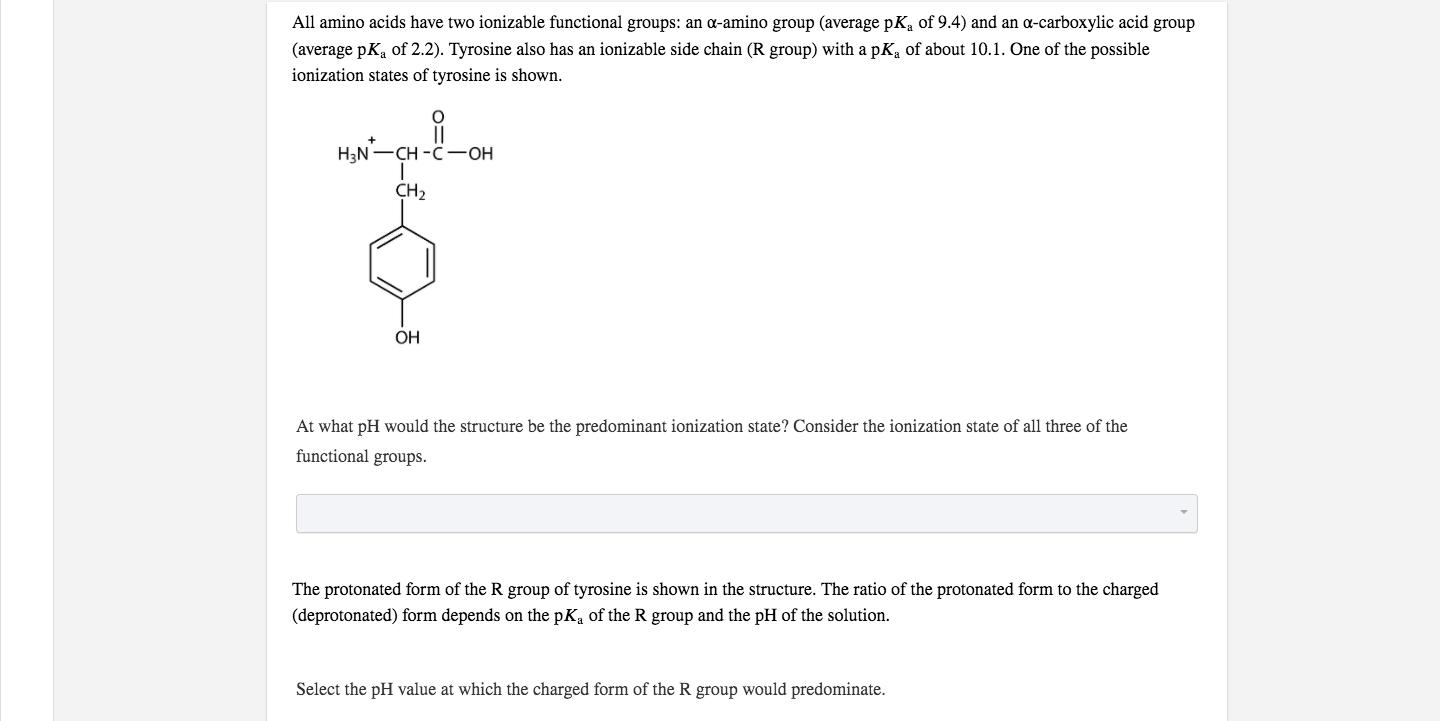
All amino acids have two ionizable functional groups: an a-amino group (average pKa of 9.4) and...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
All amino acids have two ionizable functional groups: an a-amino group (average pKa of 9.4) and an a-carboxylic acid group (average pK, of 2.2). Tyrosine also has an ionizable side chain (R group) with a pK of about 10.1. One of the possible ionization states of tyrosine is shown. H3N-CH -C-OH CH2 OH At what pH would the structure be the predominant ionization state? Consider the ionization state of all three of the functional groups. The protonated form of the R group of tyrosine is shown in the structure. The ratio of the protonated form to the charged (deprotonated) form depends on the pKa of the R group and the pH of the solution. Select the pH value at which the charged form of the R group would predominate. All amino acids have two ionizable functional groups: an a-amino group (average pKa of 9.4) and an a-carboxylic acid group (average pK, of 2.2). Tyrosine also has an ionizable side chain (R group) with a pK of about 10.1. One of the possible ionization states of tyrosine is shown. H3N-CH -C-OH CH2 OH At what pH would the structure be the predominant ionization state? Consider the ionization state of all three of the functional groups. The protonated form of the R group of tyrosine is shown in the structure. The ratio of the protonated form to the charged (deprotonated) form depends on the pKa of the R group and the pH of the solution. Select the pH value at which the charged form of the R group would predominate.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
At what pH would 95 percent of the ammonia be present as gas? Acid ionization (dissociation) constant at 25 o C = 5.62*10 -10 .
-
Which amino acids in Table 17.1 have nonpolar R groups? Highly polar groups? Relatively flat R groups? Table 17.1 QUESTION CONTINUE TO NEXT PAGE Table 17.1 Names and Formulas of the Common Amino...
-
Consider the fully protonated amino acid valine: where the numbers denote the pKa values. (b) Calculate the predominant form of valine at pH 1.0, 7.0, and 12.0. (c) Calculate the isoelectric point of...
-
Here is the end-of-year account balance information from the accounting records of Jaunty Coffee Company: Sales revenue Cash Cost of goods sold Accounts payable Capital stock Dividends Retained...
-
Rework Prob. 9-99 when the compressor isentropic efficiency is 87 percent and the turbine isentropic efficiency is 93 percent. Prob. 9-99 A gas turbine for an automobile is designed with a...
-
Given that Bit Digital was up by about 3,688 percent for 2020, why didnt all investors hold Bit Digital?
-
A face-centered cubic array of round fibers is shown in Figure 3.6. Derive the relationship between the fiber volume fraction and the given geometrical parameters. What is the maximum possible fiber...
-
Berry Company sold goods with a total selling price of $800,000 during the year. It purchased goods for $380,000 and had beginning inventory of $67,000. A count of its ending inventory determined...
-
How does memory management in RTOS differ from that in general-purpose operating systems? Discuss techniques for real-time memory allocation and their impact on system performance
-
On June 2, 2017, Betty Rice opened Betty's Art Studio in Toronto. The following transactions occurred in June: 2017 June .... 2 Betty Rice invested $12,000 in the art studio. ........... 3 Paid three...
-
How would adapting communication style fit the needs of a client or colleague from a different country and language globally? how you would tailor my communication style to put my audience at ease....
-
The following facts relate to Alschuler Corporation. 1. Deferred tax liability, January 1, 2015, 40,000. 2. Deferred tax asset, January 1, 2015, 0. 3. Taxable income for 2015, 115,000. 4. Pretax...
-
financial income of 80,000 for 2015. The following items cause taxable income to be different than pretax financial income. 1. Depreciation on the tax return is greater than depreciation on the...
-
Fuel utilization is defined as amount of initial fissile material required per unit energy (g/MWd). For example, CANDU uses natural uranium with \(0.71 \%\) U-235 and generates 7.0 GWd/t. It implies...
-
Higley Corporation has one temporary difference which will reverse and cause taxable amounts in 2015. In 2014, a new tax act set taxes equal to 45% for 2014, 40% for 2015, and 34% for 2016 and years...
-
For an efficient amount of a public good to be provided, the marginal cost of producing another unit of that good must equal the marginal benefit to each individual who consumes the good. Is this...
-
In the battle to out-blade the competition, Gillette's 2005 creation, a five-bladed razor called Fusion, leapfrogged the Schick Quattro by one blade and aimed to provide an even closer shave to the...
-
What are the four types of poultry production systems? Explain each type.
-
An experimenter makes up a solution of 0.310 mol Na3PO4, 0.100 mol Ca(NO3)2, and 0.150 mol AgNO3 in 4.000 L of water solution. Note any precipitations that occur, writing a balanced equation for...
-
Consider the complex ion [CoF6]3. a. What is the geometry? b. Which is a more likely color for this ion to absorb, red or blue? c. Would you expect this complex to be high or low spin?
-
Write a Lewis formula for each of the following, assuming that the octet rule holds for the atoms. Then obtain the formal charges of the atoms. a. ClNO b. POCl3 c. N2O (NNO)
-
Coherent states are potentially an interesting basis to consider in which to express states on the Hilbert space. Are they a good basis, satisfying qualities that we desire of a basis on the Hilbert...
-
In this chapter, we had only expressed eigenstates of the harmonic oscillator Hamiltonian through repeated action of the raising operator, \(\hat{a}^{\dagger}\). This gives us a concrete algorithm...
-
A simple model for radioactive decay of an unstable nuclear isotope is as follows.3 Consider the potential illustrated in Fig. 7.9, in which there is a hard, infinite barrier at the spatial origin, a...

Study smarter with the SolutionInn App


