Absorb ammonia from air into water at 20.0C and 1.5 atm pressure. Inlet water is recycled from
Question:
Absorb ammonia from air into water at 20.0°C and 1.5 atm pressure. Inlet water is recycled from a stripper and contains 0.2 wt% ammonia. Gas flow rate is 100.0 kmol/h. Inlet gas is 3.1 mol% ammonia. Exit gas should be 0.31 mol% ammonia. Assume L/V is constant. Equilibrium data are in Table 12-3.
a). What is the minimum flow rate of water in kg/h?
b). What is the exit concentration of the liquid, and how many equilibrium stages are needed if L = 1.5 X Lmin?
Note: Keep your units straight. In Table 12-3, 10.0 weight NH3 per 100.0 weight water = 10.0 weight/110.0 weight solution =0.090909 mass fraction.
Approach 1: Convert all units to either mass or to moles. [MW NH3 = 17.0, MW H2O = 18.0, MW air = 28.97
Approach 2: Keep liquid in weight fraction and gas in mole fraction, and plot equilibrium in this form. In the NH3 mass balance, if y is mole fraction NH3 in gas and x is weight fraction ammonia in liquid, Vy (kmol gas/h) (kmol NH3/kmol gas) = kmol NH3/h, and LX = (kg liquid/h) (kg NH3/kg liquid) = kg NH3/h. Then adjust the NH3 mass balance with the molecular weight of NH3 so that all terms are in kg NH3/h. Derive the operating equation from this form of mass balance.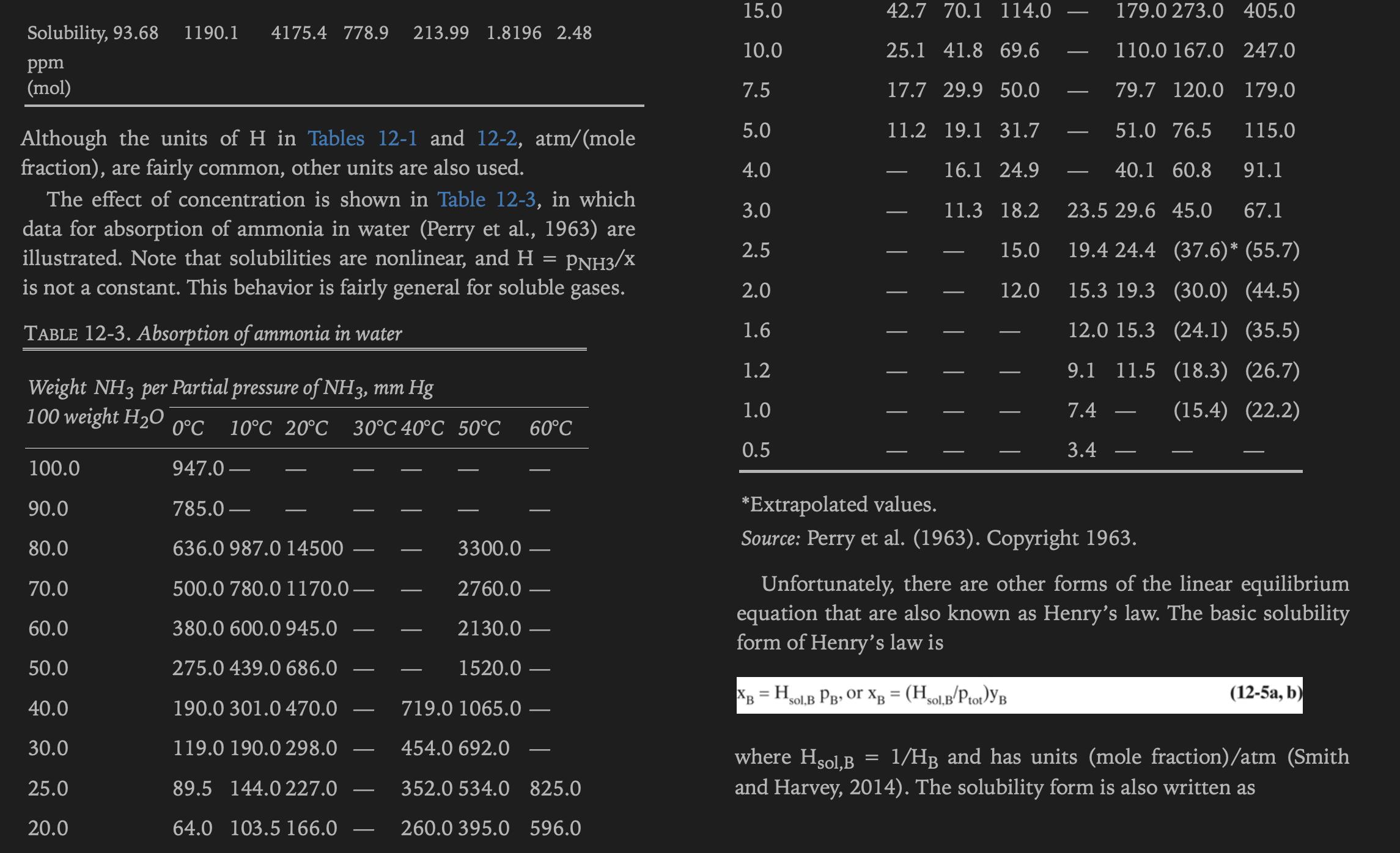

Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau




