Aleia has a water bottle whose water has gotten pretty warm. She drops ten -13 degree...
Fantastic news! We've Found the answer you've been seeking!
Question:
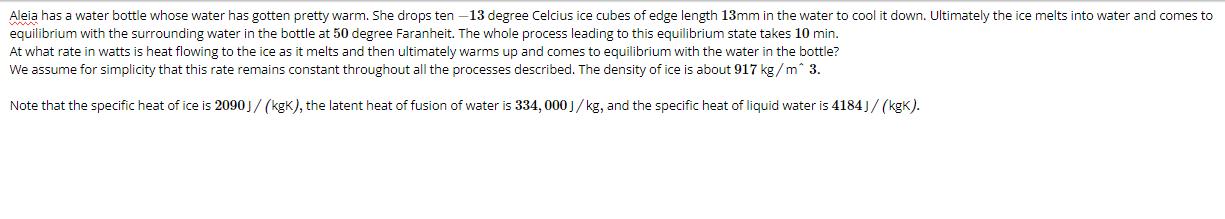
Transcribed Image Text:
Aleia has a water bottle whose water has gotten pretty warm. She drops ten -13 degree Celcius ice cubes of edge length 13mm in the water to cool it down. Ultimately the ice melts into water and comes to equilibrium with the surrounding water in the bottle at 50 degree Faranheit. The whole process leading to this equilibrium state takes 10 min. At what rate in watts is heat flowing to the ice as it melts and then ultimately warms up and comes to equilibrium with the water in the bottle? We assume for simplicity that this rate remains constant throughout all the processes described. The density of ice is about 917 kg/m^ 3. Note that the specific heat of ice is 2090 J/(kgK), the latent heat of fusion of water is 334, 000 J/kg, and the specific heat of liquid water is 4184]/(kgK). Aleia has a water bottle whose water has gotten pretty warm. She drops ten -13 degree Celcius ice cubes of edge length 13mm in the water to cool it down. Ultimately the ice melts into water and comes to equilibrium with the surrounding water in the bottle at 50 degree Faranheit. The whole process leading to this equilibrium state takes 10 min. At what rate in watts is heat flowing to the ice as it melts and then ultimately warms up and comes to equilibrium with the water in the bottle? We assume for simplicity that this rate remains constant throughout all the processes described. The density of ice is about 917 kg/m^ 3. Note that the specific heat of ice is 2090 J/(kgK), the latent heat of fusion of water is 334, 000 J/kg, and the specific heat of liquid water is 4184]/(kgK).
Expert Answer:
Related Book For 

Physics
ISBN: 978-0077339685
2nd edition
Authors: Alan Giambattista, Betty Richardson, Robert Richardson
Posted Date:
Students also viewed these physics questions
-
Use the following information for Funk Ltd to answer the questions below. Assume errors (if any) were committed by the company Funk Ltd Bank Statement, April of 2011 Date 4/1 Description Beginning...
-
A portion of the housing market in New York City (and many other cities in the world) is regulated through a policy known as rent control. In essence, this policy puts a price ceiling (below the...
-
A 28-g ice cube at its melting point is dropped into an insulated container of liquid nitrogen. How much nitrogen evaporates if it is at its boiling point of 77 K and has a latent heat of...
-
The following are some examples of typical business transactions: 1. Purchased office equipment on credit. 2. The owner contributed capital. 3. Purchased office furniture for cash. 4. Received...
-
Assume that on January 2, 2013, a Pizza Hut franchise purchased fixtures for $15,000 cash, expecting the fixtures to remain in service five years. The restaurant has depreciated the fixtures on a...
-
Why the activity "preventive maintenance" can be a value-added activity? I think it is not because (1) it neither increases customer value nor brings the product closer to completion and (2) it is...
-
Oliver Inc. makes three types of olive oil that can be sold at split-off or processed further and then sold. The joint cost for October is \(\$ 325,000\). The number of ounces in a bottle of each...
-
When Lehman was developing its Repo 105 accounting policy, did E&Y have a responsibility to be involved in that process? In general, what role should an audit firm have when a client develops an...
-
The amount of budgeted overhead costs at normal capacity of P240,000 was divided by normal capacity of 30,000 direct labor hours to arrive at the predetermined overhead rate of P8, comprised of a...
-
In an ideal reheat-regenerative cycle steam enters the high-pressure (H.P.) turbine at 90 bar, 600C. After expansion to 7 bar, some of the steam goes to a direct contact open heater and the balance...
-
This may be news to you, but hockey is getting big in Northern China. Blue Line Hockey Sticks has decided to capitalize on the trend by setting up subsidiaries in this country. You have been...
-
Sunland Corporation, an amusement park, is considering a capital investment in a new exhibit. The exhibit would cost $ 2 0 6 , 7 2 5 and have an estimated useful life of 1 0 years. It can be sold for...
-
A local radio station broadcasts signals (travelling at 3.00 108 m/s) whose wavelength is 3.102 m. At what frequency does this station broadcast?
-
How many grams of benzoic acid (molar mass = 122.12 g/mol) must be dissolved in 9.00 g of lauric acid to create a solution with a molality of 0.910 mol/kg lauric acid? (Hint: Pay attention to units)...
-
Descriptive, predictive, and prescriptive statistics allow us to manage tomorrow, today (Pease, Byerly & Fitz-enz, 2012). They can help us see what currently is and show us a path on how we can be...
-
What motivation for pursuing interstellar travel do you find interesting? Do you think the possible benefits of pursuing interstellar travel would justify significant investment and time to pursue...
-
Easter Egg and Poultry Company has $1,640,000 in assets and $695,000 of debt. It reports net income of $161,000. What is the firm's return on assets? Note: Enter your answer as a percent rounded to 2...
-
Thalina Mineral Works is one of the worlds leading producers of cultured pearls. The companys condensed statement of cash flows for the years 20182020 follows. Required Comment on Thalina Mineral...
-
The driver from Practice Problem 13.3 fills his 18.9-L steel gasoline can in the morning when the temperature of the can and the gasoline is 15.0 C and the pressure is 1.0 atm, but this time he...
-
An electron is moving at nearly light speed. A constant force of magnitude F is acting on the electron in the direction of its motion. Is the acceleration of the electron less than, equal to, or...
-
A 1.10-kg bucket is tied to a rope that is wrapped around a pole mounted horizontally on frictionless bearings. The cylindrical pole has a diameter of 0.340 m and a mass of 2.60 kg. When the bucket...
-
Decade, Inc., recorded certain capital stock transactions shown in the following journal entries: (1) issued common stock for \(\$ 25\) cash per share, (2) purchased treasury shares at \(\$ 40\) per...
-
Label each of the following characteristics of a corporation as either an (A) advantage or a (D) disadvantage: a. Limited liability b. Taxation c. Regulations d. Transferability of ownership
-
The following information relates to Ontario Components, Inc.: a. Calculate the company's return on common stockholders' equity for 2018 and 2019. b. Calculate the company's dividend yield for 2018...

Study smarter with the SolutionInn App



