Assign the symmetry elements and point group for the following molecules (a) CH (b) NHF2 (c)...
Fantastic news! We've Found the answer you've been seeking!
Question:
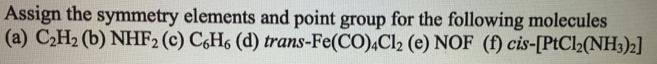
Transcribed Image Text:
Assign the symmetry elements and point group for the following molecules (a) C₂H₂ (b) NHF2 (c) C6H6 (d) trans-Fe(CO)4Cl2 (e) NOF (f) cis-[PtCl2(NH3)2] Assign the symmetry elements and point group for the following molecules (a) C₂H₂ (b) NHF2 (c) C6H6 (d) trans-Fe(CO)4Cl2 (e) NOF (f) cis-[PtCl2(NH3)2]
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Indicate what impact (+ for increase; for decrease) the following transactions would have on the accounting equation, Assets = Liabilities + Equity. A. B. C. D. E. Issued stock for cash Purchased...
-
The following molecules (AX4Y2) all have octahedral geometry. Group the molecules that are equivalent to each other. Y X' (b) (a)
-
Assign structures to the following molecules on the basis of the indicated 'H NMR spectra A-E Consider stereochemistry, where applicable (a) C4H7C1, NMR spectrum A; (b) C5H8O2, NMR spectrum B; (c)...
-
Review TP1. Review current season ticket prices for one Major League Baseball team. Choose one season ticket price area to review. A. Determine what is recognized as per ticket revenue after each...
-
A lack of reasonable care that may be characterized by the failure of auditors to follow GAAS in the conduct of the audit is known as a. Constructive fraud. b. Fraud. c. Gross negligence. d. Ordinary...
-
Costs of abnormal spoilage are losses. Explain
-
Based on the design, briefly discuss the data collection procedures to be used. Be sure to include the area rea of focus and targeted sample as part of these procedures. Develop a hypothetical...
-
OBrien Industries Inc. is a book publisher. The comparative unclassified balance sheets for December 31, 2015 and 2014 are provided below. Selected missing balances are shown by letters. The...
-
Artificial Intelligence (AI) has begun to take over many functions in the firm, including pricing, recruitment, talent promotion, advertising, loan issuance, default risk prediction, supply chain and...
-
Robson Electronics completed these selected transactions during March 2018: a. Sales of $2,050,000 are subject to an accrued warranty cost of 9%. The accrued warranty payable at the beginning of the...
-
A consumer's preferences over two goods (x1, x2) are represented by the utility function u(x1,x2) = 5x + 2x2. The income he allocates for the consumption of these two goods is m. The prices of the...
-
This table contains prices and the demands of a consumer whose behavior was observed in 5 different price-income situations. Situation P1 P2 X1 X2 A 2 2 10 70 B 2 4 70 20 C 2 2 20 30 D 6 2 10 30 E 2...
-
} for (int i=1;i <30;i++) System.out.print ("*"); System.out.println(); System.out.println("Department of Computer Science"); for (int i=1;i <30;i++) System.out.print ("*"); System.out.println(); 1.1...
-
4. (11) Suppose you own a tax preparation services company, appropriately named "MYBA Services" (MYBA = 'Minimize Your Burden of Assessment') with fixed costs of $3,000/month and marginal costs of...
-
Approximating Roots (50 points) In this problem, we will implement a square root approximator and then an nth root approximator. Recall that the nth root of a non-negative x, written , is the...
-
Calculate the mass of ascorbic acid (molecular mass = 176 g/mol) that should be dissolved in 155 g of acetic acid to cause a depression of freezing point by 1.15 K. Assume that ascorbic acid does not...
-
Jaxon's has total income of $418,300, earnings before interest and taxes of $102,600, depreciation of $59,200, and a tax rate of 30 percent. The company is fully equity funded with 15,000 shares...
-
Quadrilateral EFGH is a kite. Find mG. E H <105 G 50 F
-
What is the temperature of a two-level system of energy separation equivalent to 300 cm-I when the population of the upper state is one-half that of the lower state?
-
I.D. Chapple-Sokol, Cl Giunta, and R.G. Gordon (J Electrochem Sac 136,2993 (1989)) proposed the following radical chain mechanism for the initial stages of the gas-phase oxidation of silane by...
-
The second-order rate constant for the reaction A + 2 B --7 C + D is 0.21 dm 3 mol-J S-1. What is the concentration of C after? (a) 10 s, (b) 10 min when the reactants are mixed with initial...
-
Stadler Corporations federal income tax rate is 34 percent. It reports $100,000 depreciation expense on its financial statements and deducts $140,000 depreciation expense on its tax return. How...
-
Which of the following items is not deductible? a. Dues for club used solely for business meetings b. Directly related business entertainment c. Business gift of less than $25 in value d. Dues for...
-
John is a teacher at a local high school. During 2017, he travels three days per week to a school in the next county to work with gifted children in an after-school program that does not end until...
Working Guide To Reservoir Rock Properties And Fluid Flow 1st Edition - ISBN: 1856178250 - Free Book

Study smarter with the SolutionInn App



