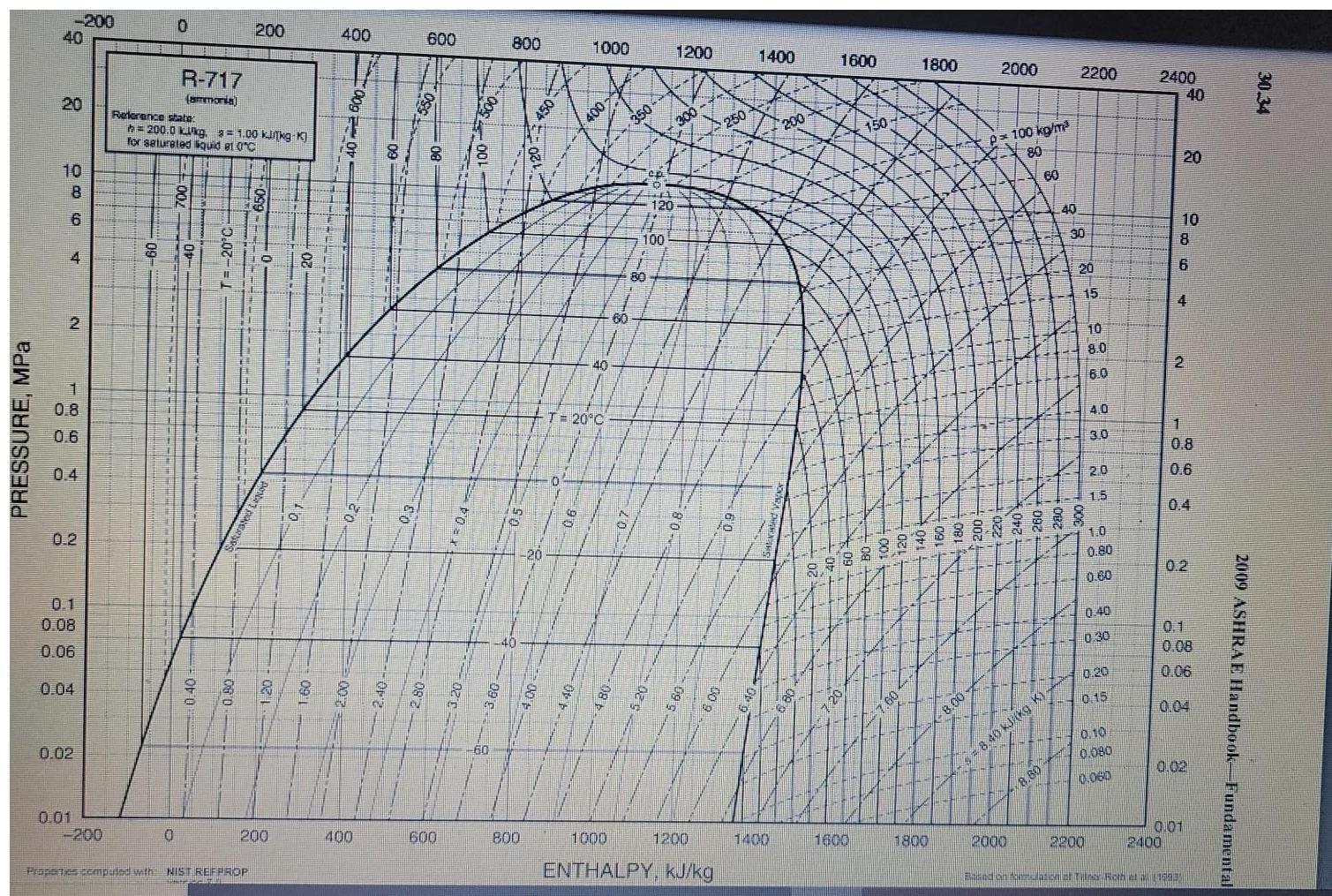
1. (30%) As a part of the liquefaction process ammonia goes through an ideal compressor and...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
1. (30%) As a part of the liquefaction process ammonia goes through an ideal compressor and a heat exchanger. After that supercritical ammonia goes through a throttling valve. Ammonia en ters the compressor at T₁ = 60°C and P₁ = 20 bar and is compressed to the pressure P₁ = 200 bar, and then cooled down in the heat exchanger to T3 = 140°C and flashed at the pressure P = 1 bar Use the attached PH diagram for ammonia to answer the following questions: A. (10) Draw this process on the attached PH diagram. B. (5) Calculate the fraction of ammonia liquefied. C. (5) Calculate the work performed in the compressor. D. (5) Calculate the amount of heat rejected in the heat exchanger. E. (5) How would the fraction of ammonia liquefied change if the amount of heat rejected in the heat exchanger remained the same, but the compressor is replaced with a non-ideal one? (Briefly explain). PRESSURE, MPa -200 40 20 10 8 6 4 2 1864 0.8 0.6 0.2 0.1 0.08 0.06 0.04 0.01 0.02 -200 60 0 0 R-717 (ammonia) Reference state h=200.0 kkg. = 1.00 kinkg-K] for saturated liquid et 0°C 0.40 200 Properties computed with NIST REFPROP O PONA 200 OB 400 20 400 600 8 8 600 500 800 15 6 40 800 450 1000 400 140 T= 20°C 250 60 ep 120 EL 100 1200 300 9 1200 3 100 1000 ENTHALPY, kJ/kg 250 1400 1400 200 1600 1600 150 1800 1800 8.00- 2000 p=100 kg/m³ 80 60 2000 4-5 = 8.40 kJ/(kg K) 40 8.80 2200 30 20 15 10 8.0 6.0 4.0 3.0 2.0 15 1.0 0.80 0.60 2200 0.40 0:30 0.20 0.15 0.10 0.080 0.060 2400 10 8 6 40 20 4 2 2400 1 0.8 06 0.2 0.08 0.06 0.04 0.01 0.02 and on fostulation af Tile Roth (1203 2009 ASHRAE Handbook-Fundamental 30.34 1. (30%) As a part of the liquefaction process ammonia goes through an ideal compressor and a heat exchanger. After that supercritical ammonia goes through a throttling valve. Ammonia en ters the compressor at T₁ = 60°C and P₁ = 20 bar and is compressed to the pressure P₁ = 200 bar, and then cooled down in the heat exchanger to T3 = 140°C and flashed at the pressure P = 1 bar Use the attached PH diagram for ammonia to answer the following questions: A. (10) Draw this process on the attached PH diagram. B. (5) Calculate the fraction of ammonia liquefied. C. (5) Calculate the work performed in the compressor. D. (5) Calculate the amount of heat rejected in the heat exchanger. E. (5) How would the fraction of ammonia liquefied change if the amount of heat rejected in the heat exchanger remained the same, but the compressor is replaced with a non-ideal one? (Briefly explain). PRESSURE, MPa -200 40 20 10 8 6 4 2 1864 0.8 0.6 0.2 0.1 0.08 0.06 0.04 0.01 0.02 -200 60 0 0 R-717 (ammonia) Reference state h=200.0 kkg. = 1.00 kinkg-K] for saturated liquid et 0°C 0.40 200 Properties computed with NIST REFPROP O PONA 200 OB 400 20 400 600 8 8 600 500 800 15 6 40 800 450 1000 400 140 T= 20°C 250 60 ep 120 EL 100 1200 300 9 1200 3 100 1000 ENTHALPY, kJ/kg 250 1400 1400 200 1600 1600 150 1800 1800 8.00- 2000 p=100 kg/m³ 80 60 2000 4-5 = 8.40 kJ/(kg K) 40 8.80 2200 30 20 15 10 8.0 6.0 4.0 3.0 2.0 15 1.0 0.80 0.60 2200 0.40 0:30 0.20 0.15 0.10 0.080 0.060 2400 10 8 6 40 20 4 2 2400 1 0.8 06 0.2 0.08 0.06 0.04 0.01 0.02 and on fostulation af Tile Roth (1203 2009 ASHRAE Handbook-Fundamental 30.34
Expert Answer:
Answer rating: 100% (QA)
A The process can be drawn on the PH diagram as follows B The fraction of ammonia liquefied can be c... View the full answer

Related Book For 

Fundamentals of Cost Accounting
ISBN: 978-1259565403
5th edition
Authors: William Lanen, Shannon Anderson, Michael Maher
Posted Date:
Students also viewed these mechanical engineering questions
-
As a part of an employment interview, you are given the partial income statement and selected financial ratios shown for Sneaky Pete's, a chain of western stores. Sneaky Pete's is organized into two...
-
What is the maximum number of stereoisomers possible for the following compound? Br HO >
-
As a part of their customer-service program, United Airlines randomly selected 10 passengers from todays 9 a.m. ChicagoTampa flight. Each sampled passenger is to be inter-viewed in depth regarding...
-
During a worldwide recession in 1983, the oil cartel began to lower prices. Why would a recession make the cartel more vulnerable to price cutting? How would the reduced demand be shared between the...
-
To exploit the strength advantage of robot manipulators and the intellectual advantage of humans, a class of manipulators called extenders has been examined [22]. The extender is defined as an active...
-
Consider 2 kmol of O 2 and 5 kmol of N 2 separated by a partition. The oxygen is at 400 K and 200 kPa. The nitrogen is at 300 K and 450 kPa. The partition is removed and the O 2 and N 2 mix....
-
According to numerous tests performed by me, what is the maximum amount of shaft current that a motor can operate with and not have EDM occur?
-
Pascarella & Wiker was retained by K & L Gates to assist in the fraud investigation requested by Le-Nature's Special Committee. What type of professional service was Pascarella & Wiker providing?...
-
What is the prevalence of Forensic Psychology? Explain the purpose of Forensic Psychology in what unique assessments are provided?
-
On 31 December 20X3, the trial balance of Joytoys showed the following chart of accounts and balances: Additional information 1 The inventory at 31 December 20X3 was valued at £19,500. 2 The...
-
Question 7 (10 points) Lord of Chips produces a special set meal which is distributed nationwide through their fast food franchisees. The following information has been obtained from the pro-forma...
-
Find the sectional lift coefficient for a thin symmetric airfoil with integrating the lifting pressure coefficient.
-
Obtain the surface pressure distribution of a spherical segment which has an apex angle of \(60^{\circ}\) and \(2 \mathrm{~m}\) radius oscillating about its nose with small amplitude.
-
Show that the enthalpy can be expressed in terms of the pressure and the density as follows: \(\mathrm{h}=2 \gamma /(\gamma-1)(\mathrm{p} / ho)\).
-
The indicative of the stall flutter is the sign of the integral under the curve of (i) lift vs vertical displacement for plunging, (ii) moment vs angle of attack for pitching. Why? In obtaining the...
-
Construct an audit questionnaire of management performance, which could be given to members of a project team. Test this on someone who has managed you. What areas would you suggest for improvement?
-
Description The writing process involves carefully planning, writing, and revising strategies to create a message that is organized, easy to read, and informative. For this assignment, you will be...
-
Transform the while loop from the previous exercise into an equivalent for loop (make sure it produces the same output).
-
When preparing cost estimates for account analysis purposes, should the costs be extracted from the historical accounting records?
-
A large company has hired your friend. She confides in you about a problem with her boss. Her boss has asked customers to sign sales agreements just before the end of the year, indicating a sale has...
-
There is no reason to investigate favorable variances; only unfavorable variances indicate problems. Do you agree?
-
Isothermal compression efficiency can be achieved by running the compressor: (a) At a very high speed (b) At a very slow speed (c) At an average speed (d) At zero speed
-
Derive the expression of work done by the compressor in isothermal compression, adiabatic compression, and polytropic compression.
-
Maximum work is done in compressing air when the compression is: (a) Isothermal compression (b) Adiabatic compression (c) Polytropic compression (d) None of these

Study smarter with the SolutionInn App


