An insulated beaker with negligible mass contains liquid water with a mass of 0.245 kg and...
Fantastic news! We've Found the answer you've been seeking!
Question:
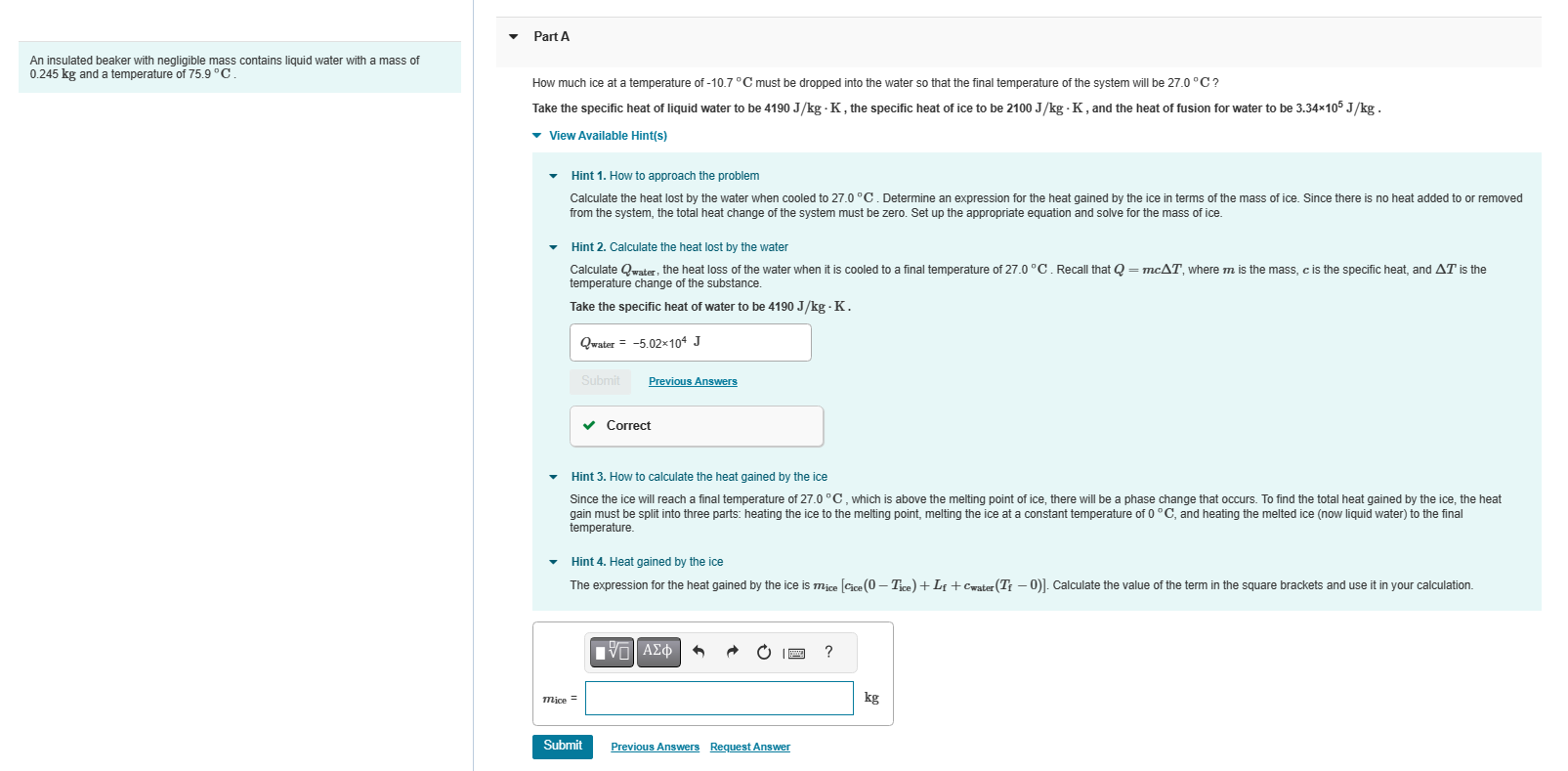
Transcribed Image Text:
An insulated beaker with negligible mass contains liquid water with a mass of 0.245 kg and a temperature of 75.9 C. Part A How much ice at a temperature of -10.7C must be dropped into the water so that the final temperature of the system will be 27.0 C? Take the specific heat of liquid water to be 4190 J/kg - K, the specific heat of ice to be 2100 J/kg - K, and the heat of fusion for water to be 3.34105 J/kg. View Available Hint(s) Hint 1. How to approach the problem Calculate the heat lost by the water when cooled to 27.0 C. Determine an expression for the heat gained by the ice in terms of the mass of ice. Since there is no heat added to or removed from the system, the total heat change of the system must be zero. Set up the appropriate equation and solve for the mass of ice. Hint 2. Calculate the heat lost by the water Calculate Qwater, the heat loss of the water when it is cooled to a final temperature of 27.0 C. Recall that Q = mcAT, where m is the mass, c is the specific heat, and AT is the temperature change of the substance. Take the specific heat of water to be 4190 J/kg. K. Qwater = -5.02104 J Submit Previous Answers Correct Hint 3. How to calculate the heat gained by the ice Since the ice will reach a final temperature of 27.0C, which is above the melting point of ice, there will be a phase change that occurs. To find the total heat gained by the ice, the heat gain must be split into three parts: heating the ice to the melting point, melting the ice at a constant temperature of 0 C, and heating the melted ice (now liquid water) to the final temperature. mice = Hint 4. Heat gained by the ice The expression for the heat gained by the ice is mice [Cice (0-Tice) + Lf+Cwater (Tf - 0)]. Calculate the value of the term in the square brackets and use it in your calculation. Submit IVE| Ima ? Previous Answers Request Answer kg An insulated beaker with negligible mass contains liquid water with a mass of 0.245 kg and a temperature of 75.9 C. Part A How much ice at a temperature of -10.7C must be dropped into the water so that the final temperature of the system will be 27.0 C? Take the specific heat of liquid water to be 4190 J/kg - K, the specific heat of ice to be 2100 J/kg - K, and the heat of fusion for water to be 3.34105 J/kg. View Available Hint(s) Hint 1. How to approach the problem Calculate the heat lost by the water when cooled to 27.0 C. Determine an expression for the heat gained by the ice in terms of the mass of ice. Since there is no heat added to or removed from the system, the total heat change of the system must be zero. Set up the appropriate equation and solve for the mass of ice. Hint 2. Calculate the heat lost by the water Calculate Qwater, the heat loss of the water when it is cooled to a final temperature of 27.0 C. Recall that Q = mcAT, where m is the mass, c is the specific heat, and AT is the temperature change of the substance. Take the specific heat of water to be 4190 J/kg. K. Qwater = -5.02104 J Submit Previous Answers Correct Hint 3. How to calculate the heat gained by the ice Since the ice will reach a final temperature of 27.0C, which is above the melting point of ice, there will be a phase change that occurs. To find the total heat gained by the ice, the heat gain must be split into three parts: heating the ice to the melting point, melting the ice at a constant temperature of 0 C, and heating the melted ice (now liquid water) to the final temperature. mice = Hint 4. Heat gained by the ice The expression for the heat gained by the ice is mice [Cice (0-Tice) + Lf+Cwater (Tf - 0)]. Calculate the value of the term in the square brackets and use it in your calculation. Submit IVE| Ima ? Previous Answers Request Answer kg
Expert Answer:
Answer rating: 100% (QA)
I see you have a question that involves a thermodynamics problem about mixing ice and water It seems like youre using an interface where some of the d... View the full answer

Related Book For 

University Physics with Modern Physics
ISBN: 978-0321696861
13th edition
Authors: Hugh D. Young, Roger A. Freedman, A. Lewis Ford
Posted Date:
Students also viewed these physics questions
-
An insulated beaker with negligible mass contains 0.250 kg of water at a temperature of 75.0C. How many kilograms of ice at temperature of -20.0C must be dropped into the water to make the final...
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
In Exercises 7681, find the domain of each function. g(x) = 4 x - 7
-
In estimating a regression based on monthly observations from January 1987 to December 2002 inclusive, you find that the coefficient on the independent variable is positive and significant at the...
-
A closed surface with dimensions a = b = 0.400 m and c = 0.600 m is located as in Figure P24.66. The left edge of the closed surface is located at position x = a. The electric field throughout the...
-
Use the relationship between force and impulse to explain how padded boxing gloves protect a boxer's hands.
-
Selected transactions for Alvarado Company during its first month in business are presented below. Sept. 1 Invested $10,000 cash in the business in exchange for common stock. 5 Purchased equipment...
-
6. 7. 8. 9. You are given a converging lens with equal radii of curvature and a diverging lens with the same radii of curvature as those of the converging lens. The lenses are made of material with n...
-
Identify 1 to 2 significant advancements in computers or computer systems in the last 10 to 15 years. Provide an example of how these advancements support different industries. How have these changes...
-
Problems 45-50 show scatter diagrams along with a best-fitting line. Match the best-fitting line in each with an appropriate equation named \(\mathrm{A}-\mathrm{H}\). A. \(y=0.5 x-4\) B. \(y=-0.5...
-
What do we mean by correlation?
-
Briefly explain the difference between these three approaches to preparing consolidated financial statements: a. Pooling-of-interests method b. Purchase method c. New-entity method
-
Nonprobability sampling assumes that some elements of the population have no chance of selection or the probability of selection can't be accurately determined. Some types of nonprobability sampling...
-
Suppose that the annual rainfall in Ferndale, California, is known to be normally distributed, with a mean of 35.5 inches and a standard deviation of 2.5 inches. About \(2.3 \%\) of the years, the...
-
One of the following is False: Select one: O Log(212) = Log(z1) + Log(z2). %3D O dlogz = z if z # 0. %3D O log(z12) = log(21) + log(z2). O loge = z + 2kni.
-
Why is inventory management important for merchandising and manufacturing firms and what are the main tradeoffs for firms in managing their inventory?
-
Two uniform solid spheres, each with mass and radius R = 0.0800 m, are connected by a short, light rod that is along a diameter of each sphere and are at rest on a horizontal tabletop. A spring with...
-
A resistor with R 1 = 25.0 is connected to a battery that has negligible internal resistance and electrical energy is dissipated R 1 by at a rate of 36.0 W. If a second resistor with R 2 = 15.0 is...
-
On Planet X, you drop a 25-kg stone from rest and measure its speed at various times. Then you use the data you obtained to construct a graph of its speed as a function of time t (Fig. P2.78). From...
-
Describe the role of a paralegal in a family law practice.
-
Define family law and identify the kinds of issues that are addressed in a family law practice.
-
Identify the kinds of resources included in a basic family law library.

Study smarter with the SolutionInn App


