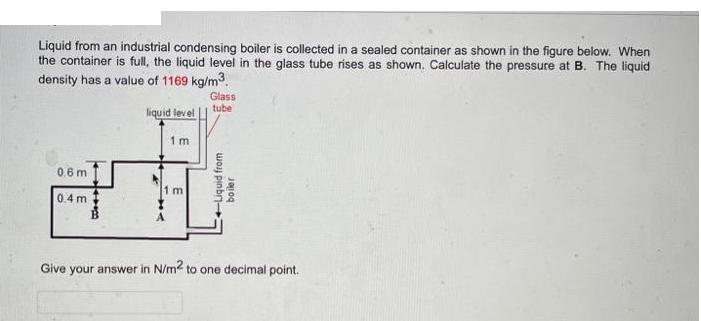
Liquid from an industrial condensing boiler is collected in a sealed container as shown in the...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Liquid from an industrial condensing boiler is collected in a sealed container as shown in the figure below. When the container is full, the liquid level in the glass tube rises as shown. Calculate the pressure at B. The liquid density has a value of 1169 kg/m³. liquid level 0.6 m 0.4 m Wee 1m 1 m Glass tube +Liquid from boiler Give your answer in N/m² to one decimal point. Liquid from an industrial condensing boiler is collected in a sealed container as shown in the figure below. When the container is full, the liquid level in the glass tube rises as shown. Calculate the pressure at B. The liquid density has a value of 1169 kg/m³. liquid level 0.6 m 0.4 m Wee 1m 1 m Glass tube +Liquid from boiler Give your answer in N/m² to one decimal point.
Expert Answer:
Answer rating: 100% (QA)
SOLUTION To calculate the pressure at point B we need to consider the h... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these accounting questions
-
An investor wishes to analyse the effects of different compounding frequencies Suppose 1000 is invested for 1 year at an interest rate of 5 per annum compounded Assume there are 365 days in 1 year
-
Your new designer chair has an S-shaped tubular metal frame that behaves just like a spring with the spring constant 13600 N/m. When your friend, who weighs 1011 N. sits on the chair, how far does it...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Violet Flowers expects to sell 3,000 plants a month. She estimated the following monthly costs: Variable Costs $9,000 Fixed Costs $15,000 During her second month of operation, Violet would like to...
-
Draw an energy diagram for a reaction with Keq = 1. What is the value of G in this reaction?
-
Using Tables 6-4, 6-5, and 6-6, determine the freight charges for the following shipments: a. A 2,500-lb shipment of paper place mats with printed advertising moving from New York to Los Angeles. b....
-
A ball falling near the surface of the Earth where air friction is not negligible eventually slows down to a constant "terminal" velocity. How is that possible given that frictional force increases...
-
Carbex, Inc., produces cutlery sets out of high-quality wood and steel. The company makes a standard cutlery set and a deluxe set and sells them to retail department stores throughout the country....
-
A ( n ) _ _ _ _ _ _ _ _ _ _ contains information about a given person, product, or event. Attribute Column Field Record
-
Work these problems relating to after-tax cash flow (ATCF) computations: 1. Compute the present value after-tax cost ("PVATC") of a $9,500,000 purchase of equipment on January 1 that your corporate...
-
Given the information below, how long is the Critical Path? Activity Time A B C D E FL 9 H - 2 2 5 1 7 2 5 6 5 10 Immediate Predecessor A B C, D E F LL. F G, H D
-
Why was interspousal immunity created, and what is its status today?
-
True Or False Many courts have abolished local governmental immunity.
-
True Or False State tort claims acts usually require that written notice of a claim be given during a designated time period after the discovery of the injury.
-
In deciding what should be disclosed to a patient, some courts a. consider the expectations of a reasonable layperson. b. use a prudent patient standard, which requires a doctor to disclose those...
-
The last-clear-chance doctrine a. mitigates the harshness of the contributorynegligence defense. b. prevents the plaintiff from recovering if both the plaintiff and defendant are inattentive and the...
-
Consider the reaction below: BH 3 + CH3OCH 3 Which of the following statements is INCORRECT? O a. BH3 is an electrophile. O b. CH3OCH3 is a Lewis base. O c. This is an example of a substitution...
-
The Pletcher Transportation Company uses a responsibility reporting system to measure the performance of its three investment centers: Planes, Taxis, and Limos. Segment performance is measured using...
-
Frederick (Freddy) Freeweights (age 42) owns and operates a health club called ''Treadmills West.'' The business is located at 4321 Arnold Street, Alpine, CA 91901. Freddy had the following income...
-
Bev and Ken Hair have been married for 3 years. They live at 3567 River Street, Springfield, MO 63126. Ken is a full-time student at Southwest Missouri State University (SMSU) and Bev works as an...
-
Yolanda earns $112,000 in 2012. Calculate the FICA tax that must be paid by: Yolanda:.....................Soc.Sec..................$__________...
-
Based on the photographs in Figure 26.13, in which segment(s) is the Antp gene normally expressed? Figure 26.13: (a) Normal fly (b) Antennapedia mutant
-
The bush baby, a small African mammal, is a remarkable jumper. Although only about 8 inches long, it can jump, from a standing start, straight up to a height of over 7 feet! Use the particle model to...
-
Hicham El Guerrouj of Morocco holds the world record in the 1500 m running race. He ran the final 400 m in a time of 51.9 s. What was his average speed in mph over the last 400 m? A. 14.2 mph B. 15.5...

Study smarter with the SolutionInn App


