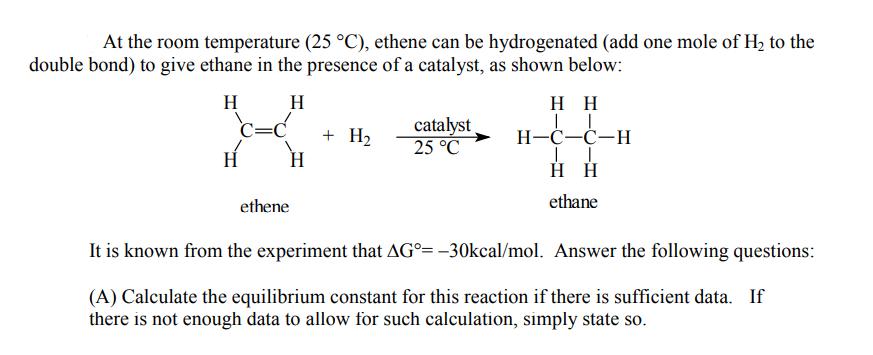
At the room temperature (25 C), ethene can be hydrogenated (add one mole of H to...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
At the room temperature (25 °C), ethene can be hydrogenated (add one mole of H₂ to the double bond) to give ethane in the presence of a catalyst, as shown below: H H C=C H ethene + H₂ catalyst 25 °C HH LL H-C-C-H H H ethane It is known from the experiment that AG°= -30kcal/mol. Answer the following questions: (A) Calculate the equilibrium constant for this reaction if there is sufficient data. If there is not enough data to allow for such calculation, simply state so. (B) Predict the sign of this AS° for this reaction. Briefly explain your reasoning. (C) Predict the sign of AH° for this reaction. Explain briefly how you arrive at this conclusion. At the room temperature (25 °C), ethene can be hydrogenated (add one mole of H₂ to the double bond) to give ethane in the presence of a catalyst, as shown below: H H C=C H ethene + H₂ catalyst 25 °C HH LL H-C-C-H H H ethane It is known from the experiment that AG°= -30kcal/mol. Answer the following questions: (A) Calculate the equilibrium constant for this reaction if there is sufficient data. If there is not enough data to allow for such calculation, simply state so. (B) Predict the sign of this AS° for this reaction. Briefly explain your reasoning. (C) Predict the sign of AH° for this reaction. Explain briefly how you arrive at this conclusion.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Calculate the Keq for this reaction if the KP = 5.205 103 at 660C. CO (g) + F2(g) + COF2(g)
-
Calculate the Keq for this reaction if the KP = 78.3 at 100C. 4HC1 (g) + O2(g) + 2H2 O(g) + 2C12 (g)
-
From the following reduction potentials (a) Calculate the equilibrium constant for .I2 (aq) + I- I-3 (b) Calculate the equilibrium constant for I2 (aq) + I- I-3 (c) Calculate the solubility (g/L) of...
-
Data from the registrar's office at Gigantic State University indicate that over the past 20 years tuition and enrollment have both increased. From this information we can conclude that: A. higher...
-
The output function of a network in expressed using Laplace transform in the following form. Find the output vo(t) as a function oftime. 12 V,(s) s(s? + 3s + 2) a 12 + 3* + 4 u(1)V 12 + 3e 2 + 4e b....
-
The helicopter shown can produce a maximum downward air speed of 80 ft/s in a 30-ft-diameter slipstream. Knowing that the weight of the helicopter and its crew is 3500 lb and assuming = 0.076 lb/ft3...
-
Cruz Manufacturing's sales slumped badly in 2002. For the first time in its his- tory, it operated at a loss. The company's income statement showed the following results from selling 600,000 units of...
-
Jones Petro Company reports the following consolidated statement of income: Operating revenues ....................$2,989 Costs and expenses: Cost of rentals and royalties ................ 543 Cost...
-
Tracy invested in a lifetime annuity that begins payments at age 65. Her life expectancy is 87. Tracy invested $800,000 in the annuity, which earns 4.5% APR, compounded monthly. With this annuity,...
-
You talk to Sally about getting paid for the work you're doing. You suggest $25 an hour and she agrees. You are only doing this temporarily since you have some extra time so you set yourself up as a...
-
PART II-PROBLEMS AND REPORTS/PRINTOUTS P-10-1. Rock Castle Construction-Setup Assume Rock Castle Construction converted from a manual system to QuickBooks. Required a. How many times did Rock Castle...
-
When can a partial dependency exist in a database? If yes, example please
-
Given the arrangement of charged particles in the figure below, find the net electrostatic force on the q = 5.35-C charged particle. (Assume 92 = 12.33 C and 93 = -19.12 C. Express your answer in...
-
is this cite correct Staff, About Amazon. "How Your Package Gets from Amazon's Warehouse to Your Front Door."USAbout Amazon, 24 July 2019,...
-
Research golf course lawsuits. Find a lawsuit, read about the lawsuit and share the information on the lawsuit with your classmates. Provide a thorough summary of the lawsuit and discuss how it might...
-
You are a homeowner with a swimming pool in your backyard. You share a fence with the neighbor behind you. While you went on vacation, your neighbor's seven year old came through a hole in the fence...
-
I need a solution for all the questions . PACKAGE B -CLASS 005 Question 2 (30 marks) IN 1. What is the present value of $10,000 received in year 5 when: a. Interest rate is 5% b. Interest rate is...
-
The domain of the variable in the expression x 3/x + 4 is________.
-
Find the closest point to b = (1, 2, - 1,3)T in the subspace W = span {(1,0,2, l)T, (1, 1,0, l)T, (2, 0, 1, - 1)T} by first constructing an orthogonal basis of W and then applying the orthogonal...
-
A bacteria colony grows according to the equation du/dt = 1.3 m. How long until the colony doubles? quadruples? If the initial population is 2, how long until the population reaches 2 million?
-
A matrix is said to be a semi-magic square if its row- sums and column sums (i.e., the sum of entries in an individual row or column) all add up to the same number. An example is whose row and column...
-
Assume that you own and manage a small clothing store. A. What kinds of financial information would you want to run your business? B. What kinds of nonfinancial information would you want (for...
-
One of the first U.S. major industries to be regulated was the railroads. Farmers and manufacturers depended on railroads to get their products to market, and often there was only one railroad...
-
The chapter indicates that two very different objectives of accounting are to provide useful information for making decisions about the future, and to provide accountability for what happened in the...

Study smarter with the SolutionInn App



