An 1865-kg airplane starts at rest on an airport runway at sea level. What is the...
Fantastic news! We've Found the answer you've been seeking!
Question:
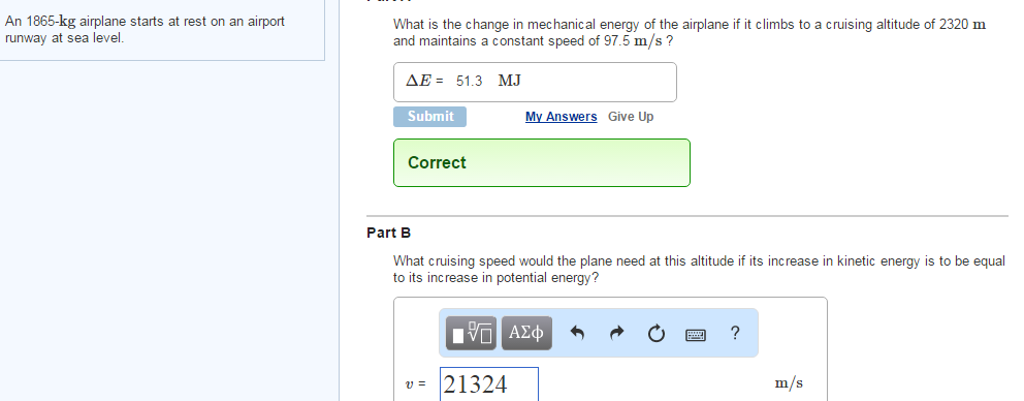
Transcribed Image Text:
An 1865-kg airplane starts at rest on an airport runway at sea level. What is the change in mechanical energy of the airplane if it climbs to a cruising altitude of 2320 m and maintains a constant speed of 97.5 m/s? ΔΕ= 51.3 MJ Submit Correct My Answers Give Up Part B What cruising speed would the plane need at this altitude if its increase in kinetic energy is to be equal to its increase in potential energy? 1Π ΑΣΦ v=21324 ? m/s An 1865-kg airplane starts at rest on an airport runway at sea level. What is the change in mechanical energy of the airplane if it climbs to a cruising altitude of 2320 m and maintains a constant speed of 97.5 m/s? ΔΕ= 51.3 MJ Submit Correct My Answers Give Up Part B What cruising speed would the plane need at this altitude if its increase in kinetic energy is to be equal to its increase in potential energy? 1Π ΑΣΦ v=21324 ? m/s
Expert Answer:
Related Book For 

Thermodynamics An Interactive Approach
ISBN: 978-0130351173
1st edition
Authors: Subrata Bhattacharjee
Posted Date:
Students also viewed these physics questions
-
What is the change in wavelength when monochromatic X-rays are scattered by electrons ons through an angle of 30?
-
What is the change in the atomic mass number for each of the reactions in the preceding two questions?
-
What is the change in internal energy of a car if you put 12.0 gal of gasoline into its tank? The energy content of gasoline is 1.3 108 J/gal. All other factors, such as the car's temperature, are...
-
If A = -2 6 1 -7 1 then det (A) = an and A-1 =
-
Discuss the concept of concurrent design. What are the advantages of this approach? How would you apply concurrent design to a group project?
-
Give a schematic figure, in the style of Figure 11.13, showing the heights of subtrees during a deletion operation in an AVL tree that triggers a trinode restructuring for the case in which the two...
-
Construct an environmental profile for a company of your choice. How might the profile be of use to management?
-
Kotari Inc. found that about 15 percent of its sales during the month were for cash. Kotari has the following accounts receivable payment experience: Percent paid in the month of sale ..........20...
-
Do you believe that Gap's apparel is out of touch with todays generation? How do you think they can improve it?
-
Table 1 shows Apple's online orders for the last week. When shoppers place an online order, several "recommended products" (upsells) are shown as at checkout an attempt to upsell See table 2 in cell...
-
Munoz Company manufactures two products. The budgeted per-unit contribution margin for each product follows: Sales price Variable cost per unit Super $ 94 (56) Supreme $125 (93) Contribution margin...
-
On January 1, 2015, Fong Co. sells property for which it had paid NT$690,000 to Chou Company, receiving in return Chous zero-interest-bearing note for NT$1,000,000 payable in 5 years. What entry...
-
What is the major difference in accounting for errors under IFRS versus U.S. GAAP?
-
At December 31, 2015, Ashley Co. has outstanding purchase commitments for purchase of 150,000 gallons, at 6.20 per gallon, of a raw material to be used in its manufacturing process. The company...
-
Under what circumstances is it appropriate to record goodwill in the accounts? How should goodwill, properly recorded on the books, be written off in order to conform with IFRS?
-
Manchester United, Inc. sold 10,000 season tickets at 2,000 each. By December 31, 2015, 8 of the 20 home games had been played. What amount should be reported as a current liability at December 31,...
-
Name few strategic plans for Emirates airlines. Which is strategic plan for Emirates airlines to know when and where globally to roll out its popular services. What are its popular services?
-
Write each fraction as a percent. 7 50
-
A vertical piston-cylinder assembly contains 10 L of air at 20oC. The cylinder has an internal diameter of 20 cm. The piston is 2 cm thick and is made of steel of density 7830 kg/m3. If the...
-
A 25 kg aluminum block initially at 225oC is brought into contact with a 25 kg block of iron at 150oC in an insulating enclosure. Determine. (a) The equilibrium temperature. (b) And the total entropy...
-
A rocket nozzle has an exit-to-throat area ratio of 4.0 and a throat area of 100 cm2. The exhaust gases are generated in a combustion chamber with stagnation pressure equal to 4 MPa and stagnation...
-
Prepare the journal entries for Kingston Dance Supplies, assuming that Kingston Dance Supplies uses a perpetual inventory system. The HST rate is 13 percent in Ontario. Explanations are not required....
-
Record the following note payable transactions of Lambda Company in the company's general journal. Explanations are not required. 2019 Jun. 1 Purchased delivery truck costing $86,000 by issuing a...
-
Suppose Detweiler Technologies borrowed \$2,000,000 on December 31, 2016, by issuing 4 percent long-term debt that must be paid in four equal annual instalments plus interest on the outstanding...

Study smarter with the SolutionInn App



