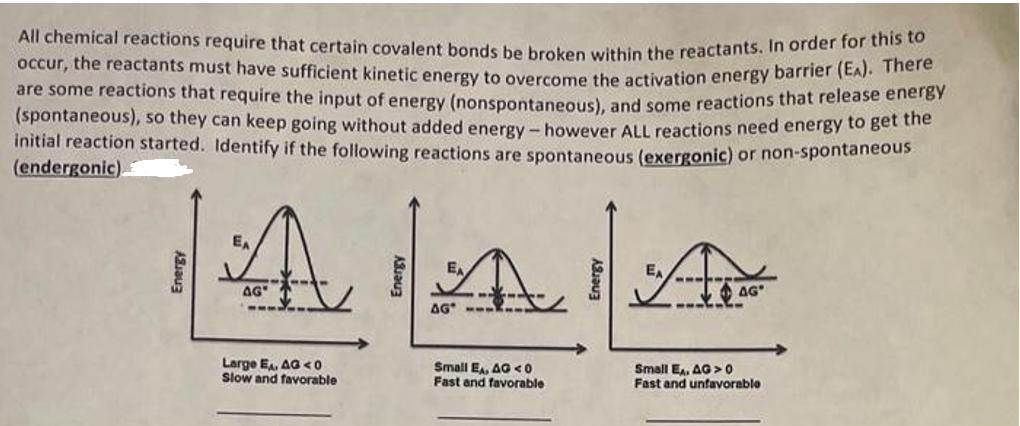
All chemical reactions require that certain covalent bonds be broken within the reactants. In order for...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
All chemical reactions require that certain covalent bonds be broken within the reactants. In order for this to occur, the reactants must have sufficient kinetic energy to overcome the activation energy barrier (EA). There are some reactions that require the input of energy (nonspontaneous), and some reactions that release energy (spontaneous), so they can keep going without added energy - however ALL reactions need energy to get the initial reaction started. Identify if the following reactions are spontaneous (exergonic) or non-spontaneous (endergonic) Energy A AG" Energy ---, AA AG Large E, AG<0 Slow and favorable Small EA, AG40 Fast and favorable Small EA, AG>0 Fast and unfavorable All chemical reactions require that certain covalent bonds be broken within the reactants. In order for this to occur, the reactants must have sufficient kinetic energy to overcome the activation energy barrier (EA). There are some reactions that require the input of energy (nonspontaneous), and some reactions that release energy (spontaneous), so they can keep going without added energy - however ALL reactions need energy to get the initial reaction started. Identify if the following reactions are spontaneous (exergonic) or non-spontaneous (endergonic) Energy A AG" Energy ---, AA AG Large E, AG<0 Slow and favorable Small EA, AG40 Fast and favorable Small EA, AG>0 Fast and unfavorable
Expert Answer:
Answer rating: 100% (QA)
Solution Step1 1 The first reaction with large activation energy EA and negative G ie G 0 is spontan... View the full answer

Related Book For 

Physics
ISBN: 978-0077339685
2nd edition
Authors: Alan Giambattista, Betty Richardson, Robert Richardson
Posted Date:
Students also viewed these biology questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Suppose you come from a part of the world that is blessed with abundant water. Demand never comes close to the available amount. Should you be careful about the amount you use or should you simply...
-
A piece of lead has the shape of a hockey puck, with a diameter of 7.5 cm and a height of 2.5 cm. If the puck is placed in a mercury bath, it floats. How deep below the surface of the mercury is the...
-
Standard addition graph. Tooth enamel consists mainly of the mineral calcium hydroxyapatite, Ca10(PO4)6(OH)2. Trace elements in teeth of archeological specimens provide anthropologists with clues...
-
A food processor claims that at most \(10 \%\) of her jars of instant coffee contain less coffee than claimed on the label. To test this claim, 16 jars of her instant coffee are randomly selected and...
-
The U.S. government subsidizes flood insurance because those who want to buy it live in the flood plain and cannot get it at reasonable rates. What inefficiency does this create?
-
The accountant at Roland Industries provides you with the following information for the first quarter: Direct labor costs Direct materials inventory, January 1 Direct materials inventory, March 31...
-
Define the following for your project: the objective, constraints, resources, the budget, and the schedule. in addition, discuss the matrix style you would prefer to use for your project and why this...
-
What's the partial pressure of carbon dioxide in a container that holds 5 moles of carbon dioxide, 3 moles of nitrogen, and 1 mole of hydrogen and has a total pressure of 1.05 atm?
-
What is the molar solubility ( in terms of mol / L ) of SrF 2 in pure water? Note the Ksp of SrF 2 is 2 . 6 x 1 0 - 1 0 Molar solubility of SrF 2 = 1 . 9 x 1 0 - 3 MMolar solubility of SrF 2 = 8 . 7...
-
Write the net ionic equation for the equilibrium that is established when ammonium iodide is dissolved in water. (Use H3O+ instead of H+. It is not necessary to include states such as (aq) or (!).) +...
-
You measure the amount of carbon-14 (the parent isotope) and nitrogen-14 (the daughter isotope) in a sample of bone and determine that 0.75 half-lives have elapsed for carbon-14. What is the age of...
-
Does a cation gain protons to form a positive charge or does it lose electrons? a) The protons in the nucleus of a cation formed due to the loss of protons and electrons. b) The cation gains protons...
-
X Limited and Y Limited agreed to amalgamate and form in your company called Limited. The balance sheet on the date of amalgamation was as under: X Co. Liabilities Equity share capital Shares of 100...
-
Consider the sections of two circuits illustrated above. Select True or False for all statements.After connecting a and b to a battery, the voltage across R1 always equals the voltage across R2.Rcd...
-
Use dimensional analysis to determine how the period T of a swinging pendulum (the elapsed time for a complete cycle of motion) depends on some, or all, of these properties: the length L of the...
-
A compass is placed directly on top of a wire (needle not shown). The current in the wire flows to the right. Which way does the north end of the needle point? Explain. (Ignore Earth's magnetic...
-
A long straight wire carries a 4.70-A current in the positive x -direction. At a particular instant, an electron moving at 1.00 Ã 107 m/s in the positive y -direction is 0.120 m from the wire....
-
Using an economic balance sheet, which of the Laws current financial assets is most concerning from an asset allocation perspective? A. Equities B. Real estate C. Fixed income Raye uses a costbenefit...
-
Using the economic balance sheet approach, the Laws economic net worth is closest to: A. $925,000. B. $1,425,000. C. $1,675,000. Raye uses a costbenefit approach to rebalancing and recommends that...
-
Raye believes the previous advisers asset class specifications for equity and derivatives are inappropriate given that, for purposes of asset allocation, asset classes should be: A. diversifying. B....

Study smarter with the SolutionInn App


