An aqueous solution of silver nitrate reacts completely with an aqueous solution of sodium phosphate to...
Fantastic news! We've Found the answer you've been seeking!
Question:
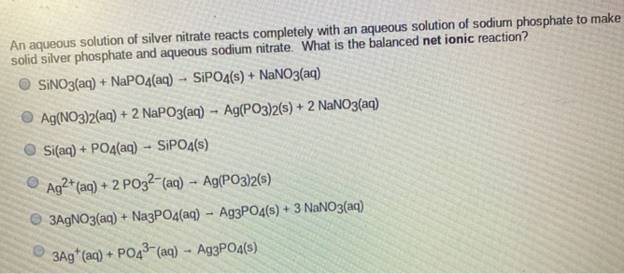
Transcribed Image Text:
An aqueous solution of silver nitrate reacts completely with an aqueous solution of sodium phosphate to make solid silver phosphate and aqueous sodium nitrate. What is the balanced net ionic reaction? SINO3(aq) + NAPO4(aq) - SIPO4(s) + NANO3(aq) Ag(NO3)2(aq) + 2 NAPO3(aq) - Ag(P03)2(s) + 2 NaNO3(aq) Si(aq) + PO4(aq) - SIPO4(s) Ag²* (aq) + 2 PO32-(aq) – Ag(PO3)2(s) O 3AGNO3(aq) + NazPO4(aq) - A93PO4(s) + 3 NANO3(aq) 3Ag (aq) + PO43 (aq) - Ag3PO4(s) An aqueous solution of silver nitrate reacts completely with an aqueous solution of sodium phosphate to make solid silver phosphate and aqueous sodium nitrate. What is the balanced net ionic reaction? SINO3(aq) + NAPO4(aq) - SIPO4(s) + NANO3(aq) Ag(NO3)2(aq) + 2 NAPO3(aq) - Ag(P03)2(s) + 2 NaNO3(aq) Si(aq) + PO4(aq) - SIPO4(s) Ag²* (aq) + 2 PO32-(aq) – Ag(PO3)2(s) O 3AGNO3(aq) + NazPO4(aq) - A93PO4(s) + 3 NANO3(aq) 3Ag (aq) + PO43 (aq) - Ag3PO4(s)
Expert Answer:
Answer rating: 100% (QA)
3 Agt 3N Firstly we sw the paumula silvere nitrgle so dium phasphate silver phasphate Sodium ... View the full answer

Related Book For 

Cambridge International AS And A Level Chemistry Coursebook
ISBN: 9781316637739
2nd Edition
Authors: Lawrie Ryan, Roger Norris
Posted Date:
Students also viewed these accounting questions
-
An aqueous solution of ammonium chloride and barium hydroxide is heated, and the compounds react to give off ammonia gas. Barium chloride solution and water are also products. Write a balanced...
-
An aqueous solution of ammonium nitrite, NH4NO2, decomposes when heated to give off nitrogen, N2. NH4NO2(s) 2H2O(g) + N2(g) This reaction may be used to prepare pure nitrogen. How many grams of...
-
An aqueous solution of a molecular compound freezes at -0.086C. What is the molality of the solution?
-
George operates a business that generated adjusted gross income of $250,000 and taxable income of $170,000 this year (before the domestic production activities deduction). Included in income was...
-
One member of the consumer group in Exercise 15 is concerned that the experiment will take too long and makes some suggestions to shorten it. Comment briefly on each idea. a) Cut the runs to 8 by...
-
Garbage trucks entering a particular waste-management facility are weighed prior to offloading their contents. Let X = the total processing time for a randomly selected truck at this facility...
-
Construct a bar graph for each of the following (assume the independent variable is Group and the dependent variable is time): a. \(\operatorname{Group} \mathrm{A}(N=21, M=14.05, s=3.63)\); Group B...
-
Breakeven point, what-if analysis Premier Products, Inc., is considering replacing its existing machine with a new and faster machine that will produce a more reliable product and will turn around...
-
Consolidated Balance Sheet Working Paper, Bargain Purchase On January 1, 2022, Paxon Corporation acquired all of the outstanding common stock of Saxon Company for $2 billion cash. The prior to the...
-
We want to design a tension spring with a spring outer diameter of 32 mm. The range of tensile load is 110 160 N, and the deformation range is 140~ 170 mm. We are going to use SWO-B as the material....
-
A fruit fly population has a gene with two alleles, Al and A2. Tests show that 70% of the gametes produced in the population contain the Al allele. If the population is in Hardy-Weinberg equilibrium,...
-
Problem 3. Suma Company has several branches all over the country. At year end, December 31, 2014 a reconciliation is being made. Before taking the effects of following information Galema Branch...
-
Hart Company sold 5,000 units for a price of $50 per unit and had the following information: Variable expenses Fixed expenses Break-even point (in dollars) $160,000 $125,000 $347,222 If the sales...
-
Prepare the following adjusting journal entry for the Wedding R Us Company: Weddings R Us Company Unadjusted Trial Balance August 31, 2023 Debit Credit Cash 124,880 Accounts Receivable 37,460 Baking...
-
3) What is the WEEKLY breakeven sales level for a clothing store that is experiencing the following costs: Rent $20,000/month; Municipal Taxes $4,000/month; Utilities $2000/month Garbage $400/month...
-
On 1/1/2022, the company purchased a car (Mazda CX9, year 2013, serial number ANK#34039) for the sales manager for $23,000. Due to the amount of travelling, the car is expected to have a useful life...
-
Raytracing is an algorithm that involves finding the point at which a ray (a line with a direction and an origin) intersects a curve or surface. We will consider a ray intersecting with an ellipse....
-
Refrigerant R-12 at 30C, 0.75 MPa enters a steady flow device and exits at 30C, 100 kPa. Assume the process is isothermal and reversible. Find the change in availability of the refrigerant.
-
a. For each pair of compounds, suggest which will have the most exothermic lattice energy. i. KCl and BaO (ionic radii are similar) ii MgI 2 and SrI 2 iii CaO and NaCl (ionic radii are similar). b....
-
a. One of the buffers in blood plasma is a mixture of dihydrogenphosphate ions (H 2 PO 4 ) and hydrogenphosphate (HPO 4 2 ) ions. i. Identify the conjugate acid and base in this buffer. ii. Write a...
-
Give the formulae of: a. Sodium chlorate(I) b. Iron(III) oxide c. Potassium nitrate(III) d. Phosphorus(III) chloride.
-
Draw a cash flow diagram of any investment that exhibits both of the following properties: 1. The investment has a 4-year life. 2. The investment has a 10 percent/year internal rate of return.
-
What is the internal rate of return of the following cash flow diagram? a. 20 percent b. 18.2 percent c. 17.5 percent d. 15 percent $30 $31 0 1 2 3 $30 $15
-
Quilts R Us (QRU) is considering investing in a new patterning attachment with the cash flow profile shown in the table below. QRU's MARR is 13.5 percent/year. a. What is the internal rate of return...

Study smarter with the SolutionInn App


