Archimedes tells us the lifting power of a balloon (how much mass it can lift) is...
Fantastic news! We've Found the answer you've been seeking!
Question:
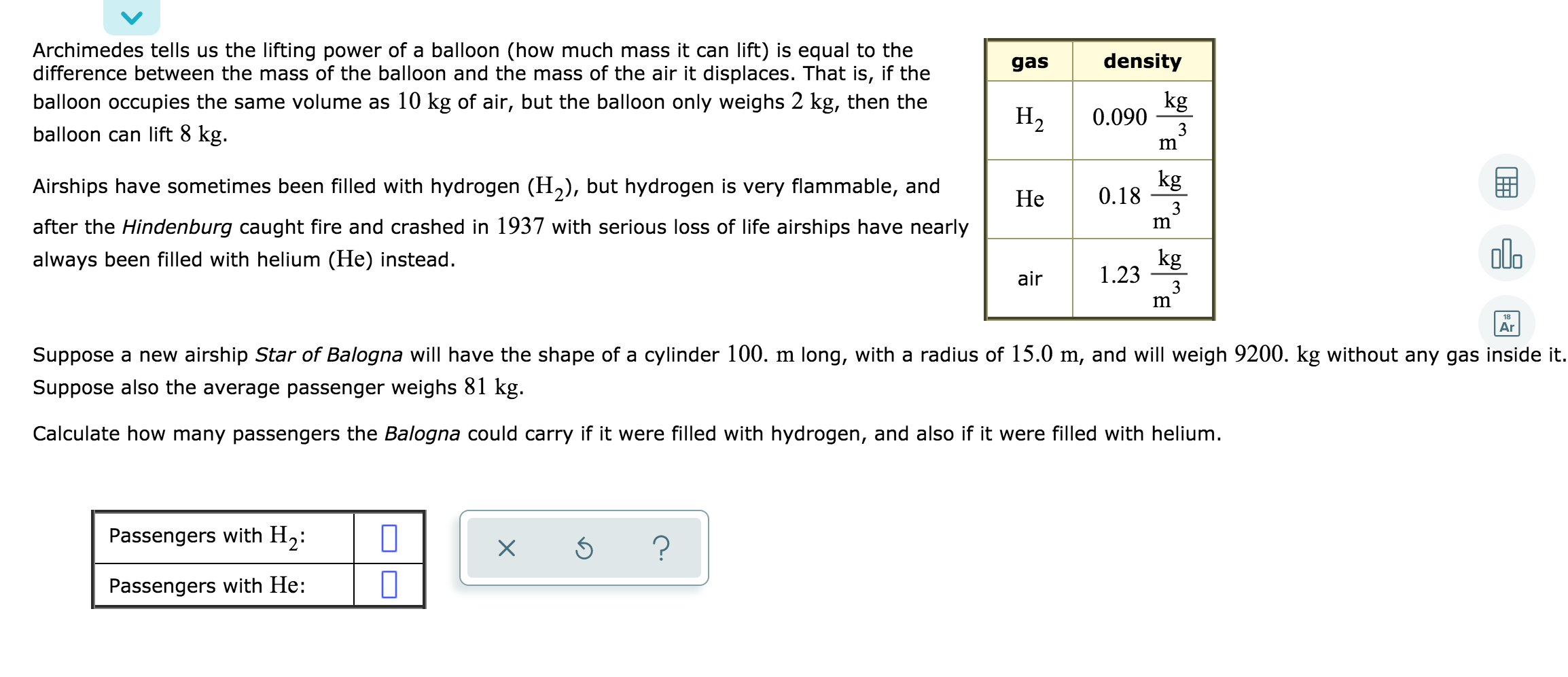
Transcribed Image Text:
Archimedes tells us the lifting power of a balloon (how much mass it can lift) is equal to the difference between the mass of the balloon and the mass of the air it displaces. That is, if the balloon occupies the same volume as 10 kg of air, but the balloon only weighs 2 kg, then the balloon can lift 8 kg. Airships have sometimes been filled with hydrogen (H₂), but hydrogen is very flammable, and after the Hindenburg caught fire and crashed in 1937 with serious loss of life airships have nearly always been filled with helium (He) instead. Passengers with H₂: Passengers with He: 0 0 X gas Ś ? H₂ He air density kg 3 0.090 0.18 1.23 m kg 3 m kg 3 m Suppose a new airship Star of Balogna will have the shape of a cylinder 100. m long, with a radius of 15.0 m, and will weigh 9200. kg without any gas inside it. Suppose also the average passenger weighs 81 kg. Calculate how many passengers the Balogna could carry if it were filled with hydrogen, and also if it were filled with helium. THE olo 18 Ar Archimedes tells us the lifting power of a balloon (how much mass it can lift) is equal to the difference between the mass of the balloon and the mass of the air it displaces. That is, if the balloon occupies the same volume as 10 kg of air, but the balloon only weighs 2 kg, then the balloon can lift 8 kg. Airships have sometimes been filled with hydrogen (H₂), but hydrogen is very flammable, and after the Hindenburg caught fire and crashed in 1937 with serious loss of life airships have nearly always been filled with helium (He) instead. Passengers with H₂: Passengers with He: 0 0 X gas Ś ? H₂ He air density kg 3 0.090 0.18 1.23 m kg 3 m kg 3 m Suppose a new airship Star of Balogna will have the shape of a cylinder 100. m long, with a radius of 15.0 m, and will weigh 9200. kg without any gas inside it. Suppose also the average passenger weighs 81 kg. Calculate how many passengers the Balogna could carry if it were filled with hydrogen, and also if it were filled with helium. THE olo 18 Ar
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A cylinder with radius R and mass M has density that increases linearly with distance r from the cylinder axis, p = ar, where a is a positive constant (a) Calculate the moment of inertia of the...
-
A constant volume tank filled with 2 kg of air rejects heat to a heat reservoir at 300 K. During the process the temperature of the air in the tank decreases to the reservoir temperature. Determine...
-
A balloon filled with helium at 25oC, 100 kPa and a volume of 0.1 m3 is moving at 10 m/s. The balloon is at an elevation of 1 km relative to an energy reference environment of T0 = 20o, p0 = 100 kPa....
-
Lungameni Enterprises (Pty) Ltd ("Lungameni Enterprises") manufactures product A, which it sells to local customers at a mark-up of 25%. Lungameni Enterprises currently absorbs its overhead costs on...
-
John Adams Companys record of transactions for the month of April was as follows. Instructions (a) Assuming that perpetual inventory records are kept in units only, compute the inventory at April 30...
-
Let Y be the binomial random variable with parameter p and n = 400. If the observed value of Y is y = 120, find a 95% confidence interval for p.
-
Question: Kelly Hackworth took a leave of absence from her job at Progressive Insurance, to care for her ailing mother. When she offered to return, Progressive refused to give her the same job or one...
-
Presented below is an aging schedule for Gille Company At December 31, 2011, the unadjusted balance in Allowance for Doubtful Accounts is a credit of $8,000.Instructions(a) Journalize and post the...
-
The following is the extract of Jordan's trial balance as at 31 December 20X7: DR $ Rent 22,000 Insurance ...
-
Perfect Parties, Inc. has several divisions, one of which provides birthday parties at their facility, and has provided the actual and planning budget results for the month of June. The Controller...
-
In the audit of accounts receivable (AR) at Charlotte Company Ltd (Charlotte) for the financial year ended 30 June 2021, the auditor is provided an aged trial balance listing all customers with the...
-
The relationship between human society and the environment, especially in the context of industrialization and fossil fuel use as the main energy source, has been characterized by uncertainty in...
-
What are the benefits and risks associated with the securitization of financial assets, and how did this practice contribute to the 2008 financial crisis ?
-
Companies use social media to promote their brand. How does Ugg use social media? What social media platforms does Ugg use to connect with its audience? Think about one of Ugg's competitorswhat is...
-
7. Like all polyfunctional organic compounds, the carbohydrates are water soluble (unless very large) and ether insoluble. Thus, they are easily distinguished from lipids, for example, which are...
-
For a variable head permeability test, the following are given: Length of specimen = 375 mm Hydraulic conductivity = 1.72 m/min Area of specimen = 1875 mm2 What should be the area of the stand pipe...
-
Review the APA Ethics page (linked below) and list three reasons ethics are important. American Psychological Association: Ethical Principles of Psychologists and Code of ConductLinks to an external...
-
Suppose the index goes to 18 percent in year 5. What is the effective cost of the unrestricted ARM?
-
Standard polystyrene solutions of known average molar masses continue to be used as for the calibration of many methods of characterizing polymer solutions. M. Kolinsky and J. Janca (J. Polym. Sci,...
-
An NO molecule has thermally accessible electronically excited states. It also has an unpaired electron, and so may be expected to be paramagnetic. However, its ground state is not paramagnetic...
-
Define the following terms: phase, constituent, component, and degree of freedom.
-
Is check forgery generally an internal or external fraud? How about credit card fraud?
-
What are major motives of fraud according to COSO?
-
How did the three Duke employees allegedly commit fraud in 2001 and 2002? Why?

Study smarter with the SolutionInn App


