Assume that Torrey Nanos drug candidate is successful in Phase I and II- its first product to
Fantastic news! We've Found the answer you've been seeking!
Question:
Assume that Torrey Nano’s drug candidate is successful in Phase I and II- its first product to make it to human trials, let alone manufacturing. It now needs to manufacture a large amount of the drug for Phase III testing.
Question: Should they vertically integrate into manufacturing?
- Apply concepts from both the transaction cost view and the capabilities view to support your argument (and if the views don’t both support your recommendation, why did you choose the direction you did).



Transcribed Image Text:
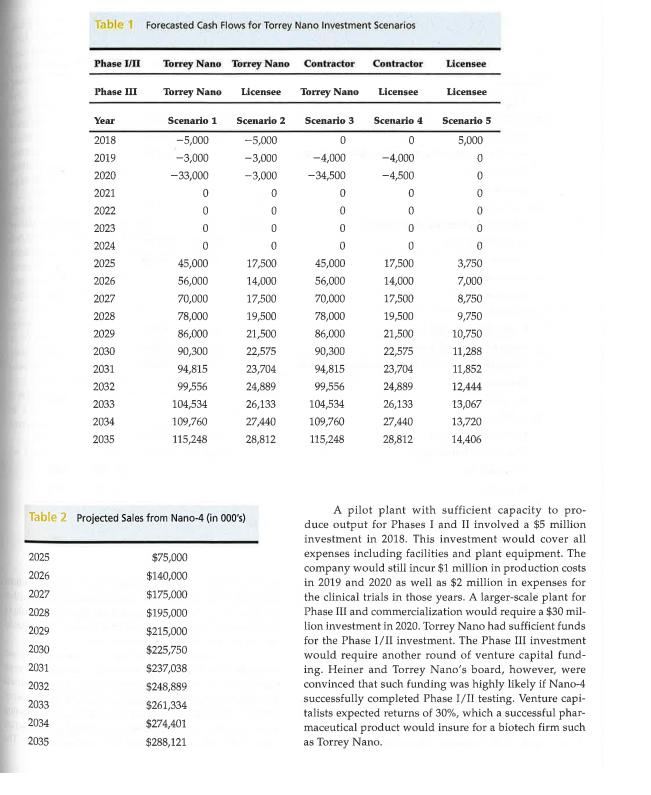
Jack Heiner, the CEO of Torrey Nano, a seven-year-old bio- technology firm, faced an important decision in the spring of 2017. Torrey Nano, based in San Diego, California was a company of 35 employees, most of whom were research sci- entists. Torrey Nano's pharmaceutical product, codename Nano-4, was almost ready for human testing. The company aspired to be a full-fledged pharmaceutical company. How- ever, like most biotech startups, it had focused on R&D since its founding. Consequently, it had done no manufacturing or marketing. With the impending human testing of Nano-4, however, it faced the decision of whether to build a small- scale pilot plant to manufacture Nano-4 for human trials. Nano-4 was the fourth product that Torrey Nano had devel- oped but the first to advance to human trials. The drug had multiple potential therapeutic applications, but Torrey Nano had focused on diabetes treatment as its first application. The company had three other products in earlier stages of development. Human testing involved three phases. Phase I, which lasted from 6-12 months, assessed basic human safety (e.g., adverse side effects, etc.). If a product was approved in Phase I, it moved to Phase II where the drug was tested with a small group of patients. Phase II examined the efficacy of a drug in treating a disease and also evaluated whether serious side effects occurred. Phase II usually lasted from one to two years. Products that succeeded in Phases I and II could go on to Phase III. Phase III, which could take two to five years to complete, tested the drug using a large sample of patients. Regulations required that whatever manufac- turing processes were used in Phase III must be the same as those used after the drug was approved and marketed commercially. The manufacturing of drugs based on nanotechnol- ogy was considered highly complex and uncertain. Drugs based on nanotechnology were particularly new in 2017 but were generally thought to hold great promise for a wide variety of diseases. Several reports suggested that manu- facturing nano-materials was both difficult to replicate and scale. Thus, it was challenging to develop effective processes for manufacturing new nano-drugs. Some nano- technology drugs, while feasible to make in the small quan- tities needed for pilot studies, were especially problematic to manufacture on a large scale. Very small differences in * This case is intended for class discussion only. Torrey Nano is a fictitious company and, while nanotechnology is believed to offer great promise for the future in pharmaceuticals, technical details in the case may not reflect reality. processes could lead to failure. Moreover, industry experts reported that firms often had difficulty transferring a manu facturing process from one facility to another. Few young companies had experience manufacturing products of the type of Nano-4. There were, however, a handful of contract manufacturers who had some experience in manufacturing involving nanotechnology. Even for these firms, though, often required considerable trial and error before mastering the process for a particular drug. Torrey Nano managers identified five possible scenar- ios for how they might handle the manufacturing of Nano- The five scenarios are listed here with cash flow estimates included in Table 1. Scenario 1: Torrey Nano builds a pilot plant for Phases I/II and a full-scale plant for Phase III. Scenario 2: Torrey Nano builds a pilot plant for Phases I/II and licenses out the manufacturing rights for Phase III. Scenario 3: Contract out manufacturing for Phases 1/ II, build a full-scale plant in-house for Phase III. Scenario 4: Contract out manufacturing for Phases 1/ II and license out manufacturing rights for Phase III. Scenario 5: Manufacturing in all phases would be done by licensee. In all five scenarios, Nano-4 would ultimately be dis tributed and marketed by an established pharmaceutical firm that would serve as Torrey Nano's and/or the eventual manufacturer's, marketing partner. If Nano-4 successfully navigated clinical testing to reach the commercial stage, ana- lysts projected sales ranging from $75 million in 2025 to over $288 million in 2035, the last year of patent protection (see Table 2). As Heiner contemplated his options, it was clear that the scenarios involving in-house manufacturing were costlier, but potentially more lucrative. Both of the first two scenarios required capital investments in 2018 and 2019 to build a pilot plant while scenarios 1 and 3 required large capital outlays to build a full-scale plant in 2020 and 2021 (see Table 3). Cash flow projections suggested that build ing a full-scale plant would allow Torrey Nano to capture a higher percentage of the revenue stream for Nano-4-$15 million licensing fee from its marketing partner in 2025 upon successful completion of clinical trials and 40 percent of royalties. Licensing the manufacturing rights after Phase II, as required by Scenarios 2 and 4, would yield a lower payout $10 million licensing fee from the marketing part ner and 10 percent royalties. Jack Heiner, the CEO of Torrey Nano, a seven-year-old bio- technology firm, faced an important decision in the spring of 2017. Torrey Nano, based in San Diego, California was a company of 35 employees, most of whom were research sci- entists. Torrey Nano's pharmaceutical product, codename Nano-4, was almost ready for human testing. The company aspired to be a full-fledged pharmaceutical company. How- ever, like most biotech startups, it had focused on R&D since its founding. Consequently, it had done no manufacturing or marketing. With the impending human testing of Nano-4, however, it faced the decision of whether to build a small- scale pilot plant to manufacture Nano-4 for human trials. Nano-4 was the fourth product that Torrey Nano had devel- oped but the first to advance to human trials. The drug had multiple potential therapeutic applications, but Torrey Nano had focused on diabetes treatment as its first application. The company had three other products in earlier stages of development. Human testing involved three phases. Phase I, which lasted from 6-12 months, assessed basic human safety (e.g., adverse side effects, etc.). If a product was approved in Phase I, it moved to Phase II where the drug was tested with a small group of patients. Phase II examined the efficacy of a drug in treating a disease and also evaluated whether serious side effects occurred. Phase II usually lasted from one to two years. Products that succeeded in Phases I and II could go on to Phase III. Phase III, which could take two to five years to complete, tested the drug using a large sample of patients. Regulations required that whatever manufac- turing processes were used in Phase III must be the same as those used after the drug was approved and marketed commercially. The manufacturing of drugs based on nanotechnol- ogy was considered highly complex and uncertain. Drugs based on nanotechnology were particularly new in 2017 but were generally thought to hold great promise for a wide variety of diseases. Several reports suggested that manu- facturing nano-materials was both difficult to replicate and scale. Thus, it was challenging to develop effective processes for manufacturing new nano-drugs. Some nano- technology drugs, while feasible to make in the small quan- tities needed for pilot studies, were especially problematic to manufacture on a large scale. Very small differences in * This case is intended for class discussion only. Torrey Nano is a fictitious company and, while nanotechnology is believed to offer great promise for the future in pharmaceuticals, technical details in the case may not reflect reality. processes could lead to failure. Moreover, industry experts reported that firms often had difficulty transferring a manu facturing process from one facility to another. Few young companies had experience manufacturing products of the type of Nano-4. There were, however, a handful of contract manufacturers who had some experience in manufacturing involving nanotechnology. Even for these firms, though, often required considerable trial and error before mastering the process for a particular drug. Torrey Nano managers identified five possible scenar- ios for how they might handle the manufacturing of Nano- The five scenarios are listed here with cash flow estimates included in Table 1. Scenario 1: Torrey Nano builds a pilot plant for Phases I/II and a full-scale plant for Phase III. Scenario 2: Torrey Nano builds a pilot plant for Phases I/II and licenses out the manufacturing rights for Phase III. Scenario 3: Contract out manufacturing for Phases 1/ II, build a full-scale plant in-house for Phase III. Scenario 4: Contract out manufacturing for Phases 1/ II and license out manufacturing rights for Phase III. Scenario 5: Manufacturing in all phases would be done by licensee. In all five scenarios, Nano-4 would ultimately be dis tributed and marketed by an established pharmaceutical firm that would serve as Torrey Nano's and/or the eventual manufacturer's, marketing partner. If Nano-4 successfully navigated clinical testing to reach the commercial stage, ana- lysts projected sales ranging from $75 million in 2025 to over $288 million in 2035, the last year of patent protection (see Table 2). As Heiner contemplated his options, it was clear that the scenarios involving in-house manufacturing were costlier, but potentially more lucrative. Both of the first two scenarios required capital investments in 2018 and 2019 to build a pilot plant while scenarios 1 and 3 required large capital outlays to build a full-scale plant in 2020 and 2021 (see Table 3). Cash flow projections suggested that build ing a full-scale plant would allow Torrey Nano to capture a higher percentage of the revenue stream for Nano-4-$15 million licensing fee from its marketing partner in 2025 upon successful completion of clinical trials and 40 percent of royalties. Licensing the manufacturing rights after Phase II, as required by Scenarios 2 and 4, would yield a lower payout $10 million licensing fee from the marketing part ner and 10 percent royalties.
Expert Answer:
Answer rating: 100% (QA)
Question Yes they should vertically merge manufacturing This is because Torrey Nanos drug is the fir... View the full answer

Related Book For 

Managerial Accounting
ISBN: 978-0078111006
14th edition
Authors: Ray Garrison, Eric Noreen and Peter Brewer
Posted Date:
Students also viewed these accounting questions
-
Based on the attached case details address the following in at least 1. External environment (threats and opportunities) 2. Internal capabilities 3. Recommended business-level strategy to follow Case...
-
Planning is one of the most important management functions in any business. A front office managers first step in planning should involve determine the departments goals. Planning also includes...
-
The Company Lalo Company, headquartered in Vaduz, is a company listed in Amsterdam, Paris and Zurich. It is the third largest small home appliance manufacturer in Europe. The company was founded in...
-
A soil sample was found to have a CEC of 24.0 cmol c kg -1 . Important: note that a cmol is not the same thing as a cmol c for divalent and trivalent ions! A. How many cmol c kg -1 of Al 3+ would be...
-
Wiart Company purchased land containing an estimated 8,000,000 tons of iron ore for $24,000,000. The land will be worth $3,600,000 without the ore after eight years of active mining. Although the...
-
A research engineer for a tire manufacturer is investigating tire life for a new rubber compound and has built 16 tires and tested them to end-of-life in a road test. The sample mean and standard...
-
What is the role of cooling towers in surface condenser ?
-
A gas turbine power plant receives a shipment of hydrocarbon fuel whose composition is uncertain but may be represented by the expression CxHy. The fuel is burned with excess air. An analysis of the...
-
Write a complete C++ program to check whether the string input is similar when it reads the same backwards as forwards using queue algorithms based on the instructions given. In the class ADTqueue,...
-
Healthwise Medical Supplies Company is located at 2400 Second Street, City, ST 12345. The company is a general partnership that uses the calendar year and accrual basis for both book and tax...
-
Biru Bookstores is considering a major expansion of its business. The details of the proposed expansion project are summarized below: The company will have to purchase RM 400,000 in equipment at...
-
Lillibridge & Friends, Incorporated provides you with the following data for its single product: Sales price per unit Fixed costs (per quarter): Selling, general, and administrative (SG&A)...
-
Why do competitive firms claim to possess market power? Do not quote from the talk, but provide an explanation framed in economic terms instead. What incentives do these firms face? How are their...
-
Keynesian Theory Monetary Theory (Chicago) Austrian School - Find more information about this Economic school of thought HERE Download HERE. From the school of economic thought you chose: Identify...
-
A manufacturer reports finished goods inventory of $750 on June 1 and $1,060 on June 30. Cost of goods manufactured for June is $5,390. What is cost of goods sold for June? Cost of Goods Sold Cost of...
-
Simplify the expression. Enter the exact answer. Hint: You can write roots as fractional exponents, for example fraction without needing a root symbol. -32 1,024 -32 1,024 as (-32/1,024)^(1/5)....
-
Use the Gordon Growth Model formula to calculate the price of a stock that will pay a dividend of $5 per share next year and the dividend is expected to remain at $5 forever. Assume a cost of capital...
-
Read Case Study Google: Dont Be Evil Unless and answer the following: Why do you think Google was adamant about not wanting to supply information requested by the government concerning the Child...
-
Pierr Manufacturing Inc. has provided the following information concerning its manufacturing costs: For example, utilities should be $1,600 per month plus $0.15 per machine-hour. The company expects...
-
Med Max buys surgical supplies from a variety of manufacturers and then resells and delivers these supplies to hundreds of hospitals. Med Max sets its prices for all hospitals by marking up its cost...
-
Harolds Roof Repair has provided the following data concerning its costs: For example, wages and salaries should be $21,380 plus $15.80 per repair-hour. The company expected to work 2,500...
-
Which of the following are boiler mountings ? (a) Economiser (b) Fusible plug (c) Super heater (d) Injector
-
Which out of the following boiler is capable of generating superheated steam without additional accessories ? (a) Cochran (b) Lancashire (c) Locomotive (d) Cornish
-
The economiser of the boiler plant is used mainly to (a) increase steam capacity (b) reduce fuel consumption (c) increase steam pressure (d) increase life of the boiler

Study smarter with the SolutionInn App


