An ideal Bose gas undergoes Bose-Einstein condensation with Bose- temperature TB. The variation of number of...
Fantastic news! We've Found the answer you've been seeking!
Question:
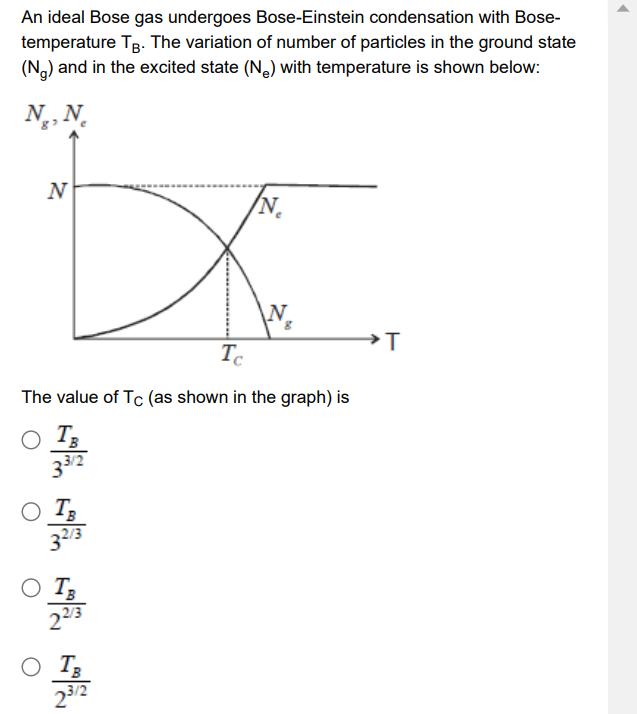
Transcribed Image Text:
An ideal Bose gas undergoes Bose-Einstein condensation with Bose- temperature TB. The variation of number of particles in the ground state (Ng) and in the excited state (Ne) with temperature is shown below: N., N. N DXC N Tc The value of Tc (as shown in the graph) is N N N 23/2 T An ideal Bose gas undergoes Bose-Einstein condensation with Bose- temperature TB. The variation of number of particles in the ground state (Ng) and in the excited state (Ne) with temperature is shown below: N., N. N DXC N Tc The value of Tc (as shown in the graph) is N N N 23/2 T
Expert Answer:
Related Book For 

Thermodynamics for Engineers
ISBN: ?978-1133112860
1st edition
Authors: Kenneth A. Kroos, Merle C. Potter
Posted Date:
Students also viewed these physics questions
-
A statistics quiz consists of true-false questions with the answers listed as shown. a. Examine the sequence and try to identify a pattern showing that the answers are not random. b. What do you...
-
An ideal gas undergoes a process from state 1 to state 2. For the following data, approximate the change in specific entropy for this process using Eq. 7.13. State T(C) P (kPa) 1 200 2 180 100 150...
-
An ideal gas undergoes a process from state 1 to state 2. For the data shown below, approximate the change in specific entropy for this process using Eq. 7.11. (ft lbm) 4.5 3.2 State T(F) P (psia) 1...
-
In Exercises 116, evaluate each algebraic expression for the given value or values of the variable(s). x 2 - 4(x - y), for x = 8 and y = 3
-
The distance between the Li+ and Cl ions in LiCl is 0.257 nm. Use this and the molecular mass of LiCl (42.4 g/mol) to compute the density of LiCl.
-
Using an Excel spreadsheet: PolarTech Inc. is considering producing a new widget. The production equipment, costing $12 million, will depreciate over a six-year period. Upon full depreciation, the...
-
A pipeline containing ammonia gas is vented to ambient air via a \(20-\mathrm{m}\) long, \(3-\mathrm{mm}\) diameter tube. What is the mass flow ( \(\mathrm{g} /\) day) of ammonia into the atmosphere?...
-
Trim Corporation acquired 100 percent of Round Corporations voting common stock on January 1, 20X2, for $400,000. At that date, the book values and fair values of Rounds assets and liabilities were...
-
I think that university students are more likely to be registered to vote than the normal population. I sample 8 university students to determine whether or not they are registered to vote. In the...
-
You are given the following information concerning four stocks: a) Using 20X0 as the base year, construct three aggregate measures of the market that simulate the Dow Jones Industrial Average, the...
-
Read the caliper and read the measurement to the significant figure 51 31 Auto
-
Green sells its $ 1 2 4 , 0 0 0 equity in a partnership to Tan for $ 9 5 , 5 0 0 . Prepare the partnership\'s journal entry to record the sale of Green\'s interest to Tan.
-
discuss the implications of pleiotropy and genetic correlation on the evolution of complex phenotypes, and how these factors impact the trade-offs among different traits within an organism ?
-
How to solve step by step these questions related to contribution margin? For the Sochi Olympics, Hudson's Bay featured maple leaf mittens. If the variable costs were $5 per pair and it made a 70%...
-
You are doing a group assignment. Your teammate Sonia does not attend your first team meeting. What should you do? Email Sonia and say, "Hi Sonia, you missed the meeting. Don't let it happen again."...
-
How does phenotypic plasticity enable organisms to adapt to fluctuating environmental conditions, and what are the molecular and genetic mechanisms that underlie this plasticity ?
-
During the year ended December 31, 2021, Kelly's Camera Shop had sales revenue of $170,000, of which $85,000 was on credit. At the start of 2021, Accounts Receivable showed a $10,000 debit balance...
-
On October 31 Juanita Ortega, owner of Outback Guide Service, received a bank statement dated October 30. Juanita found the following: 1. The checkbook has a balance of $2,551.34. 2. The bank...
-
A large air-conditioning unit processes 5 kg/s of air. The air enters with a dry-bulb temperature of 35 8C and a wet-bulb temperature of 25 8C. How much condensate is being removed each hour if the...
-
An inventor proposes an engine that utilizes the boiling water of a hot spring and suggests it can operate with an efficiency of 22% in an area where a local stream has an average temperature of 12C....
-
An automobile with a mass of 2500 lbm is traveling at 60 mph. What is the kinetic energy of the car?
-
In input or factor markets, the demand for an input is a(n) _________ demand _________ from consumers demand for the good or service.
-
At a wage below the equilibrium level, quantity _________ would exceed quantity _________, resulting in a labor _________. In this situation, employers would be forced to offer higher wages to hire...
-
The demand for labor is determined by its _________, which is the additional revenue that a firm obtains from one more unit of input.

Study smarter with the SolutionInn App



