K to 78 K What will be the new volume of the argon sample? 3 Calculate the volume of a neon gas sample if P 782 4 mm Hg. n=0.1021 mol, and T-26.2°C Styles Paragraph Part D: Behavior of Gases Using the information provided in the Chapter 11 lecture materials, solve the following problems relating to gas law behavior. 1. A sample of helium gas with a volume of 29.2 ml at 785 mm Hg is compressed at constant temperature until its volume is 15.1 ml What will be the new pressure in the sample? 2. Suppose 1.25 L of argon is cooled from 291 K to 78 K What will be the new volume of the argon sample? 3 Calculate the volume of a neon gas sample if P 782 4 mm Hg. n=0.1021 mol, and T-26.2°C Styles
Expert Answer:
Answer rating: 100% (QA)
1 To with change in the volume we PV P V 2 find out the new pressure of the wh... View the full answer

Related Book For 

Modern Portfolio Theory and Investment Analysis
ISBN: 978-1118469941
9th edition
Authors: Edwin Elton, Martin Gruber, Stephen Brown, William Goetzmann
Posted Date:
Students also viewed these accounting questions
-
12.31 Deciding on a new venture *** Ben Wong wants to buy a taxi plate in the inner Melbourne suburb of Richmond. Taxi regis- tration plates (which confer the right to own and operate a taxi) are...
-
A large distributor of oil-well drilling equipment operated over the past two years with EOQ policies based on an annual holding cost rate of 28%. Under the EOQ policy, a particular product has been...
-
Use this information to compute the following: FunTime Cruiseline offers nightly dinner cruises departing from several cities on the eastern coast of the United States including Charleston,...
-
Mr. Ajay's trial balance is as follows. Trial Balance for the year ended 31st March, 2017 Amt Debit Balances To Opening Stock To Purchases To Wages To Investment To Carriage outwards To Printing and...
-
a. Is it the long-term nominal interest rate or the long-term real interest rate that influences spending decisions? Explain why. b. How does the market determine the long-term nominal interest rate...
-
COMPUTE CASH PROVIDED BY OPERATING ACTIVITIES Mueller Company's condensed income statement for the year ended December 31, 20-2, was as follows: Net sales..........$900,000 Cost of goods...
-
In an engine working on diesel cycle, inlet pressure and temperature are 1 bar and \(20^{\circ} \mathrm{C}\). Pressure at the end of adiabatic compression is 40 bar. The ratio of expansion after...
-
Income statement data for Montana Images Company for the years ended December 31, 2012 and 2011, are as follows: a. Prepare a comparative income statement with horizontal analysis, indicating the...
-
Mercier Manufacturing produces a plastic part in three sequential departments: Extruding, Fabricating, and Packaging. Mercier uses the weighted-average process costing method to account for costs of...
-
REI sells snowboards. Assume the following information relates to REI's purchases of snowboards during September. During the same month, 100 snowboards were sold. REI uses a periodic inventory...
-
What is meant by the "Castle Doctrine" and the key reasons the doctrine has changed over the years? Contrast the "Castle Doctrine" with the "Make my Day" rules. Which do you agree with and why? If...
-
Reynolds Corp. acquired the assets of Putvan Corp. in a nontaxable merger transaction on December 1, 2018. On the date of acquisition, Putvan had a net operating loss carryover of \(\$ 500,000\)....
-
How can SHRD help support second-order strategy changes designed to produce a flatter organisational structure through delayering?
-
Tatco's pretax book income and taxable income differ because of one item, warranty expense. Tatco reports a \(\$ 200,000\) warranty expense and a related estimated liability for warranties for...
-
To what extent do you think that the lack of objectivity in performance appraisal may be overcome in performance management?
-
Imagine that you work as an HR officer for a company that is unhappy with the effectiveness of its current recruitment and selection practice. You have been asked to write a paper presenting a...
-
Where do companies generally explain the total amAshton is an investor looking at the income statements of two different companies. The first company has a very detailed income statement, and the...
-
APC16550D UART has a clock running at18.432 MHz and its baud rate is set to 2000.Determine the HEX contents of its DLM and DLL registers. Please can you explain step by step and in detail how you get...
-
For the two securities shown, plot all combinations of the two securities in space. Assume p = 1, -1, 0. For each correlation coefficient, what is the combination that yields the minimum p and what...
-
The analyst who supplied you with the information in Problem 1 has just revised her forecast. She now realizes that the growth rate of 10% can continue for only five years, after which the company...
-
Assume that the following two-index model describes returns: Assume that the following three portfolios are observed. Find the equation of the plane that must describe equilibrium returns. Portfolio...
-
Pegasus Veterinary Hospital (PVH) is a partnership of five veterinarian surgeons who specialize in treating horses. PVH is located in a $25 million facility on 20 acres of land, employing over 40...
-
A tender evaluation method specifies 60% price and 40% quality. What is the weighted score of a tender that scored 80 points (out of 100) for price and 60 for quality?
-
You are a recently appointed purchasing manager who is overseeing terms and con- ditions of contract for an important international purchase with a UN country. What rules should you be particularly...

Study smarter with the SolutionInn App


