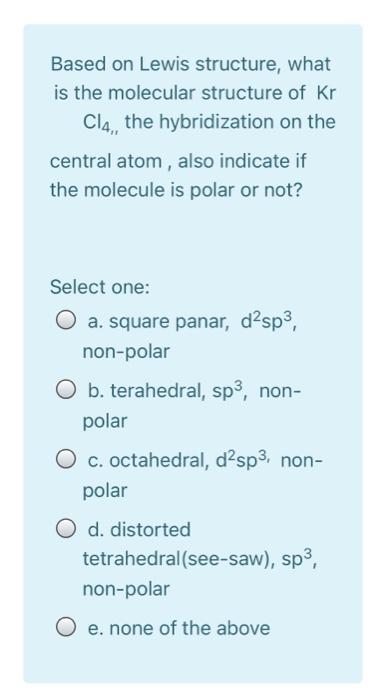
Based on Lewis structure, what is the molecular structure of Kr Cl4,, the hybridization on the...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Based on Lewis structure, what is the molecular structure of Kr Cl4,, the hybridization on the central atom, also indicate if the molecule is polar or not? Select one: a. square panar, d²sp³, non-polar O b. terahedral, sp³, non- polar O c. octahedral, d²sp³, non- polar O d. distorted tetrahedral (see-saw), sp³, non-polar O e. none of the above Based on Lewis structure, what is the molecular structure of Kr Cl4,, the hybridization on the central atom, also indicate if the molecule is polar or not? Select one: a. square panar, d²sp³, non-polar O b. terahedral, sp³, non- polar O c. octahedral, d²sp³, non- polar O d. distorted tetrahedral (see-saw), sp³, non-polar O e. none of the above
Expert Answer:
Answer rating: 100% (QA)
Ans a square planar dsp nonpolar In KrCl4 molecule the Kr atom is bonded to 4 Cl ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What is the molecular mass of each substance? 1. SO2 2. PF3
-
What is the molecular formula of a hydrocarbon that has 1 triple bond, 2 double bonds, 1 ring, and 32 carbons?
-
What is the molecular orbital configuration of HeH+? Do you expect the ion to be stable?
-
Austin and Anya Gould are a middle-aged couple with two childrenRusty, age 13, and Sam, age 11whom they adopted this year. They also bought a new home in the area to give the children a yard in which...
-
How do traditional commerce and web-based commerce differ? What aspects of traditional shopping do you prefer over online shopping? How has mobile computing altered web-based commerce?
-
A person looks at a gem with a jewelers loupea converging lens that has a focal length of 12.5 cm. The loupe forms a virtual image 30.0 cm from the lens. (a) Determine the magnification. Is the image...
-
After returning from a skiing vacation in Vermont, Leslie Adel came down with Legionnaires Disease. He claimed it was from the water drunk at the ski resort and provided by Greensprings of Vermont...
-
Chapman Company, a major retailer of bicycles and accessories, operates several stores and is a publicly traded company. The comparative balance sheet and income statement for Chapman as of May 31,...
-
A student performs a double-slit experiment using two slits spaced 0.20 mm apart and located 1.50 m from the screen. The monochromatic light source creates an interference pattern in which the...
-
Griffin and Lasky, Inc. (G&L), supplies industrial automation equipment and machine tools to the automotive industry. G&L recognizes revenue on its long-term contracts over time. Customer orders have...
-
Evaluate the following limits: 1 lim x-16 (x-4 - x-16) sin(10x) limx0 sin(5x) 1-cos(x) limx0 x
-
Choose a product or service from these examples: Fast food Class of consumer product Personal care Automobile Research online to analyze how that product or service has been advertised. Prepare...
-
Assume that the following balance sheets are stated at book value. Suppose the fair market value of acquired firm's fixed assets is $12,000 versus the book value shown. The acquiring firm pays...
-
The company usings standard costs for inventory and are billed actual costs. When inventory is received, the company makes the following: Dr. Inventory $Standard Cr. Inventory Suspense $Standard When...
-
The Black Horse is currently considering a project that will produce cash inflows of $11,000 a year for three years followed by $6,500 in Year 4. The cost of the project is $38,000. What is the...
-
Arthur holds a well-diversified portfolio of high quality, large-cap stocks. The current value of Arthur's portfolio is RM600,000 but he is concerned that the market is heading for a big fall over...
-
Steady As She Goes Incorporated will pay a year-end dividend of $2.60 per share, Investors expect the dividend to grow at a rate of 6% indefinitely. a. If the stock currently sells for $26.00 per...
-
What does non-recourse financing mean?
-
A certain oxygen atom has the electron configuration 1s22s22px22py2. How many unpaired electrons are present? Is this an excited state for oxygen? In going from this state to the ground state, would...
-
Consider the general reaction aA + bB cC and the following average rate data over a specific time period t: A/t = 0.0080 mol L-1s-1 B/t = 0.0120 mol L-1s-1 C/t = 0.0160 mol L-1s-1 Determine a set...
-
Why does the molecular orbital model do a better job in explaining the bonding in NO- and NO than the hybrid orbital model?
-
Emily throws a soccer ball out of her dorm window to Allison, who is waiting below to catch it. If Emily throws the ball at an angle of 30 below horizontal with a speed of 12 m/s, how far from the...
-
In the 2016 Olympics in Rio, after the 50 m freestyle competition, a problem with the pool was found. In lane 1 there was a gentle 1.2 cm/s current flowing in the direction that the swimmers were...
-
In punting a football, the kicker tries to maximize both the distance of the kick and its hang timethe time that the ball is in the air. A kicker gets off a great punt with a hang time of 5.0 s that...

Study smarter with the SolutionInn App


