Argon gas (10.0 g) is added to a 10.0 L chamber fitted with a piston at...
Fantastic news! We've Found the answer you've been seeking!
Question:
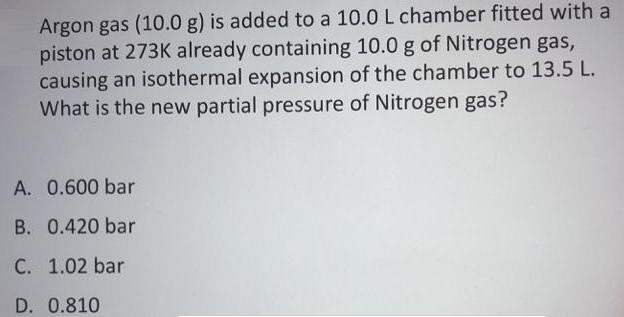
Transcribed Image Text:
Argon gas (10.0 g) is added to a 10.0 L chamber fitted with a piston at 273K already containing 10.0 g of Nitrogen gas, causing an isothermal expansion of the chamber to 13.5 L. What is the new partial pressure of Nitrogen gas? A. 0.600 bar B. 0.420 bar C. 1.02 bar D. 0.810 Argon gas (10.0 g) is added to a 10.0 L chamber fitted with a piston at 273K already containing 10.0 g of Nitrogen gas, causing an isothermal expansion of the chamber to 13.5 L. What is the new partial pressure of Nitrogen gas? A. 0.600 bar B. 0.420 bar C. 1.02 bar D. 0.810
Expert Answer:
Answer rating: 100% (QA)
Moles of argon gas added 10399 where 399 molar mass of Ar Moles of nitrogen 1028 m... View the full answer

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these accounting questions
-
An ideal regenerator (T3 = T5) is added to a simple ideal Brayton cycle (see Fig. P9-99). Air enters the compressor of this cycle at 16 psia and 100°F; the pressure ratio is 11; and the maximum...
-
A 15.9-g sample of sodium carbonate is added to a solution of acetic acid weighing 20.0 g. The two substances react, releasing carbon dioxide gas to the atmosphere. After reaction, the contents of...
-
A 10 mL sample is added to a 300 mL BOD bottle. Dilution water is added to the sample bottle and the initial dissolved-oxygen concentration is measured as 8.5 mg/L. After the sample is sealed, the...
-
You are the assistant vice president in charge of production for a firm that produces computers. Your firm's production function is f(L,K) = min (L,K) Where L and K are the quantities of the two...
-
First Local Bank would like to improve customer service at its drive- in facility by reducing waiting and transaction times. On the basis of a pilot study, the banks process manager estimates the...
-
A study that found when a larger body is labeled as normal size, people who see it become less concerned about overeating. What do you think? Will our cultures new focus on body positivity ironically...
-
Two machines are used to fill cans. The machines are supposed to fill each can with 12 ounces of liquid. The amounts actually filled follow a normal distribution. A simple random sample of 10 cans is...
-
McNamara Limiteds ledger shows the following balances on December 31, 2012: Preferred shares outstanding: 25,000 shares ...........$ 625,000 Common shares outstanding: 40,000 shares...
-
Exercise 21-23 (Algo) Cash flows from operating activities (direct method) [LO21-3] Portions of the financial statements for Myriad Products are provided below: Sales MYRIAD PRODUCTS COMPANY Income...
-
Adam Rust looked at his mechanic and sighed. The mechanic had just pronounced a death sentence on his road-weary car. The car had served him wellat a cost of $500 it had lasted through four years of...
-
At an annual growth rate of 3%, how many years will it take an initial investment of $1,000 to increase to $2,000? At an annual growth rate of 7%, how many years will it take an initial investment of...
-
Calculate DVA in Example 24.6. Assume that default can happen in the middle of each month. The default probability of the bank is 0.001 per month for the two years and the recovery rate in the event...
-
Xanadu Mining is considering three mutually exclusive alternatives, as shown in the table below. MARR is 10 percent/year. a. What is the present worth of each alternative? b. Which alternative should...
-
What is the formula for the delta of a call option on a stock that provides no income?
-
The present worth of a multiyear investment with all positive cash flows (incomes) other than the initial investment is \(P W=\$ 10,000\) at \(M A R R=i \%\). If MARR changes to \((i+1) \%\), the...
-
Two numerically controlled drill presses are being considered by the production department of Zunnis Manufacturing; one must be selected. Comparison data is shown in the table below. MARR is 10...
-
If H is an infinite dimensional Hilbert space, show that no orthonormal basis for X is a Hamel basis. Show that a Hamel basis is uncountable. The temperature of a room is 24.3 C. What is this...
-
Problem 2. (0.6 points, 0.2 points for each question) (a) A company turns its inventory 2 times a month. Its months-of-supply = Its days-of-supply = Please show your analysis below: _months. days. (1...
-
Water in a 5 - cm-deep pan is observed to boil at 98oC. At what temperature will the water in a 40 - cm-deep pan boil? Assume both pans are full of water.
-
Heat is supplied to an absorption refrigeration system from a geothermal well at 110oC at a rate of 5 105 kJ/h. The environment is at 25oC, and the refrigerated space is maintained at - 18oC....
-
A 2.4-m high 200-m2 house is maintained at 22C by an air-conditioning system whose COP is 3.2. It is estimated that the kitchen, bath, and other ventilating fans of the house discharge a houseful of...
-
The needle indicator of an electronic instrument is connected to a torsional viscous damper and a torsional spring. If the rotary inertia of the needle indicator about its pivot point is \(25...
-
Find the responses of systems governed by the following equations of motion for the initial conditions \(x(0)=1, \dot{x}(0)=0\) : a. \(2 \ddot{x}+8 \dot{x}+16 x=0\) b. \(3 \ddot{x}+12 \dot{x}+9 x=0\)...
-
Find the frequency of oscillation and time constant for the systems governed by the following equations: a. \(\ddot{x}+2 \dot{x}+9 x=0\) b. \(\ddot{x}+8 \dot{x}+9 x=0\) c. \(\ddot{x}+6 \dot{x}+9 x=0\)

Study smarter with the SolutionInn App


