At the end of 2018, Practice Corporation reports the following amounts Cash Equipment Accounts payable Interest...
Fantastic news! We've Found the answer you've been seeking!
Question:
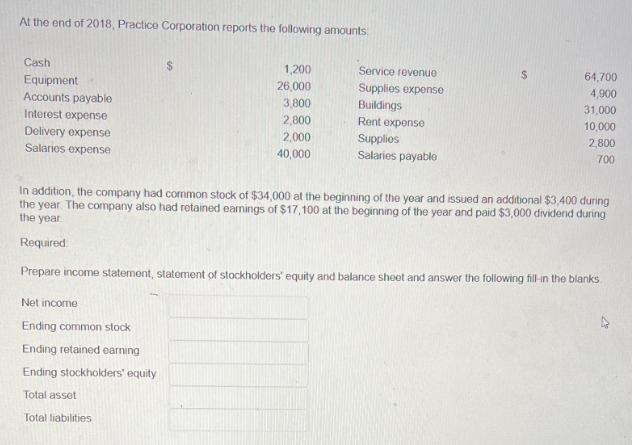
Transcribed Image Text:
At the end of 2018, Practice Corporation reports the following amounts Cash Equipment Accounts payable Interest expense Delivery expense Salarios expense 1,200 26,000 Net income Ending common stock Ending retained earning Ending stockholders' equity Total asset Total liabilities 3,800 2,800 2,000 40,000 Service revenue Supplies expense Buildings Rent expense Supplies Salaries payable 64,700 4,900 31,000 10,000 2,800 700 In addition, the company had common stock of $34,000 at the beginning of the year and issued an additional $3,400 during the year. The company also had retained earnings of $17,100 at the beginning of the year and paid $3,000 dividend during the year Required. Prepare income statement, statement of stockholders' equity and balance shoot and answer the following fill-in the blanks At the end of 2018, Practice Corporation reports the following amounts Cash Equipment Accounts payable Interest expense Delivery expense Salarios expense 1,200 26,000 Net income Ending common stock Ending retained earning Ending stockholders' equity Total asset Total liabilities 3,800 2,800 2,000 40,000 Service revenue Supplies expense Buildings Rent expense Supplies Salaries payable 64,700 4,900 31,000 10,000 2,800 700 In addition, the company had common stock of $34,000 at the beginning of the year and issued an additional $3,400 during the year. The company also had retained earnings of $17,100 at the beginning of the year and paid $3,000 dividend during the year Required. Prepare income statement, statement of stockholders' equity and balance shoot and answer the following fill-in the blanks
Expert Answer:
Answer rating: 100% (QA)
To prepare the income statement statement of stockholders equity and balance sheet we need to organi... View the full answer

Related Book For 

Interpreting and Analyzing Financial Statements
ISBN: 978-0132746243
6th edition
Authors: Karen P. Schoenebeck, Mark P. Holtzman
Posted Date:
Students also viewed these accounting questions
-
Read the case study "Southwest Airlines," found in Part 2 of your textbook. Review the "Guide to Case Analysis" found on pp. CA1 - CA11 of your textbook. (This guide follows the last case in the...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Q1. The following transactions occurred during Year 2, the second year of business for Nancy Nanny Child Care. Record each transaction in proper journal entry format below using debits and credits....
-
Yang Company purchased 2,000 widgets and has 400 widgets in its ending inventory at a cost of $90 each and a current replacement cost of $80 each. The net realizable value of each unit in the ending...
-
Both surfaces of a double convex lens have radii of 31.0cm. If the focal length is 28.9cm, what is the index of refraction of the lens material?
-
Gasoline (assumed C8H18) is burned steadily with air in a jet engine. If the airfuel ratio is 18 kg air/kg fuel, determine the percentage of theoretical air used during this process.
-
The Bonferroni-adjusted P-value is always greater than the uncorrected P-value. In Exercises 5 and 6, determine whether the statement is true or false. If the statement is false, rewrite it as a true...
-
Refer to Problem 10-1. What is the projects PI?
-
Explain the concept of Inflation Targeting within the context of Ghana. What is the primary objective of Monetary Policy in Ghana? Discuss the tools used by the Bank of Ghana in the conduct of...
-
1. Suppose Siscos altercation occurred while he was en route to his house during an unpaid one-hour lunch break. Would Sisco still be eligible for compensation? Why or why not? 2. Would Sisco have...
-
The coefficient of thermal expansion of concrete and steel are alpha_concrete = 1.2 x 10-5/C and alpha_steel = 1.1 x 10-5/C, respectively. The length of the steel conforms to the size of the...
-
As a Dedicated Service Assistant in the Jockey Club Retail and Telephone Registration Department, How the performance information is gathered and reason why by using a 360-degree performance...
-
The reaction of a-pinene oxide with aqueous acid includes a complex rearrangement. What would the reaction look like if a simpler molecule-cyclohexene oxide - reacted with aqueous acid and no...
-
Forrett Company borrowed $100,000 on October 1, 2023, by signing a $100,000, 8%, 4-month note. Required: Record the issuance of the note, the adjusting journal entry for year-end on 12/31/23, and the...
-
What are the salient considerations in the selection of an appropriate team/team members for IT-driven? transformational project? What could Dr. Banerjee have done differently to make the project...
-
Determining the exact concentration of HC1. Based on the information below, what can you conclude about this lab? What is the deviation? Results: THAM molar mass: 121.1 g/mol Sample 1 Sample 2 Sample...
-
Complete worksheet and enter closing entries on Journal Account Titles Cash Accounts Recievable Supplies Prepaid Insurance Equipment Accum Depr.-Equipment Buildings Accum Depr.- Buildings Accounts...
-
What is EBIT/eps analysis? What information does it provide managers?
-
Which financial statement reports: a. Whether assets are primarily financed with debt or equity? (BS / IS / SE / CF) b. Whether the company was profitable or not? (BS / IS / SE / CF) c. Cash received...
-
Q1. Prepare a multi-step Income Statement for both companies in the space provided below. Q2. Assuming all transactions are cash transactions, prepare a Statement of Cash Flows. Also prepare the...
-
Q1. FINANCIAL TRIVIA For the fiscal years ending below put a large circle in the box of the company that you guess has . a. The greatest amount of assets. (This one is completed for you.) b. The...
-
The consistent mass matrix of a bar element is given by a. \(\frac{ho A l}{6}\left[\begin{array}{ll}2 & 1 \\ 1 & 2\end{array} ight]\) b. \(\frac{ho A l}{6}\left[\begin{array}{rr}2 & -1 \\ -1 &...
-
How are fixed boundary conditions incorporated in the finite element equations?
-
True or False. The system mass matrix is always singular unless the boundary conditions are incorporated.

Study smarter with the SolutionInn App


