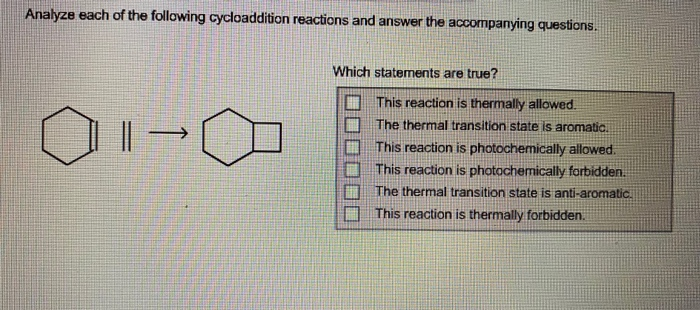
Analyze each of the following cycloaddition reactions and answer the accompanying questions. >> Which statements are...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Analyze each of the following cycloaddition reactions and answer the accompanying questions. →>> Which statements are true? This reaction is thermally allowed. The thermal transition state is aromatic. This reaction is photochemically allowed. This reaction is photochemically forbidden. The thermal transition state is anti-aromatic. This reaction is thermally forbidden. Analyze each of the following cycloaddition reactions and answer the accompanying questions. →>> Which statements are true? This reaction is thermally allowed. The thermal transition state is aromatic. This reaction is photochemically allowed. This reaction is photochemically forbidden. The thermal transition state is anti-aromatic. This reaction is thermally forbidden.
Expert Answer:
Answer rating: 100% (QA)
The correct choices are This reaction is photochemically ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Analyze each of the following scenarios. Provide a brief explanation of your analysis and answers to each one. a. A governmental unit acquired land, buildings, other improvements, and certain...
-
Each of the following reactions will be encountered at some point in this text. Classify each one according to whether the organic substrate is oxidized or reduced in the process. (a) (b) (c) (d) ...
-
Which reaction in each of the following pairs will take place more rapidly? a. b. c. d. e. CHHO CH,CHO CH OH I CHOH Br DMSO, CHOHBr EtOH CHzBr NH3 CH3NH3 + Br Et CH,NH,Br CHBNH EtOH, CH NH, + Br
-
Plot the six risks on a probability/impact matrix using the project impact matrix 2023 template. Write one to two sentences stating the rationale for how you determined the quadrant placement for...
-
If you were employed as a salesperson, but were asked to work from your home (as a telecommuter) on days that you were not out in the field, what would you like, and dislike, about this...
-
Which of the following is a Type II subsequent event? a. Bankruptcy of a customer subsequent to year-end, which would be considered when evaluating the adequacy of the allowance for uncollectible...
-
Top managers of Chase Financial Services are reviewing company per- formance for 2009. The income statement reports a 20% increase in net income over 2008. However, most of the net-income increase...
-
In 2013, Copia Company had current assets of $155,000 and current liabilities of $100,000, of which accounts payable were $65,000. Cost of goods sold was $425,000, merchandise inventory increased by...
-
3. Markov analysis The training program for production supervisors at a particular company consists of twophases. Phase 1, which involves 3 weeks of classroom work, is followed by phase 2, which is a...
-
A certain newspaper provides the net asset value, the year-to-date percent return, and the three-year percent return for 882 mutual funds at the end of 2017. Assume that a simple random sample of 12...
-
An investor is considering purchasing a property (unleveraged) that is expected to have annual net operating income as follows: Year 1: 100,000; Year 2: 105,000; Year 3: 110,000; Year 4: 115,000; and...
-
Why are operational and financial metrics important to good management?
-
What is the purpose of a trustee?
-
What do we call the quantity force distance, and what quantity does it change?
-
How do the cash management practices of large and small businesses differ?
-
What are the two primary factors that affect a loans interest rate?
-
Consider a damped oscillator with m = 0.2 kg, k = 100 N/m, and b = 5 N-s/m. The oscillator is driven by a force F= (1.6 N) cos 20r. (a) If the displacement is given by x = A cos(wt - 8), what are the...
-
What are the key dimensions of critical thinking 2. Watch the NBC Learn video on Diet Scams. What types of claims are made in this video Are they valid Elaborate on your responses. Discuss this video...
-
The alkene mixture obtained on dehydration of 2,2-dimethylcyclohexanol contains appreciable amounts of 1,2-dimethylcyclohexene. Give a mechanistic explanation for the formation of this product.
-
Write the structures of the enol forms of 2-butanone that react with chlorine to give 1-chloro-2-butanone and 3-chloro-2-butanone.
-
Which product, compound A, B, or C, would you expect to be formed when 1-methyl-1,2-epoxycyclopentane of the absolute configuration shown is allowed to stand in methanol containing a few drops of...
-
You have just been telephoned by the chief accountant of a listed company client, Randerston plc, to tell you that there has been a computer breakdown and that some parts of the data concerning...
-
Consider the following situations: (a) Assume that you are a partner in a two partner practice with total practice income of 250 000. One of your clients (a private limited company with a turnover of...
-
You have been asked by your audit partner to be senior in charge of the audit of a small public limited company. Unbeknown to the partner, you hold 1000 of the 100 000 shares in the company. Do you...

Study smarter with the SolutionInn App


