b) A system consists of air initially at state 1, where P = 100 kPa, T...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
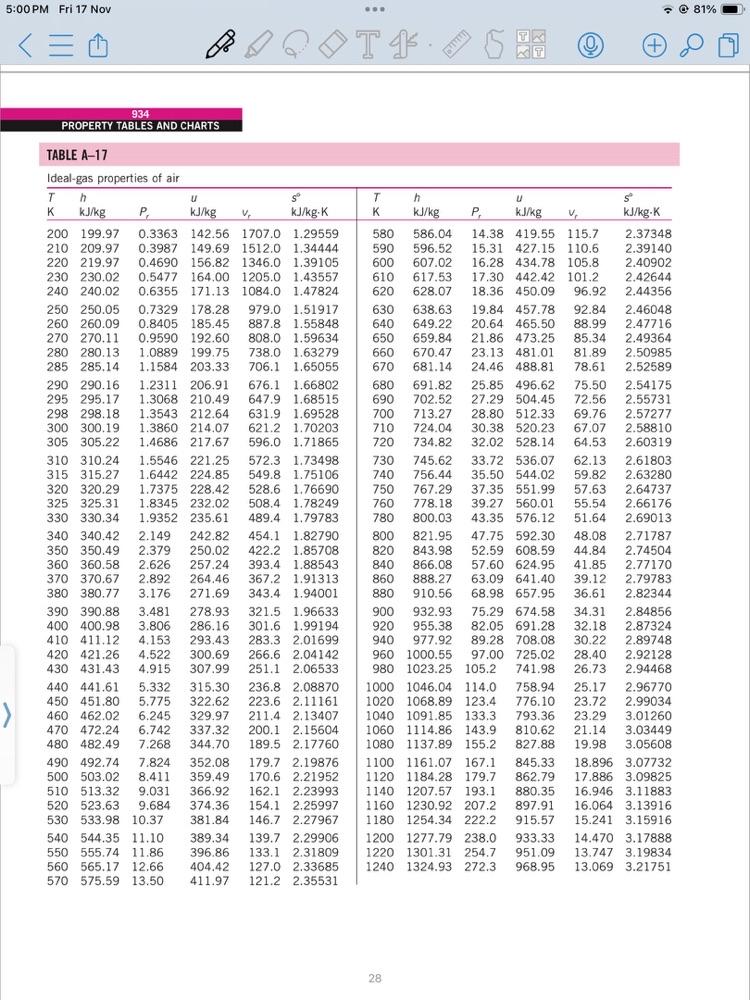
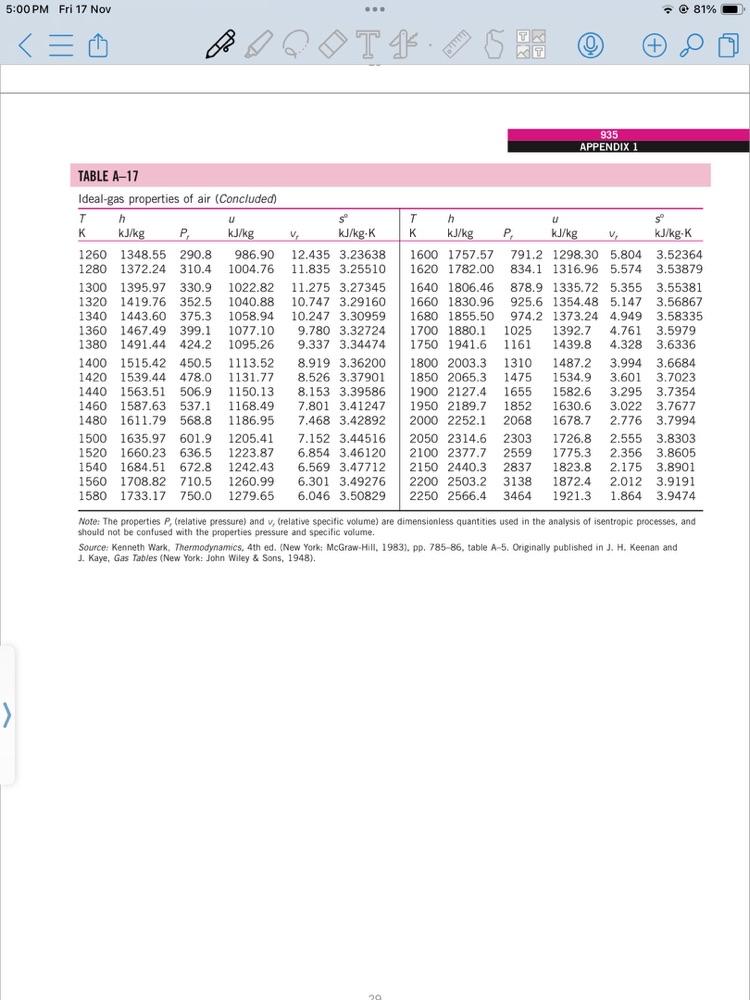
b) A system consists of air initially at state 1, where P = 100 kPa, T = 350K. The system undergoes the following process: Process 1 Process 2 2: adiabatic compression to P2 = 1500 kPa, v = 0.1359 m/kg 3: constant volume cooling to T3 = 300K a) Show P-v diagram b) Determine work and heat transfer for each process using data from Table A-17 and constant specific heat c) Discuss your findings (10 marks) 5:00 PM Fri 17 Nov 934 PROPERTY TABLES AND CHARTS TABLE A-17 Ideal-gas properties of air P T - 81% (+ Th U T h U K kJ/kg P kJ/kg V kJ/kg-K K kJ/kg P kJ/kg V kJ/kg-K 200 199.97 0.3363 142.56 1707.0 1.29559 580 586.04. 14.38 419.55 115.7 2.37348 210 209.97 0.3987 149.69 1512.0 1.34444 220 219.97 0.4690 156.82 1346.0 1.39105 590 596.52 15.31 427.15 110.6 2.39140 600 607.02 16.28 434.78 105.8 2.40902 230 230.02 0.5477 164.00 1205.0 1.43557 610 617.53 17.30 442.42 101.2 2.42644 240 240.02 0.6355 171.13 1084.0 1.47824 620 628.07 18.36 450.09 96.92 2.44356 250 250.05 0.7329 178.28 979.0 1.51917 630 638.63 19.84 457.78 92.84 2.46048 260 260.09 0.8405 185.45 270 270.11 0.9590 192.60 280 280.13 1.0889 199.75 285 285.14 1.1584 203.33 290 290.16 1.2311 206.91 295 295.17 1.3068 210.49 647.9 1.68515 298 298.18 1.3543 212.64 631.9 1.69528 300 300.19 1.3860 214.07 621.2 1.70203 305 305.22 1.4686 217.67 596.0 1.71865 310 310.24 1.5546 221.25 572.3 1.73498 315 315.27 1.6442 224.85 549.8 1.75106 320 320.29 1.7375 228.42 528.6 1.76690 325 325.31 1.8345 232.02 508.4 1.78249 330 330.34 1.9352 235.61 489.4 1.79783 340 340.42 2.149 242.82 454.1 1.82790 350 350.49 2.379 250.02 422.2 1.85708 360 360.58 2.626 257.24 393.4 1.88543 370 370.67 2.892 264.46 367.2 1.91313. 380 380.77 3.176 271.69 343.4 1.94001 390 390.88 3.481 278.93 321.5 1.96633 400 400.98 3.806 286.16 301.6 1.99194 410 411.12 4.153 293.43 283.3 2.01699 420 421.26 4.522 300.69 266.6 2.04142 430 431.43 4.915 307.99 251.1 2.06533 440 441.61 5.332 315.30 236.8 2.08870 450 451.80 5.775 322.62 223.6 2.11161 460 462.02 6.245 329.97 211.4 2.13407 470 472.24 6.742 337.32 200.1 2.15604 480 482.49 7.268 344.70 189.5 2.17760 490 492.74 7.824 352.08 179.7 2.19876 500 503.02 8.411 359.49 170.6 2.21952 510 513.32 9.031 366.92 162.1 2.23993 520 523.63 9.684 374.36 154.1 2.25997 530 533.98 10.37 381.84 146.7 2.27967 540 544.35 11.10 550 555.74 11.86 560 565.17 12.66 570 575.59 13.50 887.8 1.55848 640 649.22 20.64 465.50 88.99 2.47716 808.0 1.59634 650 659.84. 21.86 473.25 85.34 2.49364 738.0 1.63279 706.1 1.65055 660 670.47 23.13 481.01 81.89 2.50985 670 681.14 24.46 488.81 78.61 2.52589 676.1 1.66802 680 691.82 25.85 496.62 75.50 2.54175 690 702.52 27.29 504.45 72.56 2.55731 700 713.27 28.80 512.33 69.76 2.57277 710 724.04 30.38 520.23 67.07 2.58810 720 730 740 756.44 750 767.29 760 778.18 780 800.03 800 821.95 734.82 32.02 528.14 64.53 2.60319 745.62 33.72 536.07 62.13 2.61803 35.50 544.02 37.35 551.99. 57.63 39.27 560.01 43.35 576.12 59.82 2.63280 2.64737 55.54 2.66176 51.64 2.69013 47.75 592.30 48.08 2.71787 820 843.98 52.59 608.59 44.84 2.74504 840 860 866.08 57.60 624.95 41.85 2.77170 888.27 63.09 641.40 39.12 2.79783 880 910.56 900 920 68.98 657.95 36.61 2.82344 932.93 955.38 75.29 674.58 34.31 2.84856 82.05 691.28 32.18 2.87324 940 977.92 389.34 396.86 139.7 2.29906 133.1 2.31809 404.42 411.97 127.0 2.33685 121.2 2.35531 960 1000.55 980 1023.25 105.2 1000 1046.04 114.0 1020 1068.89 123.4 1040 1091.85 133.3 1060 1114.86 143.9 1080 1137.89 155.2 1100 1161.07 167.1 1120 1184.28 179.7 1140 1207.57 193.1 1160 1230.92 207.2 1180 1254.34 222.2 1200 1277.79 238.0 1220 1301.31 254.7 1240 1324.93 272.3 89.28 708.08 97.00 725.02 741.98 758.94 25.17 2.96770 776.10 23.72 2.99034 793.36 23.29 3.01260 810.62 21.14 3.03449 827.88 19.98 3.05608 845.33 18.896 3.07732 862.79 880.35 897.91 30.22 2.89748 28.40 2.92128 26.73 2.94468 17.886 3.09825 16.946 3.11883 16.064 3.13916 915.57 15.241 3.15916 933.33 14.470 3.17888 951.09 968.95 13.747 3.19834 13.069 3.21751 28 5:00 PM Fri 17 Nov T. TABLE A-17 Ideal-gas properties of air (Concluded) 935 APPENDIX 1 - 81% T h u T h K kJ/kg P, kJ/kg V, kJ/kg-K K kJ/kg 1260 1348.55 290.8 986.90 1280 1372.24 310.4 1300 1395.97 330.9 1320 1419.76 352.5 1340 1443.60 375.3. 1360 1467.49 399.1 1380 1491.44 424.2 1400 1515.42 450.5 1420 1539.44 478.0 1440 1563.51 506.9 1460 1587.63 537.1 1480 1611.79 568.8 1004.76 1022.82 1040.88 1058.94 1077.10 1095.26 12.435 3.23638 11.835 3.25510 11.275 3.27345 10.747 3.29160 10.247 3.30959 9.780 3.32724 9.337 3.34474 1113.52 8.919 3.36200 1131.77 8.526 3.37901 1150.13 8.153 3.39586 1168.49 7.801 3.41247 1186.95 7.468 3.42892 1600 1757.57 1620 1782.00 1640 1806.46 1660 1830.96 1680 1855.50 1700 1880.1 1750 1941.6 1800 2003.3 1310 1850 2065.3 1475 1900 2127.4 1655 1950 2189.7 1852 2000 2252.1 2068 kJ/kg P 1298.30 791.2 5.804 3.52364 834.1 1316.96 5.574 3.53879 878.9 1335.72 5.355 3.55381. 925.6 1354.48 5.147 3.56867 974.2 1373.24 4.949 3.58335 1025 1392.7 4.761 3.5979 1161 1439.8 4.328 3.6336 1487.2 3.994 3.6684 1534.9 V kJ/kg-K 3.601 3.7023 1582.6 3.295 3.7354 1630.6 3.022 3.7677 1678.7 2.776 3.7994 1500 1635.97 601.9 1520 1660.23 636.5 1540 1684.51 672.8 1242.43 1560 1708.82 710.5 1260.99 1580 1733.17 750.0 1279.65 1205.41 7.152 3.44516 1223.87 6.854 3.46120 6.569 3.47712 6.301 3.49276 6.046 3.50829 2050 2314.6 2303 2100 2377.7 2559 2150 2440.3 2837 2200 2503.2 3138 2250 2566.4 3464 1726.8 2.555 3.8303 1775.3 2.356 3.8605 1823.8 2.175 3.8901 1872.4 2.012 3.9191 1921.3 1.864 3.9474 Note: The properties P, (relative pressure) and v, (relative specific volume) are dimensionless quantities used in the analysis of isentropic processes, and should not be confused with the properties pressure and specific volume. Source: Kenneth Wark. Thermodynamics, 4th ed. (New York: McGraw-Hill, 1983), pp. 785-86, table A-5. Originally published in J. H. Keenan and J. Kaye, Gas Tables (New York: John Wiley & Sons, 1948). 20 b) A system consists of air initially at state 1, where P = 100 kPa, T = 350K. The system undergoes the following process: Process 1 Process 2 2: adiabatic compression to P2 = 1500 kPa, v = 0.1359 m/kg 3: constant volume cooling to T3 = 300K a) Show P-v diagram b) Determine work and heat transfer for each process using data from Table A-17 and constant specific heat c) Discuss your findings (10 marks) 5:00 PM Fri 17 Nov 934 PROPERTY TABLES AND CHARTS TABLE A-17 Ideal-gas properties of air P T - 81% (+ Th U T h U K kJ/kg P kJ/kg V kJ/kg-K K kJ/kg P kJ/kg V kJ/kg-K 200 199.97 0.3363 142.56 1707.0 1.29559 580 586.04. 14.38 419.55 115.7 2.37348 210 209.97 0.3987 149.69 1512.0 1.34444 220 219.97 0.4690 156.82 1346.0 1.39105 590 596.52 15.31 427.15 110.6 2.39140 600 607.02 16.28 434.78 105.8 2.40902 230 230.02 0.5477 164.00 1205.0 1.43557 610 617.53 17.30 442.42 101.2 2.42644 240 240.02 0.6355 171.13 1084.0 1.47824 620 628.07 18.36 450.09 96.92 2.44356 250 250.05 0.7329 178.28 979.0 1.51917 630 638.63 19.84 457.78 92.84 2.46048 260 260.09 0.8405 185.45 270 270.11 0.9590 192.60 280 280.13 1.0889 199.75 285 285.14 1.1584 203.33 290 290.16 1.2311 206.91 295 295.17 1.3068 210.49 647.9 1.68515 298 298.18 1.3543 212.64 631.9 1.69528 300 300.19 1.3860 214.07 621.2 1.70203 305 305.22 1.4686 217.67 596.0 1.71865 310 310.24 1.5546 221.25 572.3 1.73498 315 315.27 1.6442 224.85 549.8 1.75106 320 320.29 1.7375 228.42 528.6 1.76690 325 325.31 1.8345 232.02 508.4 1.78249 330 330.34 1.9352 235.61 489.4 1.79783 340 340.42 2.149 242.82 454.1 1.82790 350 350.49 2.379 250.02 422.2 1.85708 360 360.58 2.626 257.24 393.4 1.88543 370 370.67 2.892 264.46 367.2 1.91313. 380 380.77 3.176 271.69 343.4 1.94001 390 390.88 3.481 278.93 321.5 1.96633 400 400.98 3.806 286.16 301.6 1.99194 410 411.12 4.153 293.43 283.3 2.01699 420 421.26 4.522 300.69 266.6 2.04142 430 431.43 4.915 307.99 251.1 2.06533 440 441.61 5.332 315.30 236.8 2.08870 450 451.80 5.775 322.62 223.6 2.11161 460 462.02 6.245 329.97 211.4 2.13407 470 472.24 6.742 337.32 200.1 2.15604 480 482.49 7.268 344.70 189.5 2.17760 490 492.74 7.824 352.08 179.7 2.19876 500 503.02 8.411 359.49 170.6 2.21952 510 513.32 9.031 366.92 162.1 2.23993 520 523.63 9.684 374.36 154.1 2.25997 530 533.98 10.37 381.84 146.7 2.27967 540 544.35 11.10 550 555.74 11.86 560 565.17 12.66 570 575.59 13.50 887.8 1.55848 640 649.22 20.64 465.50 88.99 2.47716 808.0 1.59634 650 659.84. 21.86 473.25 85.34 2.49364 738.0 1.63279 706.1 1.65055 660 670.47 23.13 481.01 81.89 2.50985 670 681.14 24.46 488.81 78.61 2.52589 676.1 1.66802 680 691.82 25.85 496.62 75.50 2.54175 690 702.52 27.29 504.45 72.56 2.55731 700 713.27 28.80 512.33 69.76 2.57277 710 724.04 30.38 520.23 67.07 2.58810 720 730 740 756.44 750 767.29 760 778.18 780 800.03 800 821.95 734.82 32.02 528.14 64.53 2.60319 745.62 33.72 536.07 62.13 2.61803 35.50 544.02 37.35 551.99. 57.63 39.27 560.01 43.35 576.12 59.82 2.63280 2.64737 55.54 2.66176 51.64 2.69013 47.75 592.30 48.08 2.71787 820 843.98 52.59 608.59 44.84 2.74504 840 860 866.08 57.60 624.95 41.85 2.77170 888.27 63.09 641.40 39.12 2.79783 880 910.56 900 920 68.98 657.95 36.61 2.82344 932.93 955.38 75.29 674.58 34.31 2.84856 82.05 691.28 32.18 2.87324 940 977.92 389.34 396.86 139.7 2.29906 133.1 2.31809 404.42 411.97 127.0 2.33685 121.2 2.35531 960 1000.55 980 1023.25 105.2 1000 1046.04 114.0 1020 1068.89 123.4 1040 1091.85 133.3 1060 1114.86 143.9 1080 1137.89 155.2 1100 1161.07 167.1 1120 1184.28 179.7 1140 1207.57 193.1 1160 1230.92 207.2 1180 1254.34 222.2 1200 1277.79 238.0 1220 1301.31 254.7 1240 1324.93 272.3 89.28 708.08 97.00 725.02 741.98 758.94 25.17 2.96770 776.10 23.72 2.99034 793.36 23.29 3.01260 810.62 21.14 3.03449 827.88 19.98 3.05608 845.33 18.896 3.07732 862.79 880.35 897.91 30.22 2.89748 28.40 2.92128 26.73 2.94468 17.886 3.09825 16.946 3.11883 16.064 3.13916 915.57 15.241 3.15916 933.33 14.470 3.17888 951.09 968.95 13.747 3.19834 13.069 3.21751 28 5:00 PM Fri 17 Nov T. TABLE A-17 Ideal-gas properties of air (Concluded) 935 APPENDIX 1 - 81% T h u T h K kJ/kg P, kJ/kg V, kJ/kg-K K kJ/kg 1260 1348.55 290.8 986.90 1280 1372.24 310.4 1300 1395.97 330.9 1320 1419.76 352.5 1340 1443.60 375.3. 1360 1467.49 399.1 1380 1491.44 424.2 1400 1515.42 450.5 1420 1539.44 478.0 1440 1563.51 506.9 1460 1587.63 537.1 1480 1611.79 568.8 1004.76 1022.82 1040.88 1058.94 1077.10 1095.26 12.435 3.23638 11.835 3.25510 11.275 3.27345 10.747 3.29160 10.247 3.30959 9.780 3.32724 9.337 3.34474 1113.52 8.919 3.36200 1131.77 8.526 3.37901 1150.13 8.153 3.39586 1168.49 7.801 3.41247 1186.95 7.468 3.42892 1600 1757.57 1620 1782.00 1640 1806.46 1660 1830.96 1680 1855.50 1700 1880.1 1750 1941.6 1800 2003.3 1310 1850 2065.3 1475 1900 2127.4 1655 1950 2189.7 1852 2000 2252.1 2068 kJ/kg P 1298.30 791.2 5.804 3.52364 834.1 1316.96 5.574 3.53879 878.9 1335.72 5.355 3.55381. 925.6 1354.48 5.147 3.56867 974.2 1373.24 4.949 3.58335 1025 1392.7 4.761 3.5979 1161 1439.8 4.328 3.6336 1487.2 3.994 3.6684 1534.9 V kJ/kg-K 3.601 3.7023 1582.6 3.295 3.7354 1630.6 3.022 3.7677 1678.7 2.776 3.7994 1500 1635.97 601.9 1520 1660.23 636.5 1540 1684.51 672.8 1242.43 1560 1708.82 710.5 1260.99 1580 1733.17 750.0 1279.65 1205.41 7.152 3.44516 1223.87 6.854 3.46120 6.569 3.47712 6.301 3.49276 6.046 3.50829 2050 2314.6 2303 2100 2377.7 2559 2150 2440.3 2837 2200 2503.2 3138 2250 2566.4 3464 1726.8 2.555 3.8303 1775.3 2.356 3.8605 1823.8 2.175 3.8901 1872.4 2.012 3.9191 1921.3 1.864 3.9474 Note: The properties P, (relative pressure) and v, (relative specific volume) are dimensionless quantities used in the analysis of isentropic processes, and should not be confused with the properties pressure and specific volume. Source: Kenneth Wark. Thermodynamics, 4th ed. (New York: McGraw-Hill, 1983), pp. 785-86, table A-5. Originally published in J. H. Keenan and J. Kaye, Gas Tables (New York: John Wiley & Sons, 1948). 20
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
"internet radios" for streaming audio, and personal video recorders and players. Describe design and evaluation processes that could be used by a start-up company to improve the usability of such...
-
Consider three (ideally other) countries for which evidence features here. What are the key influences on cross-national comparative variation in the place and role of the HRM function?
-
You serve as the executor for the estate of Willy Rock. The following transactions occur during June 2013: a. Willy's estate included a municipal bond with a fair value of $500,000. On the date of...
-
Use the Trapezoid Rule to approximate with R = 2, 4, and 8. For each value of R, take n = 4, 8, 16, and 32, and compare approximations with successive values of n. Use these approximations to...
-
What is the difference between a predator and a situational (accidental) fraudster?
-
County Beverage Drive-Thru, Inc., operates a chain of beverage supply stores in Northern Illinois. Each store has a single service lane; cars enter at one end of the store and exit at the other end....
-
5. Suppose that acts on density operators for Q with Kraus representation () - k=1 Let U M, (C), and define operators = 1, k BkUkjAj. , Show that B,..., B., are also Kraus operators for E. 6. Even...
-
Labour force data for country Eturia (all in millions) are shown in the table below. Enter your responses below rounded to one decimal place. 2013 2014 2015 Population (millions) 20 20.5 21.02...
-
Sales VC o U-Develop had 3 departments: Print Department. . Camera Department Frame Department o Print Department is currently operating at a loss Cameras CM Less: FC Rent Salaries Selling & Admin....
-
The Future of Auditing Over the next few years, investors and regulators will expect more predictive analytics in both the official audit opinion and periodic business performance updates. To that...
-
Perform a horizontal analysis providing both the amount and percentage change. (Round place. Decreases should be indicated by a minus sign.) Cash Accounts receivable Inventory Long-term assets Total...
-
How could you provide redundancy ( ( additional to the main PtP wireless link ) ) to a temporary remote wireless telemetry station? Keep cost in mind.
-
O a. Unearned revenue O b. Accounts payable O c. Product warranty is the money received from a customer for goods or services that have yet to be delivered or produced.
-
Calliastra has five shirts, six skirts, seven pairs of trousers, and eight dresses. She can select either a skirt or a pair of trousers to go with a shirt, or she can wear only a dress. How many...
-
Determine the values of the given trigonometric functions directly on a calculator. The angles are approximate. tan 0.8035
-
Make up and solve an original problem. The guidelines are given in Problem VO do Ub UR VO
-
Consider the following system of gas-phase reactions: AXrX=k1CA1/2k1=0.004(mol/dm3)1/2min1ABrB=k2CAk2=0.3 min1AYrY=k3CA2k3=0.25 dm3/molmin B is the desired product, and X and Y are foul pollutants...
-
Consider a cylindrical batch reactor that has one end fitted with a frictionless piston attached to a spring. The reaction A + B 8C A cylindrical batch reactor is placed. A frictionless piston on...
-
Identify a key position that you intend to make during your presentation. Using Table 14.2 as a guide, write out how you could use the PREP (PositionReasonsExamplePosition) method to effectively...
-
Identify a topic of interest. Create a storyboard to outline the titles, content, and related story line of your PowerPoint slides.
-
Create an electronic slide presentation for your topic of interest. Ensure that it conveys your key messages effectively.

Study smarter with the SolutionInn App


