Analyze the polarity of each bond in the following organic compound. H || a) Which...
Fantastic news! We've Found the answer you've been seeking!
Question:
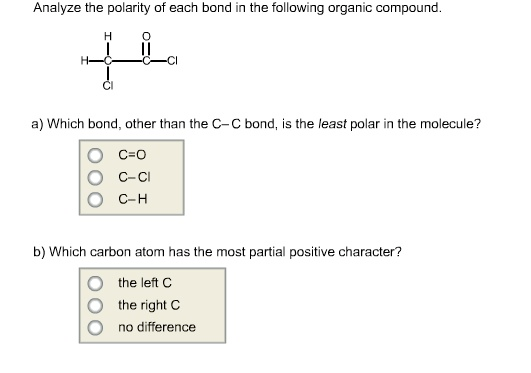
Transcribed Image Text:
Analyze the polarity of each bond in the following organic compound. ملل H || a) Which bond, other than the C-C bond, is the least polar in the molecule? C=O C-CI C-H b) Which carbon atom has the most partial positive character? the left C the right C no difference Analyze the polarity of each bond in the following organic compound. ملل H || a) Which bond, other than the C-C bond, is the least polar in the molecule? C=O C-CI C-H b) Which carbon atom has the most partial positive character? the left C the right C no difference
Expert Answer:
Answer rating: 100% (QA)
ANSWER General Guidance If the electronegativity of the two bonded atoms are different then the bond ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A carbonoxygen double bond in a certain organic molecule absorbs radiation that has a frequency of 6.0 1013 s-1. a. What is the wavelength of this radiation? b. To what region of the spectrum does...
-
Which compound has bond angles nearest to 120? (a) O = C = S (b) CHI3 (c) H2C = O (d) H-C=C-H (e) CH4
-
A compound containing only carbon and hydrogen is 85.63% C by mass. Reaction of this compound with H2O produces a secondary alcohol as the major product and a primary alcohol as the minor product. If...
-
What is the capacity for each of the containers in Problems 4754? (Give answers in the U.S. system to the nearest tenth of a gallon or in metric to the nearest tenth of a liter.) Orange 25 cm Juice...
-
What are general compliance requirements? Provide several examples.
-
Steel can be produced using three different methods: (1) A basic process using coke that produces steel ingots, (2) Continuous casting, or (3) An electric furnace using steel scrap. The following...
-
Based on the following pedigree for a trait determined by a single gene (affected individuals are shown as filled symbols), state whether it would be possible for the trait to be inherited in each of...
-
Winkler Furniture manufactures two different types of china cabinets: a French provincial model and a Danish Modern model. Each cabinet produced must go through three departments: carpentry,...
-
3. A region has the peak-period electricity demand in MWh given by D(r) = { 40,008 rif r 64 104,000 1,000r if r64 6 Exercises for Markets Module by Metin akanyldrm where r is the wholesale price in...
-
The owner of Neros company has hired you to analyze her company's performance and financial position, as well as the positions of Iits competitors, Centar and Xermix. However, the data Neros abtained...
-
The balance sheets of Forest Company and Garden Company are presented below as at December 31, Year 8. Additional Information Forest acquired 90% of Garden for $207,900 on July 1, Year 1, and...
-
using the classical model closed economy what are the effects on the variables below of a temporary increase in government spending. --- Output --- Real Interest ---Price level --- Wage rate ( real )...
-
If aggregate demand does not change, aggregate supply policies for economic growth are? explain them
-
Problem P12-1-1 Fowler and Vermeer agree to form a partnership. Fowler's investment consists of the following: Fowler's Accounts Cash A/R AFDA Merchandise Inventory Equipment Accum. Depr. A/P Notes...
-
Many different variables affect the oil market. Forecast and predict how the following will affect the equilibrium price and quantity of the oil market. Each case should be analyzed through the...
-
Please explain the calculations. ASSUMPTIONS Sales Price per Unit $49.99 Gross Margin = (Revenues - cost of goods sold) / Revenues 25% Depreciation & amortization as a % of capital expenditures 25%...
-
The balancing act governments have in efforts to provide legislation that protects employees, as well as employers, is significant. Discuss why it is important to make sure both sides are protected...
-
Using the theoretical sampling strategy, how many samples of size 4 (n = 4) can be drawn from a population of size: (a) N = 5? (b) N = 8? (c) N = 16? (d) N = 50?
-
Use the accompanying phase diagram for carbon to answer the following questions. a. How many triple points are in the phase diagram? b. What phases can coexist at each triple point? c. What happens...
-
What mass of ZnS (Ksp = 2.5 10-22) will dissolve in 300.0 mL of 0.050 M Zn(NO3)2? Ignore the basic properties of S2-.
-
For the following half reaction, E cell = 22.07 V: AlF 6 3 + 3e - Al + 6F 2 Using data from Table 11.1, calculate the equilibrium constant at 25 o C for the reaction Al 3+ (aq) 1 6F 2 (aq) AlF 6 3...
-
Are the initial rates lower on a fixed-rate loan or a variable-rate loan? Why?
-
Why are mortgage payments not included in the debt limit ratio?
-
Visit the Consumer Financial Protection Bureau Web site (www.consumerfinance.gov). Based on the public information available, describe what role the bureau plays in providing consumer education,...

Study smarter with the SolutionInn App


