Balance the following acidic skeleton half reaction: Cr07 (aq) Cr+ (aq) A) Enter the coefficient...
Fantastic news! We've Found the answer you've been seeking!
Question:
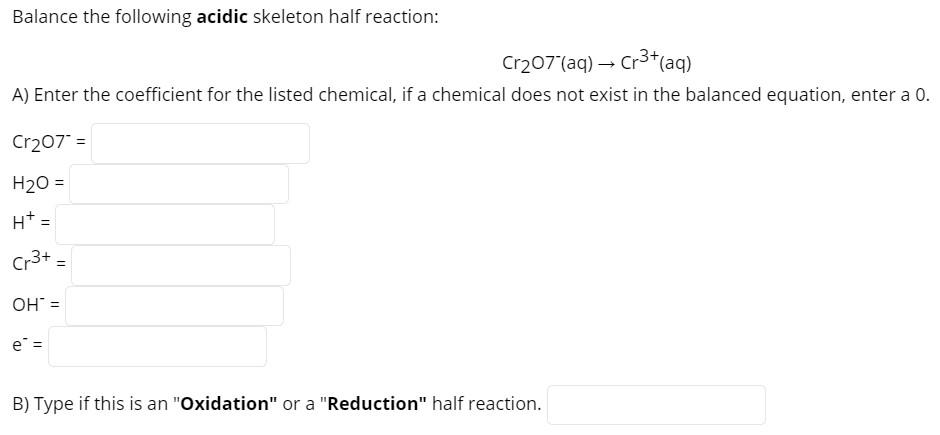
Transcribed Image Text:
Balance the following acidic skeleton half reaction: Cr₂07 (aq) → Cr³+ (aq) A) Enter the coefficient for the listed chemical, if a chemical does not exist in the balanced equation, enter a 0. Cr207" = H₂O = H* = Cr3+ OH = e = B) Type if this is an "Oxidation" or a "Reduction" half reaction. = Balance the following acidic skeleton half reaction: Cr₂07 (aq) → Cr³+ (aq) A) Enter the coefficient for the listed chemical, if a chemical does not exist in the balanced equation, enter a 0. Cr207" = H₂O = H* = Cr3+ OH = e = B) Type if this is an "Oxidation" or a "Reduction" half reaction. =
Expert Answer:
Answer rating: 100% (QA)
CHOHaq CrO aq CHOaq Cr aq Oxidation Half Reaction CH3OH CH... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
A theoretically correct measure of income does not exist in the real world in which accountants must operate. Required a. What is meant by the phrase a theoretically correct measure of income? b. Why...
-
The hydride ion does not exist in water because it has a greater attraction for the hydronium ion than the hydroxide ion does. Write the equation for the reaction that occurs when calcium hydride is...
-
U.S. requirement of reporting comprehensive income does not exist in the United Kingdom, as the WPP Group does not report comprehensive income anywhere in these statements. However, WPP Group does...
-
Consider the pistoncylinder arrangement shown in the sketch below. The gas forces on each side of the piston, assuming there are no frictional forces at the pistoncylinder interface, balance the...
-
Lee Company started the period with cash of $22,000, common stock of $12,000, and retained earnings of $10,000. Lee engaged in the following transactions in 2013: Transaction During 2013 1. On...
-
A U.S. firm must make a payment of 1 million yen to a Japanese firm that has sold the U.S. firm sets of Japanese baseball-player trading cards. The U.S. firm begins with a dollar checking account....
-
The Forensic Accounting Box of this chapter noted that forensic accounting techniques are being used to help pick stocks for investing. How might identifying accounting concerns be useful for...
-
Joseph Biggs owns his own ice cream truck and lives 30 miles from a Florida beach resort. The sale of his products is highly dependent on his location and on the weather. At the resort, his profit...
-
A beer manufacturer noticed that the cost of hops increased four times in the last year. A description of this would be found in the _ _ _ _ _ _ _ _ Blank section of the SWOT analysis.
-
The following rates were observed for a first-order, irreversible reaction, carried out on a spherical catalyst: dp = 0.625 cm robs=0.09 mol/gcat.hr dp = 0.10 cm Tobs = 0.275 mol/gcat.hr Strong...
-
Summary : Write a summary of what the financial statements indicate about the companys financial health and performance. Purpose : What kind of information would these financial statements provide to...
-
Assume that, upon separation from his wife or partner, a man who has believed himself to be the father of the child he has been raising suddenly has doubts and wishes to have genetic testing done to...
-
How are constitutive equations used in vibrations modeling?
-
Scott and Heidi were married in 1990. Heidi gave birth to five children during their marriage. Scott initiated divorce proceedings in October 2012. Custody and placement of Scott and Heidi's two...
-
Assume that a man and woman who know each other casually have sexual intercourse one time. As a result, she becomes pregnant and decides to have the baby. Based on this fleeting moment of intimacy,...
-
Thinking back to the case of Michael H. v. Gerald D., do you think the biological father should have been permitted to establish his paternal rights, or do you think the Court was right to bar his...
-
(Java)Music playlist: - A program needs an array that can hold at least 50-100 songs. - There is 3 function: search the titles, sort the titles and randomize - Please include a comment to explain...
-
Using (1) or (2), find L(f) if f(t) if equals: t cos 4t
-
The first step in the preparation of lead from its ore (galena, PbS) consists of roasting the ore. 2PbS(s) + 3O2(g) 2SO2(g) + 2PbO(s) Calculate the standard enthalpy change for this reaction, using...
-
Consider two flasks that contain different pure liquids at 20C. The liquid in one flask, substance A, has a molar mass of 100 g/mol and has hydrogen bonding. The liquid in the other flask, substance...
-
Ozone decomposes to oxygen gas. 2O3(g) 3O2(g) A proposed mechanism for this decomposition is What is the rate law derived from this mechanism? O3 2 + O (fast, equilibrium) slow)
-
The following summarized data relate to Robert Corporation's current operations: Required Prepare a multiple-step income statement for Robert Corporation for the year. Include earnings per share...
-
Net sales, net income, and total asset figures for Janice Controls, Inc., for five consecutive years are given below (Janice manufactures pollution controls): Required a. Calculate trend percentages,...
-
Presented below is selected information for Turner Company: Required a. Calculate the following ratios for 2019. The 2018 results are given for comparative purposes. b. Comment on the changes between...

Study smarter with the SolutionInn App


