B. Calculate AHsolution (Na2S04 10 H20). (0.4) oehoga ded C. Calculate your experimental value for AHnydration...
Fantastic news! We've Found the answer you've been seeking!
Question:

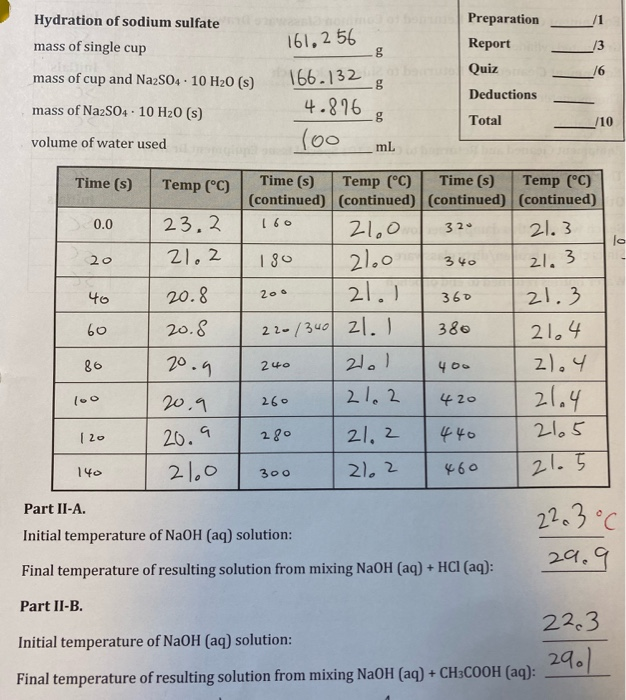
Transcribed Image Text:
B. Calculate AH°solution (Na2S04· 10 H20). (0.4) oehoga ded C. Calculate your experimental value for AH°nydration (NazSO4) applying Hess's law. Show the addition of the appropriate net ionic equations used to obtain AH°hydration (NazSO4). (0.5) Hydration of sodium sulfate Preparation 161,2 56 g mass of single cup Report 13 Quiz 16 mass of cup and NazS04 · 10 H20 (s) 166.132 g Deductions mass of NazS04· 10 H20 (s) 4.876 Total /10 (00 volume of water used mL Time (s) Time (s) Temp (°C) (continued) (continued) (continued) (continued) 21. 3 Time (s) Temp (°C) Temp (°C) 0.0 23,2 160 21.0 320 21.0 21.1 21.2 180 20 21.3 340 20.8 21.3 40 200 360 20.8 22-/340 21. 380 21,4 60 20.9 21.1 2).4 86 240 400 21.4 2165 20.9 21.2 420 l00 260 21, 2 440 20.9 280 |20 21.0 21, 2 21.5 140 300 460 Part II-A. 22.3°C Initial temperature of NaOH (aq) solution: 29.9 Final temperature of resulting solution from mixing NaOH (aq) + HCI (aq): Part II-B. 22.3 Initial temperature of NaOH (aq) solution: 29.1 Final temperature of resulting solution from mixing NaOH (aq) + CH3COOH (aq): B. Calculate AH°solution (Na2S04· 10 H20). (0.4) oehoga ded C. Calculate your experimental value for AH°nydration (NazSO4) applying Hess's law. Show the addition of the appropriate net ionic equations used to obtain AH°hydration (NazSO4). (0.5) Hydration of sodium sulfate Preparation 161,2 56 g mass of single cup Report 13 Quiz 16 mass of cup and NazS04 · 10 H20 (s) 166.132 g Deductions mass of NazS04· 10 H20 (s) 4.876 Total /10 (00 volume of water used mL Time (s) Time (s) Temp (°C) (continued) (continued) (continued) (continued) 21. 3 Time (s) Temp (°C) Temp (°C) 0.0 23,2 160 21.0 320 21.0 21.1 21.2 180 20 21.3 340 20.8 21.3 40 200 360 20.8 22-/340 21. 380 21,4 60 20.9 21.1 2).4 86 240 400 21.4 2165 20.9 21.2 420 l00 260 21, 2 440 20.9 280 |20 21.0 21, 2 21.5 140 300 460 Part II-A. 22.3°C Initial temperature of NaOH (aq) solution: 29.9 Final temperature of resulting solution from mixing NaOH (aq) + HCI (aq): Part II-B. 22.3 Initial temperature of NaOH (aq) solution: 29.1 Final temperature of resulting solution from mixing NaOH (aq) + CH3COOH (aq):
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
4 cubed + 10 X 20 + 8 squared 23
-
What is the shear capacity of the RC beam described below considering the steel reinforcement and using the formula: VRsyAw 2fyd cot 8/s The shear reinforcement in the beam is provided by sets of...
-
To heat 1 cup of water (250 cm3) to make coffee, you place an electric heating element in the cup. As the water temperature increases from 20C to 65C, the temperature of the heating element remains...
-
Ted and Joan Kramer have assets of $290,000 and are planning for Joan to enter a nursing home. The Kramers would like to apply for Medicaid benefits to pay for Joan's care. What should they do to...
-
Discuss the brand personalities of Kodak, Mountain Dew, and Prudential Insurance, and describe how these brand personalities were created.
-
Company name Calix Limited CXL Millions 2023 2022 2021 2020 2019 2018 Income Statement Data Net Sales revenue 18.6 18.5 19.2 14.1 3.3 4.1 Cost of goods sold 12.6 13.3 14.2 10.7 2.0 2.7 Gross Profit...
-
The following data were obtained from the year-end financial statements of The Arcadia Company: Required Compute the following ratios for The Arcadia Company: (1) Return on Sales, (2) Return on...
-
The following account balances were included in Bromley Companys balance sheet on December 31, 2015: Land............ $ 100,000 Land improvements........ 20,000 Buildings........... 300,000 Machinery...
-
Paul and Lee form a partnership. Paul's equity from capital contributions is $80,000 and Lee's equity from capital contributions is $100,000. The company has $200,000 in total liabilities. What would...
-
Prepare a simple tax provision and the journal entries to calculate the current and the deferred tax provision (benefit) using the following data: a. ABC, Inc. reported pre-tax book income of...
-
The following data consists of birth weights (pounds) of a sample of newborn babies at a local hospital: Calculate the following: a. Range Range= 0 9.5 9.6 6.4 8.8 8.8 8.9 8.4 8.7 7.5 8.4 8.2 9.4
-
Boat for $3M. He was to return his acceptance in the course of Post. By a letter also dated the 1st February, Mark informed Joe that he was selling his Toyota Rav 4 car for $1, 500,000.00. He stated...
-
A rainstorm produced a 90-million-volt lightning bolt, but only for 0.004 seconds. The bolt struck a conducting pipe and the current passing through the pipe was 400 amperes. (a) What was the total...
-
A field party is capable of making taping observations with a standard deviation of 0.010 ft per 100-ft tape length. What standard deviation would be expected in a distance of 200 ft taped by this...
-
Approximately how long will it take for $1000 to double if the interest rate is 7%?
-
Based on the information provided. Calculate the operating profit. Water and electricity R8 000 Depreciation R11 500 Sales R54 000 Trade receivables R20 000 Trade creditors R12 000 Rent expense R2 500
-
1.What type of property can be used for a 1031 Tax Exchange and why would such an exchange be used? and Why do investors care how the IRS classifies property either as real or personal property.
-
Calculate the electrical conductivity of a fiber-reinforced polyethylene part that is reinforced with 20 vol % of continuous, aligned nickel fibers.
-
Suppose that 2.5 mmol Ar (g) occupies 72 dm3 at 298 K and expands to 100 dm3, Calculate G for the process.
-
The diffusion coefficient of I in hexane at 25C is 4.05 x 10-9 m2 S-1. Estimate the time required for an iodine molecule to have a root mean square displacement of 1.0 cm.
-
In the gas-phase reaction A + B ;:='0C + 2 D, it was found that, when 2.00 mol A, 1.00 mol B, and 3.00 mol D were mixed and allowed to come to equilibrium at 25C, the resulting mixture contained 0.79...
-
The equity method is often described as a one-line consolidation. Discuss the similarities of the equity method and consolidated reporting. Discuss how these methods differ in their financial...
-
Accumulated other comprehensive income (AOCI) appears as a subsection of shareholders equity on the balance sheet. Discuss the components of AOCI. What information does the AOCI convey to financial...
-
Marketable securities are classified under U.S. GAAP as either trading securities or available-for-sale securities. Regardless of their classification, marketable securities are accounted for at...

Study smarter with the SolutionInn App


