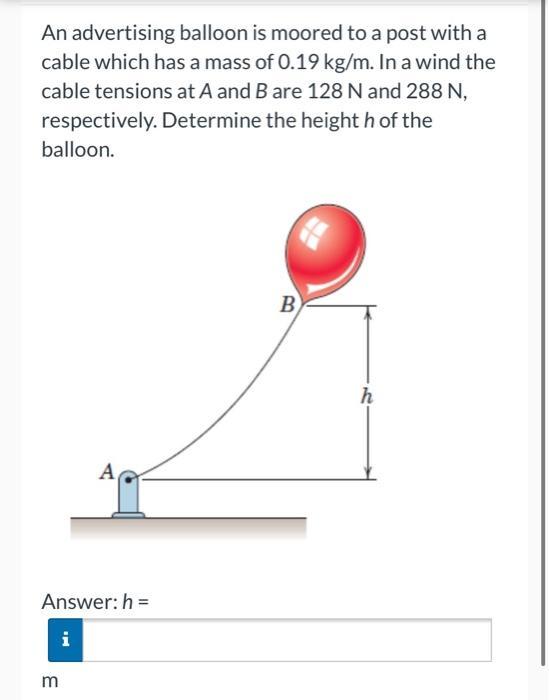
An advertising balloon is moored to a post with a cable which has a mass of...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
An advertising balloon is moored to a post with a cable which has a mass of 0.19 kg/m. In a wind the cable tensions at A and B are 128 N and 288 N, respectively. Determine the height h of the balloon. Answer: h= m A IM B h An advertising balloon is moored to a post with a cable which has a mass of 0.19 kg/m. In a wind the cable tensions at A and B are 128 N and 288 N, respectively. Determine the height h of the balloon. Answer: h= m A IM B h
Expert Answer:
Answer rating: 100% (QA)
Solution We have given Mass of the cable per unit length ... View the full answer

Related Book For 

Posted Date:
Students also viewed these physics questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The Fashion Rack has a monthly accounting period. The firms chart of accounts is shown below and on the next page. The journals used to record transactions are the sales journal, purchases journal,...
-
A cable with a restrainer at the bottom hangs vertically from its upper end (see figure). The cable has an effective cross-sectional area A = 40 mm2 and an effective modulus of elasticity E = 130...
-
Solve Loe 4 (a) x (b) fo e-y/2 dy 8 X 1+x dx -6 1 ( c ) = = 14 dx (c) - 3 x -
-
In which section of the income statement would a credit balance in Cash Short and Over be reported?
-
European Imports is an importer of silver, brass, and furniture items from Spain. Eloise Stiles is the general manager of European Imports. Stiles employs two other people in the business. Michelle...
-
The following figures relate to the retail business of J Clarke for the month of May 19X9. Goods which are on sale fall into two categories, A and B. Calculate for each category: Cost of goods sold...
-
1. Although the case with OFCCP is closed, we wonder if there are any less discriminatory explanations possible for why our women sales reps on average earned less than men. If so, what are they? 2....
-
Don's landscaping service purchased a backhoe for $12,000. It has a useful life of 5 years and a residual value of $1750. Based on this information, what is the straight-line depreciation for one...
-
Rina's Performance Pizza is a small restaurant in Denver that sells gluten-free pizzas. Rina's very tiny kitchen has barely enough room for the three ovens in which her workers bake the pizzas. Rina...
-
What are the fundamental functions of the nucleus in a eukaryotic cell?
-
A factory has two service departments P and Q and three production departments A, B, and C. You are supplied with the following information:Expenses of Service Departments P and Q are apportioned as...
-
The first model is the Rationalist Economics, and the second is the Behavioral Economics. Behavioral economists point out how the emotion interferes with our financial decision making. Crash of 2008...
-
Given the definition of the Haskell function len in this chapter, prove by induction the following: Consider the following (conect, but inefficient) Haskell implementation of the familiar Fibonacci...
-
What are the pros and cons of implementing the just-in-time inventory system. Do the pros outweigh the cons?
-
The required rate of return for AT&T, Inc (T) is 14.5%. Its expected ROE is 17.5% and its expected EPS is $2.5. If the firm's retention ratio is 30%, calculate its justified trailing and forward P/E...
-
Seth Erkenbeck, a recent college graduate, has just completed the basic format to be used in preparing the statement of cash flows (indirect method) for ATM Software Developers. All amounts are in...
-
During the month, services performed for customers on account amounted to $7,500 and collections from customers in payment of their accounts totaled $6,000. At the end of the month, the Accounts...
-
Phosphorous acid, H3PO3, has the structure (HO)2PHO, in which one H atom is bonded to the P atom, and two H atoms are bonded to O atoms. For each bond to an H atom, decide whether it is polar or...
-
Identify each of the following as an acid or a base in terms of the Arrhenius concept. Give the chemical equation for the reaction of the substance with water, showing the origin of the acidity or...
-
Hydrogen peroxide, H2O2, is a colorless liquid. A concentrated solution of it is used as a source of oxygen for rocket propellant fuels. Dilute aqueous solutions are used as a bleach. Analysis of a...
-
Using the Internet, find some universities that offer forensic accounting courses.
-
On the Internet find the KPMG India Fraud Survey Report, 2002. a. How many chief executive officers re- ceived the questionnaire? b. How many of the respondents do not have a written fraud policy? c....
-
Using the Internet, locate and review these sites: a. National Fraud Center b. The Fraud Detectives Consultant Network c. Milberg Weiss Bershad Hynes & Lerach d. Mark R. Simmons e. Communications...

Study smarter with the SolutionInn App


