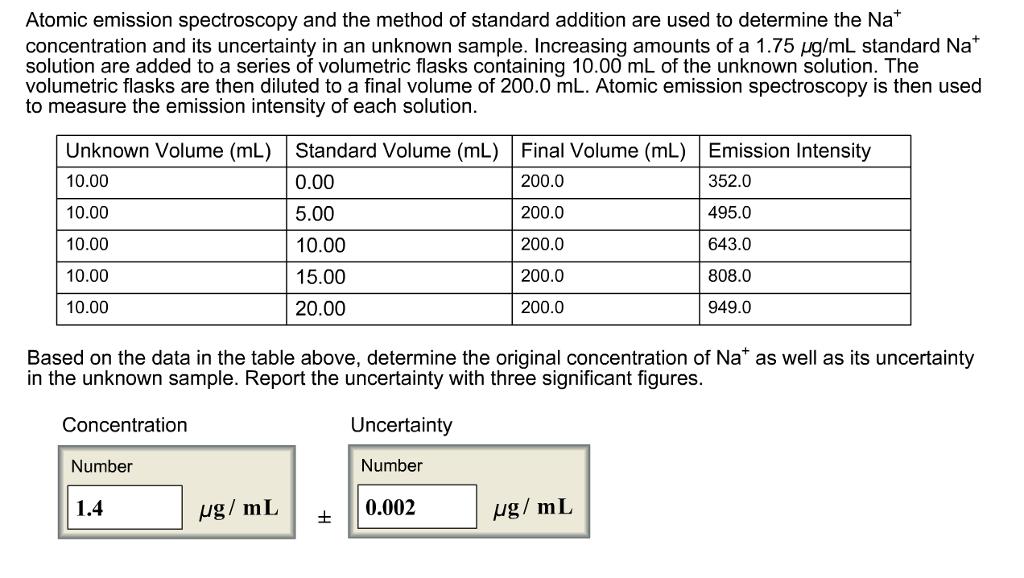
Atomic emission spectroscopy and the method of standard addition are used to determine the Na* concentration...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Atomic emission spectroscopy and the method of standard addition are used to determine the Na* concentration and its uncertainty in an unknown sample. Increasing amounts of a 1.75 ug/mL standard Na* solution are added to a series of volumetric flasks containing 10.00 mL of the unknown solution. The volumetric flasks are then diluted to a final volume of 200.0 mL. Atomic emission spectroscopy is then used to measure the emission intensity of each solution. Unknown Volume (mL) Standard Volume (mL) Final Volume (mL) Emission Intensity 10.00 0.00 200.0 352.0 10.00 5.00 200.0 495.0 10.00 10.00 200.0 643.0 10.00 15.00 200.0 808.0 10.00 20.00 200.0 949.0 Based on the data in the table above, determine the original concentration of Na* as well as its uncertainty in the unknown sample. Report the uncertainty with three significant figures. Concentration Uncertainty Number Number 1.4 µg/ mL 0.002 ug/ mL Atomic emission spectroscopy and the method of standard addition are used to determine the Na* concentration and its uncertainty in an unknown sample. Increasing amounts of a 1.75 ug/mL standard Na* solution are added to a series of volumetric flasks containing 10.00 mL of the unknown solution. The volumetric flasks are then diluted to a final volume of 200.0 mL. Atomic emission spectroscopy is then used to measure the emission intensity of each solution. Unknown Volume (mL) Standard Volume (mL) Final Volume (mL) Emission Intensity 10.00 0.00 200.0 352.0 10.00 5.00 200.0 495.0 10.00 10.00 200.0 643.0 10.00 15.00 200.0 808.0 10.00 20.00 200.0 949.0 Based on the data in the table above, determine the original concentration of Na* as well as its uncertainty in the unknown sample. Report the uncertainty with three significant figures. Concentration Uncertainty Number Number 1.4 µg/ mL 0.002 ug/ mL
Expert Answer:
Related Book For 

Fundamentals of Analytical Chemistry
ISBN: 978-0495558286
9th edition
Authors: Douglas A. Skoog,Donald M. West, F. James Holler, Stanley R. Crouch
Posted Date:
Students also viewed these chemistry questions
-
The method of standard additions was used to determine nitrite in a soil sample. A 1.00-mL portion of the sample was mixed with 24.00 mL of a colorimetric reagent, and the nitrite was converted to a...
-
In Monopoly, dice are used to determine which player gets to move first. Suppose there are two players in the game. Each player rolls a die and the player with the higher number gets to move first....
-
An analyst plans to use P/E and the method of comparables as a basis for recommending purchasing shares of one of two peer - group companies in the business of manufacturing personal digital...
-
In 2020, Neighbor Co-Op Inc. sells 1,100 beverages in glass bottles and receives a $1.00 deposit for each returnable bottle sold. As of December 31, 2020, a total of 880 glass bottles were returned...
-
Mrs. STs corporate employer has a cafeteria plan under which its employees can receive a $3,000 year-end Christmas bonus or enroll in a qualified medical reimbursement plan that pays up to $3,000 of...
-
Find the measures of the angles in Fig. 2.13. 3 A C- 1 5 62 6 AB || CD 3 Fig. 2.13 B D
-
What are some ways to steal cash? What can help to prevent cash and check thefts?
-
The reorder point is defined as the lead-time demand for an item. In cases of long lead times, the lead-time demand and thus the reorder point may exceed the economic order quantity Q*. In such...
-
View Policies Current Attempt in Progress Suppose that the following information is available for Home Depot Corporation and Lowes Corporation: Home Depot Corporation Lowes Corporation (in millions)...
-
"Part 1: The Performance Lawn Equipment database contains data needed to develop a pro forma income statement. Dealers selling PLE products all receive 18% of sales revenue for their part of doing...
-
Use the contingency table to the right to determine the probability of events a. What is the probability of event A' b. What is the probability of event A and B c. What is the probability of event A'...
-
ed Campbell Company established a predetermined variable overhead cost rate at $24.90 per direct labor hour. The actual variable overhead cost rate was $23.10 per hour. The planned level of labor...
-
A painter needs to cover a triangular region 62 meters by 66 meters by 73 meters. A can of paint covers 70 square meters. How many cans will be needed?
-
Explain how events in financial markets can contribute (either exacerbate or cause) recessions. You should explain both the chain of causality and give a recent example of a development in financial...
-
if current ratio increases and quick ratio decreases what is most likely going on ?
-
Suppose the function f and its derivative f' have values as given in the following table. 1 f(z) f(z) 8 10 221 3 4 7 19 52 6 7 8 9 4 6 20 3 17 8 15 14 8 13 14 Compute L* f(=)f (s) da Enter the exact...
-
[Chapter 10: Creating Section Views in AutoCAD] P10-5) Draw the following object converting the front view into a full section. Draw the appropriate cutting plane line. It is not necessary to include...
-
Solve each equation or inequality. |6x8-4 = 0
-
Why is a Walden reductor always used with solutions that contain appreciable concentrations of HCl?
-
List the types of substances to which each of the following separation methods is most applicable: (a) Supercritical fluid chromatography. (b) Capillary zone electrophoresis. (c) Flow FFF.
-
Consider curves for the titration of 0.10 M NaOH and 0.010 M NH3 with 0.10 M HCl. (a) Briefly account for the differences between curves for the two titrations. (b) In what respect will the two...
-
The bidask spread (II) Consider the spot quotations in pounds per dollar (/\($)\) and dollars per pound (\($/)\) in Table 3.15: a. Compute the bid quotations in American and European terms. b....
-
Interest rate parity Consider Figure 3.29, which indicates (approximately) the difference between interest rates in the United States and the UK, Rus Ruk on a one-year T-bill (i.e. a two-year bill...
-
Points quotations In Table 3.16, consider the spot quotations in dollars per pound (\($/)\) and the forward quotations in basis points (1/10,000). On the basis of the points quotations in Table 3.16,...

Study smarter with the SolutionInn App



