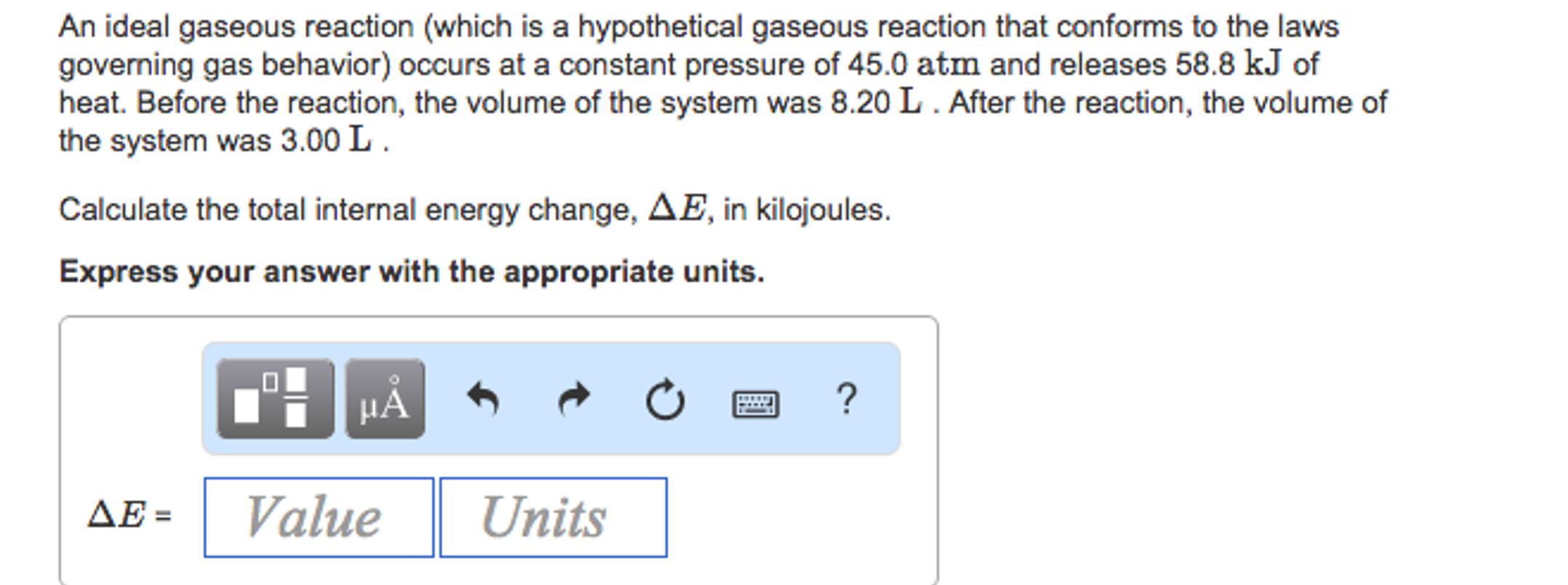
An ideal gaseous reaction (which is a hypothetical gaseous reaction that conforms to the laws governing...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
An ideal gaseous reaction (which is a hypothetical gaseous reaction that conforms to the laws governing gas behavior) occurs at a constant pressure of 45.0 atm and releases 58.8 kJ of heat. Before the reaction, the volume of the system was 8.20 L. After the reaction, the volume of the system was 3.00 L. Calculate the total internal energy change, AE, in kilojoules. Express your answer with the appropriate units. μÅ ΔΕΞ Value Units ? An ideal gaseous reaction (which is a hypothetical gaseous reaction that conforms to the laws governing gas behavior) occurs at a constant pressure of 45.0 atm and releases 58.8 kJ of heat. Before the reaction, the volume of the system was 8.20 L. After the reaction, the volume of the system was 3.00 L. Calculate the total internal energy change, AE, in kilojoules. Express your answer with the appropriate units. μÅ ΔΕΞ Value Units ?
Expert Answer:
Answer rating: 100% (QA)
Internal energy formula is AU Q W Where Q heat transferred to t... View the full answer

Related Book For 

Fundamentals Of Financial Management
ISBN: 9780273713630
13th Revised Edition
Authors: James Van Horne, John Wachowicz
Posted Date:
Students also viewed these accounting questions
-
An ideal gas is heated at a constant pressure of 2.0 105 Pa from a temperature of - 73C to a temperature of +27C. The initial volume of the gas is 0.10 m3. The heat energy supplied to the gas in...
-
A gas is compressed at a constant pressure of 0.800 atm from 9.00 L to 2.00 L. In the process, 400 J of energy leaves the gas by heat. (a) What is the work done on the gas? (b) What is the change in...
-
An ideal gas is heated at a constant pressure of 2.0 10 5 Pa from a temperature of 73C to a temperature of +27C. The initial volume of the gas is 0.10 m 3 . The heat energy supplied to the gas in...
-
Are the statements in (a) and (b) below saying the same thing? (Hint: think about the tradeoffs between concerns of intra- and intergenerational equity.) (a) Sustainability is principally about...
-
In the accompanying tables you will find consolidated balance sheets for the commercial banking system and the 12 Federal Reserve Banks. Use columns 1 through 3 to indicate how the balance sheets...
-
The green division of Campana Company reported the following data for the current year: Sales...........................................$3,000,000 Variable...
-
The FBI computed the proportion of violent crimes in the United States falling into each of four categories. A simple random sample of 500 violent crimes committed in California were categorized in...
-
Design for Supply Chain (DfSC) is a systematic method of ensuring the best t between the design of a product throughout its lifetime and its supply chain members' resources and capabilities. Even...
-
On June 30, Sharper Corporation's stockholders' equity section of its balance sheet appears as follows before any stock dividend or split. Sharper declares and immediately distributes a 50% stock...
-
The names of the employees of Matson Office Systems and their regular salaries are shown in the following payroll register. Note that Wayne and Young are paid monthly on the last payday, while all...
-
A random sample of 10 college students was drawn from a large university. Their ages are 22, 17, 27, 20, 23, 21, 24, 21, 19, and 24 years. a. Use the critical value method to test whether we can...
-
Johnson's Pic has the following costs related to a new project:Marketing costs already spent: 1 5 , 0 0 0 1 5 , 0 0 0 Variable costs associated with the project: 7 2 , 5 0 0 7 2 , 5 0 0 Additional...
-
Solve. Assume all variables are positive. P T = -1, for R R R =
-
Interpret the results of the FMS test results. Are there any similarities between theresults?Whatis youroverallanalysis? Chooseandexplainthemostappropriate resistanceexercise...
-
Load distribution of a wing with finite span. If the load distribution on a finite-span wing is as shown, sketch the trailing vortex system, indicating the magnitude and direction of the trailing...
-
Baskin Company's net income last year was $98,000. Changes in the company's balance sheet accounts for the year appear below: Increases (Decreases) Debit balances: Cash $24,000 Accounts receivable...
-
Metal spheres A and B are identical except sphere A has a charge of -4.0nC while sphere B has a charge of +3.0nC. If the spheres are brought into contact, do electrons move from A to B or B to A? How...
-
MgO prevents premature evaporation of Al in a furnace by maintaining the aluminum as Al2O3. Another type of matrix modifier prevents loss of signal from the atom X that readily forms the molecular...
-
Does the dollar size of the invoice affect the percentage annual interest cost of not taking discounts? Illustrate with an example.
-
What are compensating bank balances, and why are they not the same for all depositors?
-
In choosing the composition of short-term financing, what factors should be considered?
-
What statutory and mandatory professional pronouncements require a review to be published?
-
List two items that may be found in the operating section and two items that may be found in the financial review section of an OFR.
-
What statutory and mandatory professional requirements require directors to submit a report to shareholders?

Study smarter with the SolutionInn App


