The weak base ethylamine (CH5NH (aq)) has a K of 6.4 x104. a. Write the equilibrium...
Fantastic news! We've Found the answer you've been seeking!
Question:

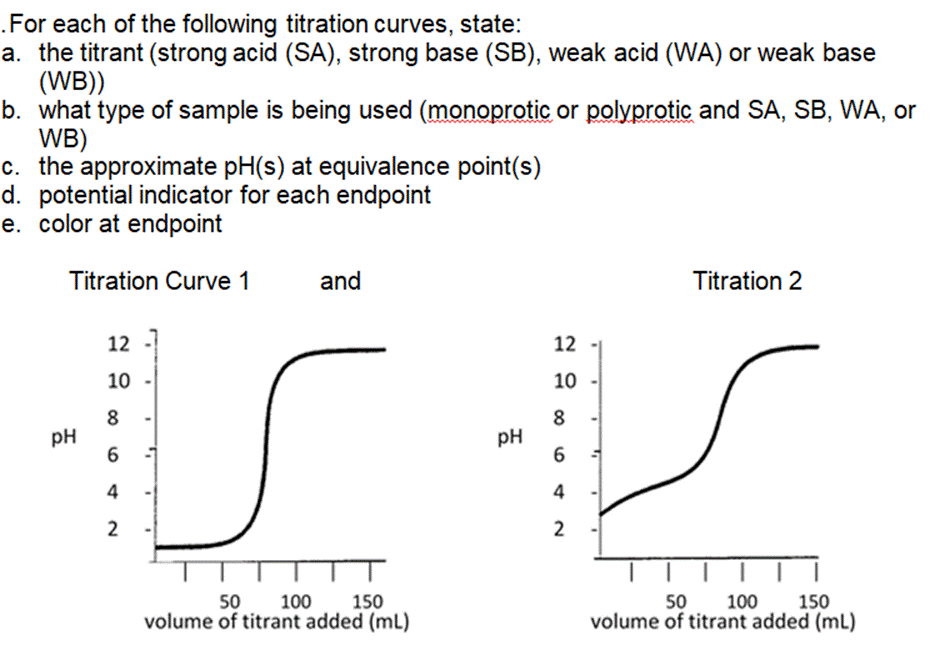
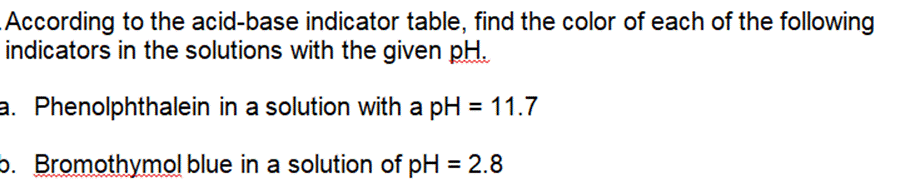
Transcribed Image Text:
The weak base ethylamine (C₂H5NH₂ (aq)) has a K₂ of 6.4 x104. a. Write the equilibrium equation for the ionization of ethylamine. b. What concentration of C₂H5NH2 (aq) is required to produce an ethylamine solution with a pH of 12.102? .For each of the following titration curves, state: a. the titrant (strong acid (SA), strong base (SB), weak acid (WA) or weak base (WB)) b. what type of sample is being used (monoprotic or polyprotic and SA, SB, WA, or WB) c. the approximate pH(s) at equivalence point(s) d. potential indicator for each endpoint e. color at endpoint Titration Curve 1 PH 12 10 8 6 4 2 and v T T 50 150 100 volume of titrant added (ml) PH 12 10 8 6 4 2 Titration 2 50 100 150 volume of titrant added (ml) According to the acid-base indicator table, find the color of each of the following indicators in the solutions with the given pH. a. Phenolphthalein in a solution with a pH = 11.7 . Bromothymol blue in a solution of pH = 2.8 The weak base ethylamine (C₂H5NH₂ (aq)) has a K₂ of 6.4 x104. a. Write the equilibrium equation for the ionization of ethylamine. b. What concentration of C₂H5NH2 (aq) is required to produce an ethylamine solution with a pH of 12.102? .For each of the following titration curves, state: a. the titrant (strong acid (SA), strong base (SB), weak acid (WA) or weak base (WB)) b. what type of sample is being used (monoprotic or polyprotic and SA, SB, WA, or WB) c. the approximate pH(s) at equivalence point(s) d. potential indicator for each endpoint e. color at endpoint Titration Curve 1 PH 12 10 8 6 4 2 and v T T 50 150 100 volume of titrant added (ml) PH 12 10 8 6 4 2 Titration 2 50 100 150 volume of titrant added (ml) According to the acid-base indicator table, find the color of each of the following indicators in the solutions with the given pH. a. Phenolphthalein in a solution with a pH = 11.7 . Bromothymol blue in a solution of pH = 2.8
Expert Answer:
Answer rating: 100% (QA)
SOLUTION a The equilibrium equation for the ionization of ethylamine C2H5NH2 can be written as follows C2H5NH2 aq H2O l C2H5NH3 aq OH aq In this equat... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
What rms voltage is required to produce an rms current of 2.1 A in a 66-mH inductor at a frequency of 25 Hz?
-
What four conditions are required to produce an ideal metallurgical bond?
-
What pressure of nitrogen gas is required to produce a collision rate of 5.00 x 1019 S-1 at 525 K on a circular surface of diameter 2.0 mm?
-
In Exercises simplify the ratio of factorials. (n + 1)! n!
-
What is meant by due diligence? How does a traditional registration differ from a shelf registration?
-
Students scores on a test in a public administration course follow a normal distribution with a mean of 150 points and a standard deviation of 12. One student who scored 161 on the test and received...
-
What is the proper accounting for depreciation when new or additional information becomes available that causes a company to change its estimates of useful life or residual value?(Appendix)
-
Statement of Budgetary Resources. The trial balance of the Federal Science Administration, as of August 31, 2011, follows: Required Prepare a statement of budgetary resources for the 11 months ended...
-
Solve the LP problem. If no optimal solution exists, indicate whether the feasible region is empty or the objective function is unbounded. (Enter EMPTY if the region is empty. Enter UNBOUNDED if the...
-
The proposed rates were not in the range the CEO expected given the pricing analysis. The CEO has asked the pricing actuary to verify the total projected loss cost excluding potential large storm...
-
Consider a suburban mall that generates total lease income of $40m per year. Operating expenses of running the mall are $7m per year. The mall is financed by an interest-only commercial mortgage with...
-
Instead of 1 loan been disbursed, assumed a portfolio of loans been disbursed with % distribution of tenure (on amount), interest rate and overall disbursal as follows : Overall portfolio disbursal...
-
Solve the following exact differential equation: (y cos(x) + 2xe) + (sin(x) + xev - 1)y = 0
-
A fixed income arbitrage hedge fund focuses on Treasury bond trading. Bond A is a ten-year Treasury bond (Notes) which was issued eight years ago in a low interest environment. Bond B is a three-year...
-
What are the mechanisms of cellular senescence, and how do senescent cells contribute to aging and the development of age-related diseases ?
-
The diameters of the main rotor and tail rotor of a single - engine helicopter are 7 . 5 5 m and 1 . 0 4 m , respectively. The respective rotational speeds are 4 4 6 rev / min and 4 , 1 4 0 rev / min...
-
The following is an LP model designed to be analyzed using Solver. The dialog box for the Evaluate Formula auditing tool, when applied to the formula in Cell 11, and the successive step-by-step output
-
Beginning with a country that has a trade deficit, demonstrate graphically what will happen to a countrys potential output with globalization if that countrys costs of production fall. Explain your...
-
In a friendly game of handball, you hit the ball essentially at ground level and send it toward the wall with a speed of 16 m/s at an angle of 37 above the horizontal. (a) What amount of time is...
-
The light used in this experiment has a wavelength of 511 nm. (a) If the width of the slit is decreased, will the angle to the first dark fringe above the central bright fringe increase or decrease?...
-
A uniform electric field with a magnitude of 6860 N/C points in the positive x direction. Find the change in electric potential energy when a +14.5-(C charge is moved 5.75 cm in (a) The positive x...
-
A firm is earning $8 per year. It can earn .20 (after corporate tax) on reinvested funds. Based on a constant dividend the firm's value is $40.27. If the funds are reinvested the firm's value after...
-
What does the practice and theory of international audit include aside from knowledge of ISAs?
-
Evaluate this quote: Every international business, large or small, should have an annual audit by an independent auditor. Why must an auditor review the financial statements of a company each year?

Study smarter with the SolutionInn App


