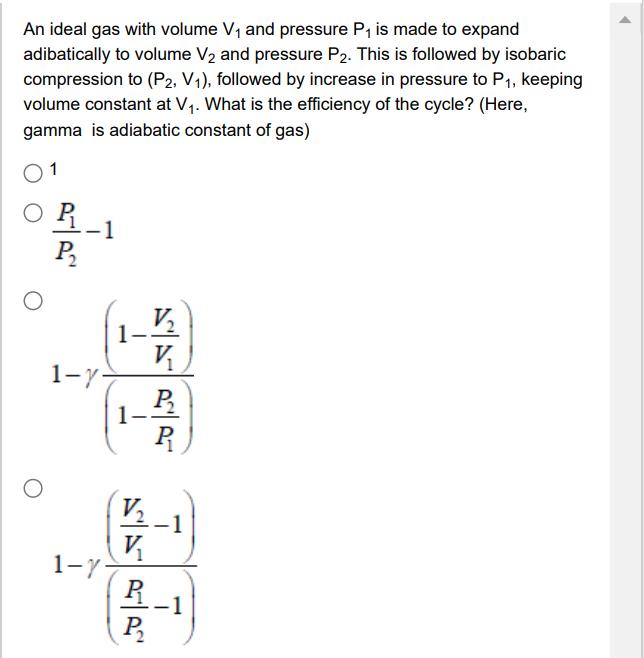
An ideal gas with volume V and pressure P is made to expand adibatically to volume...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
An ideal gas with volume V and pressure P is made to expand adibatically to volume V and pressure P2. This is followed by isobaric compression to (P2, V1), followed by increase in pressure to P, keeping volume constant at V. What is the efficiency of the cycle? (Here, gamma is adiabatic constant of gas) 1 P 1-y- 1-7- 1- V V V V R P P R -1 An ideal gas with volume V and pressure P is made to expand adibatically to volume V and pressure P2. This is followed by isobaric compression to (P2, V1), followed by increase in pressure to P, keeping volume constant at V. What is the efficiency of the cycle? (Here, gamma is adiabatic constant of gas) 1 P 1-y- 1-7- 1- V V V V R P P R -1
Expert Answer:
Related Book For 

Introduction To Chemical Engineering Thermodynamics
ISBN: 9781259696527
8th Edition
Authors: J.M. Smith, Hendrick Van Ness, Michael Abbott, Mark Swihart
Posted Date:
Students also viewed these physics questions
-
An ideal gas with constant heat capacities undergoes a change of state from conditions T1, P1 to conditions T2, P2. Determine (H (J mol-1) and (S (J mol-1 K-1) for one of the following cases. (a) T1...
-
An ideal gas with the adiabatic exponent y goes through a process p = Po- aV, where Po and a are positive constants, and V is the volume. At what volume will the gas entropy have the maximum value?
-
An ideal gas with the adiabatic exponent undergoes a process in which its internal energy relates to the volume as U = aV, where a and a are constants. Find: (a) The work performed by the gas and the...
-
A solid of constant density is bounded below by the plane z = 0, on the sides by the elliptical cylinder x 2 + 4y 2 = 4, and above by the plane z = 2 - x. a. Find x and y. b. Evaluate the integral...
-
A series RLC circuit with L = 10 mH, C = 2 F, and R = 5 is driven by a generator with a maximum emf of 100 V and a variable angular frequency . Find (a) The resonant frequency 0 and (b) Irms at...
-
Your client, Cookie, Inc., has asked you to prepare select portions of its 2023 Federal tax return (Form 1120). In order to prepare the 2023 tax return, you must correctly reconcile Cookie's...
-
Which component of the rate of return on investment (profit margin or investment turnover) focuses on efficiency in the use of assets and indicates the rate at which sales are generated for each...
-
Total June 2010 sales for Roys Catering are expected to be $450,000. Of each months sales, 80 percent is expected to be on credit. The Accounts Receivable balance at May 31 is $119,600; of which...
-
Brewers Company agrees to lend Cubs Company $3,480,000 on September 1, 2023. Cubs Company signs a $3,480,000, 5%, 36-month note. Assume Cubs Company accrues interest monthly and that all interest is...
-
Two companies competing in the same industry are being evaluated by a bank that can lend money to only one of them. Summary information from the financial statements of the two companies follows:...
-
XYZ Corp. is preparing its year end for December 31, 2016.An inventory count was performed correctly at December 31, 2016 and all entries were made to record the correct amount of inventory at Dec...
-
The Muffin House is an industrial bakery in Ogunquit, Maine. They produce baked goods in large batches. For each batch they record a range of information. A sample of this information is provided in...
-
A single-degree-of-freedom system experiences the periodic excitation in Fig. 1. The excitation is constant for time intervals of duration T/2 followed by time intervals also of duration T/2 during...
-
Supposer une obligation du gouvernement du Canada aux caractristiques suivantes: livraison : 16 mars 2024 avec l'intrt couru chance : 1e septembre 2033 4 taux du coupon (nominal) : 10% valeur...
-
White Manufacturings 2008 organizational structure is seemingly a depiction of a Project Management Office (PMO). Discuss the primary functions of a PMO as well as the four (4) ways in which the PMO...
-
Clear Restorations purchased a floor machine for $10,520. Delivery costs totaled $1,250. The salvage value is $1,350 after 8 years. Using the straight-line depreciation method, calculate the book...
-
Salsa Company is considering an investment in technology to improve its operations. The investment cos $248,000 and will yield the following net cash flows. Management requires a 10% return on...
-
As economic conditions change, how do banks adjust their asset portfolio?
-
If a liquid solution of HCl in water, containing 1 mol of HCl and 4.5 mol of H 2 O, absorbs an additional 1 mol of HCl(g) at a constant temperature of 25C, what is the heat effect?
-
If the heat capacity of a substance is correctly represented by an equation of the form, C P =A+BT+CT 2 show that the error resulting when C P H is assumed equal to C P evaluated at the arithmetic...
-
Consider a binary mixture for which the excess Gibbs energy is given by G E /RT = 2.6x 1 x 2 . For each of the following overall compositions, determine whether one or two liquid phases will be...
-
The following information is taken from the Fossil, Inc. 2015 annual report: Calculate Fossils actual and sustainable rate of growth in sales. How do the two rates of growth compare? What advice...
-
Presented below are selected financial data from the 2015 annual report of the Bristol-Myers Squibb Company: Required Using the ratio definitions from Exhibit 4.6, calculate the financial ratios for...
-
Presented below are selected financial data from the 2015 annual report of The Boeing Company: Required Using the ratio definitions from Exhibit 4.6, calculate the financial ratios for The Boeing...

Study smarter with the SolutionInn App



