The enthalpy of fusion of Ice is 6.02 k.J mol-. The heat capacity of H2O is...
Fantastic news! We've Found the answer you've been seeking!
Question:
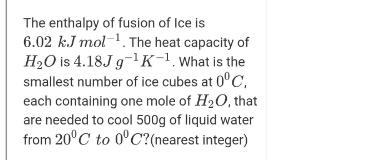
Transcribed Image Text:
The enthalpy of fusion of Ice is 6.02 k.J mol-. The heat capacity of H2O is 4.18J g-1K-1. What is the smallest number of ice cubes at 0°C, each containing one mole of H20, that are needed to cool 500g of liquid water from 20°C to 0°C?(nearest integer) The enthalpy of fusion of Ice is 6.02 k.J mol-. The heat capacity of H2O is 4.18J g-1K-1. What is the smallest number of ice cubes at 0°C, each containing one mole of H20, that are needed to cool 500g of liquid water from 20°C to 0°C?(nearest integer)
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
The enthalpy of fusion of water is 6.01 kJ/mol. Sunlight striking Earth's surface supplies 168 W per square meter (1 W = 1 watt = 1 J/s). (a) Assuming that melting of ice is only due to energy input...
-
For ammonia (NH3) the enthalpy of fusion is 5.65 kJ/ mol, and the entropy of fusion is 28.9 J K-1 mol-1. a. Will NH3(s) spontaneously melt at 200. K? b. What is the approximate melting point of...
-
Do you think that projects that are needed to comply with regulations, such as the international anti-money-laundering (AML) laws that require firms to know your customer (KYC), should undergo the...
-
Protein Blenders, Inc., made a contract with Gingerich to buy from him the shares of stock of a small corporation. When the buyer refused to take and pay for the stock, Gingerich sued for specific...
-
You have a logarithmic utility function U(W) = In W, and your current level of wealth is $5,000. (a) Suppose you are exposed to a situation that results in a 50/50 chance of winning or losing $1,000....
-
A pet food company has the business objective of having the weight of a can of cat food come as close to the specified weight as possible. Realizing that the size of the pieces of meat contained in a...
-
Compare the objectives of a product to its features.
-
Holt Company purchased a computer for $8,000 on January 1, 2011. Straight-line depreciation is used, based on a 5-year life and a $1,000 salvage value . In 2013, the estimates are revised. Holt now...
-
What is the output of the following C code? 1 #include 2 3 void xMethod(int n) { 5 } 676945 8 10 11 printf("%d",n+1); void xMethod(double n) { printf("%f",n+2); } void xMethod(long n) { 12 13. } 14...
-
Cherry Cotta makes custom ordered clay pots for residential gardens. Below is cost information regarding its latest job. a. Materials were purchased on account. $18,996 purchased b. A materials...
-
The system in Fig. P2.48 is open to 1 atm on the right side. (a) If L = 120 cm, what is the air pressure in container A? (b) Conversely, if p = 135 kPa, what is the length L?
-
A 2,000-kg car moving east at 10.0 m/s collides with a 3,000-kg car moving north. The cars stick together and move as a unit after the collision, at an angle of 38.0 north of east and at a speed of...
-
A billiard ball moving at 5.85 m/s strikes a stationary ball of the same mass. After the collision, the first ball moves at 4.79 m/s, at an angle of 35 with respect to the original line of motion....
-
The original question and answers are shown below, but my question is how do we get the value:Pound0.98 when answering the part B question? Assume a forward contract on pound sterling. Suppose the...
-
Consider a daily stock return time series (continuously compounded) that follows a Gaussian White Noise model: R~ N(0.002, 0.022). We can write this as R = u + et, where: a. has a mean of: b. has a...
-
1. Suppose the United States economy is represented by the following equations : Z = C + I + G C = 100 + .Y D T = 200 I = 30 Y D = Y - T G = 100 a) Which variables are endogenous and which are...
-
1. Advantages and Disadvantages of Banking Regulations: Thoroughly investigate the advantages, such as financial stability and consumer protection, as well as the disadvantages, such as potential...
-
Use the information given about the angles and to find the exact value of: (a) sin( + ) (b) cos( + ) (c) sin( - ) (d) tan ( + ) (e) sin(2) (f) cos (2) (g) sin /2 (h) cos/2 cos = 4/5, 0 < < /2; cos =...
-
A compound whose empirical formula is XF3 consists of 65% F by mass. What is the atomic mass of X?
-
Write balanced chemical equations to correspond to each of the following descriptions:
-
Which of these compounds can be a member of an isomer pair? In each case where isomerism is possible, identify the type or types of isomerism. [Sections 24.2, 24.4] CH2 C C-OH --O NHE Cl (b) CH3CH2CH...
-
The control volume shown in Figure \(P 2.15\) has steady, incompressible flow, and all properties are uniform at the inlet and outlet. For \(u_{1}=1.256 \mathrm{MJ} / \mathrm{kg}\) and...
-
(a) List the forces exerted on the spring-cart system of Figure 15.1 right after it is released, and draw a freebody diagram for each object in the system. (b) Which of these forces do work on the...
-
(a) In Figure 15.2e, the cart's displacement from the equilibrium position is maximum. Is the \(x\) component of the cart's acceleration at that instant positive, negative, or zero? (b) At which...

Study smarter with the SolutionInn App



