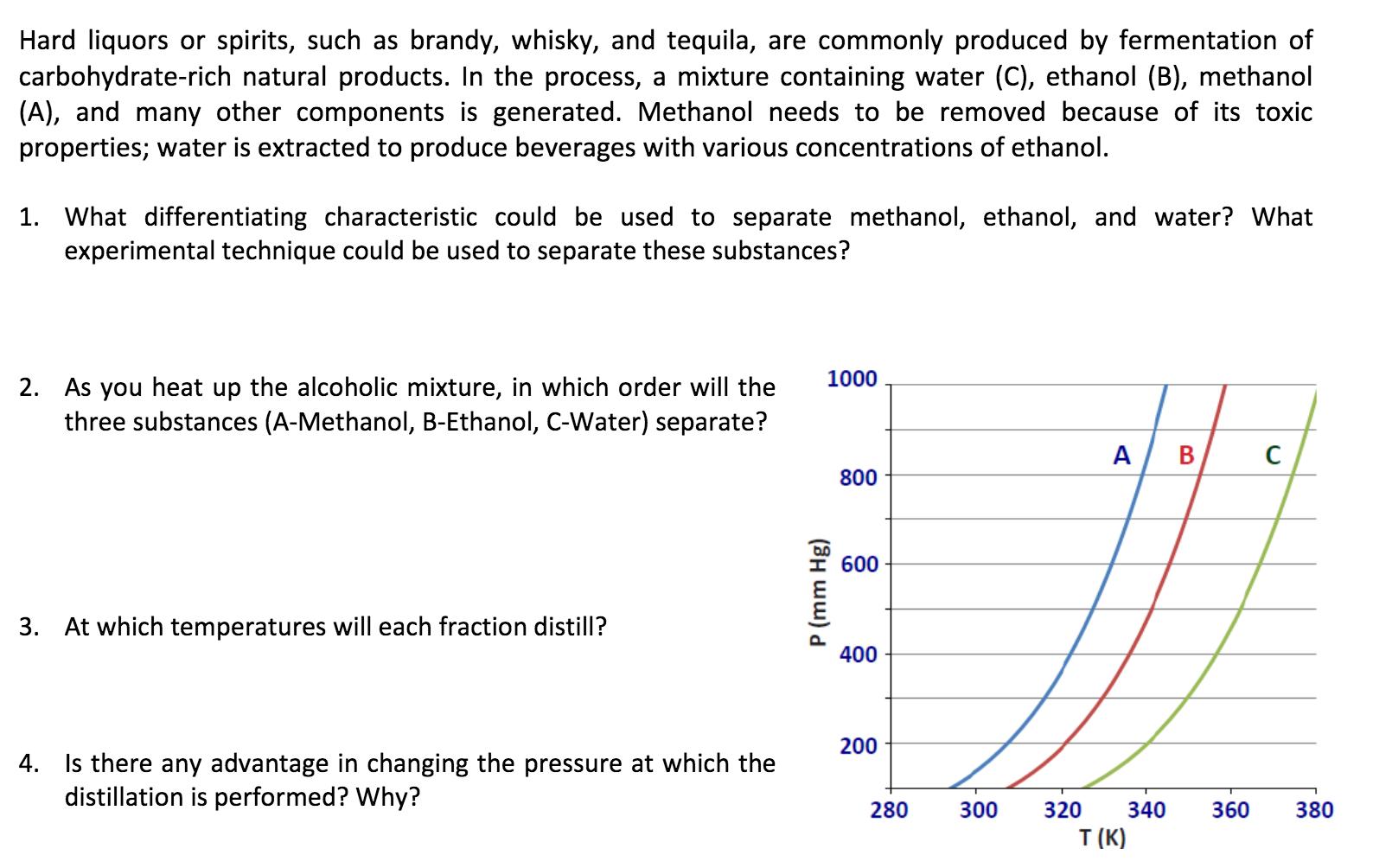
Hard liquors or spirits, such as brandy, whisky, and tequila, are commonly produced by fermentation of...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Hard liquors or spirits, such as brandy, whisky, and tequila, are commonly produced by fermentation of carbohydrate-rich natural products. In the process, a mixture containing water (C), ethanol (B), methanol (A), and many other components is generated. Methanol needs to be removed because of its toxic properties; water is extracted to produce beverages with various concentrations of ethanol. 1. What differentiating characteristic could be used to separate methanol, ethanol, and water? What experimental technique could be used to separate these substances? 1000 2. As you heat up the alcoholic mixture, in which order will the three substances (A-Methanol, B-Ethanol, C-Water) separate? A B 800 600 3. At which temperatures will each fraction distill? 400 200 4. Is there any advantage in changing the pressure at which the distillation is performed? Why? 280 300 320 340 360 380 T (K) P (mm Hg) Hard liquors or spirits, such as brandy, whisky, and tequila, are commonly produced by fermentation of carbohydrate-rich natural products. In the process, a mixture containing water (C), ethanol (B), methanol (A), and many other components is generated. Methanol needs to be removed because of its toxic properties; water is extracted to produce beverages with various concentrations of ethanol. 1. What differentiating characteristic could be used to separate methanol, ethanol, and water? What experimental technique could be used to separate these substances? 1000 2. As you heat up the alcoholic mixture, in which order will the three substances (A-Methanol, B-Ethanol, C-Water) separate? A B 800 600 3. At which temperatures will each fraction distill? 400 200 4. Is there any advantage in changing the pressure at which the distillation is performed? Why? 280 300 320 340 360 380 T (K) P (mm Hg)
Expert Answer:
Answer rating: 100% (QA)
ans 1 the differentiating character is used their difference in boili... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these organizational behavior questions
-
Consider a situation where two firms who normally compete in prices decide to form a cartel. They implement a grim trigger pricing strategy where they each receive half of the monopoly profits when...
-
Which substances in Table 10.3 would you expect to deviate most from ideal-gas behavior at low temperature and high pressure? Which would deviate least? Explain. TABLE 10.3Van der Waals Constants for...
-
The following solid substances are in separate but unlabeled test tubes: Al2(SO4)3 18H2O, BaCl2 2H2O, KOH. Describe how you could identify the compounds by chemical tests using only these...
-
Select the best answer. 1. A local chapter of the Society for Protection of the Environment beneted from the voluntary services of two attorneys. One served as a member of the Society's board of...
-
The cantilever beam ACB shown in the figure has flexural rigidity EI = 2.1 x 106 k-in.2 Calculate the downward deflections δC and δB at points C and B, respectively, due to...
-
Why do purchases appear as expenses on an income statement?
-
Describe various principles identified in the medical code of ethics.
-
Dr. Lillian Fok, a New Orleans psychologist, specializes in treating patients who are agoraphobic (i.e., afraid to leave their homes). The following table indicates how many patients Dr. Fok has seen...
-
If demand is such that, then P = 40 - 20 a.MR = 400-202 b. MR = 40-40 C. MR 40-Q
-
Josie is an accounting supervisor at Monk & Sons Realty. She instructs her subordinate, Maria, to make certain accounting entries in the company's books that will increase revenue by a material...
-
Using if--- else statements, write a grading programs. Consider the following grading systems 70 100 first class honors 60 69 Second upper division 50 59 Second lower division 40 49 Pass 0 39...
-
Instructions: Watch videos entitled "Abnormal Profit, Normal Profit & Loss" and "Objectives of the Firm" and submit a response to this Discussion Forum. In your response, make sure to consider the...
-
Calculate the carbon footprint of a household, i.e. the total annual CO emissions of the household, assuming the household annually consumes (1) 1220 (1+12) kWh electricity 8 (the CO emission factor...
-
Andrew volunteered for the American Red Cross after a recent hurricane. He traveled 200 miles and helped the victims of the disaster in the clean up for five days. He also donated $1,500 to the...
-
Determine the income tax consequences of the share redemption to the major shareholder. Start by calculating the taxable non-eligible dividend. Proceeds of Redemption Less: Paid-up Capital ITA 84(3)...
-
The equilibrium constant, Kc, for the following reaction is 1.2910-2 at 600 K. CoCl2(g) CO(g) + Cl2(g) Calculate the equilibrium concentrations of reactant and products when 0.323 moles of COCl2(g)...
-
Trendy Toes produces sports socks. The company has fixed expenses of $75,000 and variable expenses of $0.75 per package. Each package sells for $1.50. The number of packages Trendy Toes needed to...
-
Write a paper about medication error system 2016.
-
The average concentration of carbon monoxide in air in an Ohio city in 2006 was 3.5 ppm. Calculate the number of CO molecules in 1.0 L of this air at a pressure of 759 torr and a temperature of 22oC.
-
The fact that water on Earth can readily be found in all three states (solid, liquid, and gas) is in part a consequence of the fact that the triple point of water (T = 0.01 C, P = 0.006 atm) falls...
-
All the stable isotopes of boron, carbon, nitrogen, oxygen, and fluorine are shown in the chart in the right hand column (in red), along with their radioactive isotopes with t1/2 > 1 min (in blue)....
-
Based on Exhibit 1, which statement is most likely correct? A . Company A has below-average liquidity risk. B . Company B has above-average solvency risk. C . Company A has made one or more...
-
Using the information presented in Exhibit 4, the quick ratio for SAP Group at 31 December 2017 is closest to: A . 1.00. B . 1.07. C . 1.17.
-
Using the information presented in Exhibit 14, the fi nancial leverage ratio for SAP Group at December 31, 2017 is c losest to: A . 1.50. B . 1.66. C . 2.00.

Study smarter with the SolutionInn App


