Chapter 10: Nucleophilic Substitution Reaction II The Kinetics of Solvolysis- SN1 Reaction Write Prelab: Purpose, Introduction,...
Fantastic news! We've Found the answer you've been seeking!
Question:

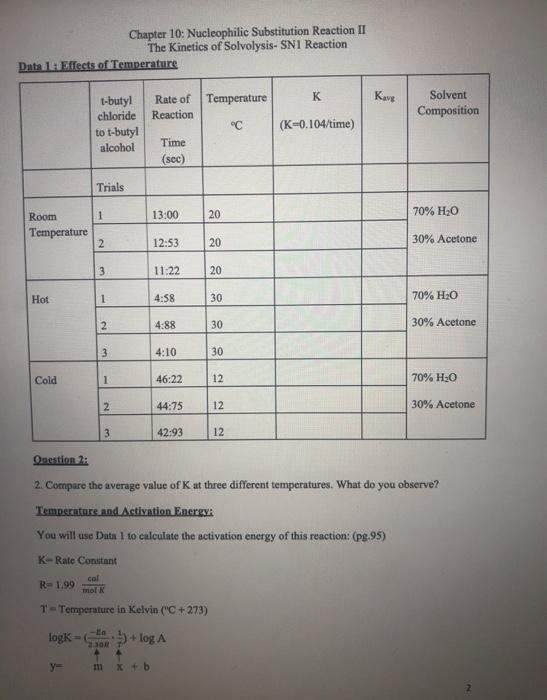

Transcribed Image Text:
Chapter 10: Nucleophilic Substitution Reaction II The Kinetics of Solvolysis- SN1 Reaction Write Prelab: Purpose, Introduction, Procedures, and Table of Physical Constants Explanation: In the experiment, we are going to examine the rate of tert-Butyl alcohol+ Cr tert-Butyl chloride + H₂O H₂O in this reaction is a solvent and nucleophile. Sometimes, in an SN1 reaction the solvent acts as the nucleophile. This is called solvolysis. Factors which affect the rate of reaction: A. The concentration of the reactants B. The solvent C. The temperature Concentration of the Reactants: A. The reaction involves two separate steps. First the leaving group departs, and then the nucleophile attacks the carbocation. The first step is the rate-determining step. The rate law for the reaction is as follows: Rate = k [tert-butyl chloride] The rate law doesn't involve water so changing the concentration of the nucleophile has no effect on the rate The rate law does involve [tert-butyl chloride], so increasing the concentration of the substrate increases the rate. Likewise, decreasing the concentration of the substrate decreases the rate. Solvent: B. See explanation for A. Temperature: C. A rise in temperature should accelerate a bond breaking reaction. The rate determining step involves bond breaking. So, we would predict that an increase in temperature should result in production of more tert-butyl and thus increases the rate of the reaction. Question 1: 1. Write the mechanism of the reaction. Data 1: Effects of Temperature Room Temperature Hot Cold 1-butyl chloride to t-butyl alcohol Trials y= 1 2 3. 1 2 3 1 Chapter 10: Nucleophilic Substitution Reaction II The Kinetics of Solvolysis- SN1 Reaction 2 3 Rate of Temperature Reaction Time (sec) m x + b 13:00 12:53 11:22 4:58 4:88 4:10 46:22 44:75 42:93 20 20 T-Temperature in Kelvin ("C +273) logk=+log A 20 30 30 30 12 12 12 °℃ K (K-0.104/time) Kavg Solvent Composition 70% H₂O 30% Acetone 70% H₂O 30% Acetone Question 2: 2. Compare the average value of K at three different temperatures. What do you observe? Temperature and Activation Energy: You will use Data 1 to calculate the activation energy of this reaction: (pg.95) K-Rate Constant col R=1.99 mol K 70% H₂O 30% Acetone Question 3: 3. Plot-log Kang (y axis) vs (x axis) for the three temperatures. Use Excel. Question 4: 4. Why did you have to make graphs? t-butyl chloride to .m-slope 5. Calculate the energy of activation with units. Data 2: Effect of solvent (Read pg. 94) Rate-[tert-Butyl chloride] t-butyl alcohol Trials 1 2 Ea 2.30R 3 Chapter 10: Nucleophilic Substitution Reaction II The Kinetics of Solvolysis- SN1 Reaction Time (sec) 9.97 8.03 7.69 Temperature °C 20 20 20 K- 0.104/time Kava Solvent composition 9. Does water participate directly in the reaction? 10. Predict the effect if the solvent were 60% water: 40% acetone. 80% H₂0 20% Acetone Use Data 2 to Answer Questions: 6. Use average to determine K. 7. What is the effect observed from increasing the water from 70% to 80% of the solvent mixture? What about the substrate? 8. What is the function of acetone? I Chapter 10: Nucleophilic Substitution Reaction II The Kinetics of Solvolysis- SN1 Reaction Write Prelab: Purpose, Introduction, Procedures, and Table of Physical Constants Explanation: In the experiment, we are going to examine the rate of tert-Butyl alcohol+ Cr tert-Butyl chloride + H₂O H₂O in this reaction is a solvent and nucleophile. Sometimes, in an SN1 reaction the solvent acts as the nucleophile. This is called solvolysis. Factors which affect the rate of reaction: A. The concentration of the reactants B. The solvent C. The temperature Concentration of the Reactants: A. The reaction involves two separate steps. First the leaving group departs, and then the nucleophile attacks the carbocation. The first step is the rate-determining step. The rate law for the reaction is as follows: Rate = k [tert-butyl chloride] The rate law doesn't involve water so changing the concentration of the nucleophile has no effect on the rate The rate law does involve [tert-butyl chloride], so increasing the concentration of the substrate increases the rate. Likewise, decreasing the concentration of the substrate decreases the rate. Solvent: B. See explanation for A. Temperature: C. A rise in temperature should accelerate a bond breaking reaction. The rate determining step involves bond breaking. So, we would predict that an increase in temperature should result in production of more tert-butyl and thus increases the rate of the reaction. Question 1: 1. Write the mechanism of the reaction. Data 1: Effects of Temperature Room Temperature Hot Cold 1-butyl chloride to t-butyl alcohol Trials y= 1 2 3. 1 2 3 1 Chapter 10: Nucleophilic Substitution Reaction II The Kinetics of Solvolysis- SN1 Reaction 2 3 Rate of Temperature Reaction Time (sec) m x + b 13:00 12:53 11:22 4:58 4:88 4:10 46:22 44:75 42:93 20 20 T-Temperature in Kelvin ("C +273) logk=+log A 20 30 30 30 12 12 12 °℃ K (K-0.104/time) Kavg Solvent Composition 70% H₂O 30% Acetone 70% H₂O 30% Acetone Question 2: 2. Compare the average value of K at three different temperatures. What do you observe? Temperature and Activation Energy: You will use Data 1 to calculate the activation energy of this reaction: (pg.95) K-Rate Constant col R=1.99 mol K 70% H₂O 30% Acetone Question 3: 3. Plot-log Kang (y axis) vs (x axis) for the three temperatures. Use Excel. Question 4: 4. Why did you have to make graphs? t-butyl chloride to .m-slope 5. Calculate the energy of activation with units. Data 2: Effect of solvent (Read pg. 94) Rate-[tert-Butyl chloride] t-butyl alcohol Trials 1 2 Ea 2.30R 3 Chapter 10: Nucleophilic Substitution Reaction II The Kinetics of Solvolysis- SN1 Reaction Time (sec) 9.97 8.03 7.69 Temperature °C 20 20 20 K- 0.104/time Kava Solvent composition 9. Does water participate directly in the reaction? 10. Predict the effect if the solvent were 60% water: 40% acetone. 80% H₂0 20% Acetone Use Data 2 to Answer Questions: 6. Use average to determine K. 7. What is the effect observed from increasing the water from 70% to 80% of the solvent mixture? What about the substrate? 8. What is the function of acetone? I
Expert Answer:
Answer rating: 100% (QA)
Question we observed that as temperature is higher than rate of react... View the full answer

Related Book For 

Human Resource Management
ISBN: 978-0078029127
12th edition
Authors: John Ivancevich, Robert Konopaske
Posted Date:
Students also viewed these management leadership questions
-
What personal qualities do you think are necessary for an employee to be successful at a company such as Zappos? How would you select for those qualities? Explain.
-
What social media platforms do you think are most critical for Lyon College to engage?
-
What services do you think are most important to todays increasingly diverse workforce? Why?
-
Products are 1. Lip gloss or lipstick with SPF 2. Custom facial sheets you can make online or in store pickup and have it send to your home based on your needs 3. A foundation sunscreen Price Provide...
-
Reexamine Exhibit 13.11. Explain the purpose of each performance measure. As a manager, which performance measure would you want to use. Which measure would you want used to evaluate you? Why? How...
-
A thin metallic wire of thermal conductivity k, diameter D, and length 2L is annealed by passing an electrical current through the wire to induce a uniform volumetric heat generation q. The ambient...
-
Given the background reading, list three verbal deception and three non-verbal deception cues that you believe that Aldrich Ames might exhibit and describe why
-
Evaluating a cost center including flexible budgeting concepts Koch Medical Equipment Company makes a blood pressure measuring kit. Albert Kaiser is the production manager. The production departments...
-
2. What is the computational complexity of the following function Mystery1? int Mystery1(int n) { int sum = 0; for (int i=0; i
-
1. a. How much did the materials cost per unit change between June and July? b. How much did the conversion cost per unit change between June and July? 2. Which is the optimal choice of material and...
-
2. Determine the amount of slack in cach of a project's activities (presented below), and identify those that are on the critical path Immediate Predecessor(s) None Estimated Activity Duration (in...
-
Gabriel sold CosmoVerse stock that he purchased 300 shares at $22.09 per share one year ago for a 20% loss. (Round your answer to 2 decimal places.) How much did Gabriel pay in total for their shares...
-
(Philippine government) Individual Output Describe how livelihood strategies interact with the institutional and government goals Explain how such arrangements facilitate or constrain access to...
-
3. (30 points) Use a diagram with schedules A and B as the ones in DFS (Dornbusch, Fischer, and Samuelson, 1977). Suppose the world consists of the U.S. and China, and that Chinese productivity in...
-
critically evaluate, using academic sources , If the UK had already joined the Euro, would the economic effects of Brexit have been bigger, smaller, or the same? Notes: (I would need to read up on...
-
What should you do before you begin defining an account set in Inventory Control?
-
A wave is described by the formula x(t) = (0.23 m) cos( 1,380t- 255x) Assuming the wave number, k, and angular frequency, w, are in SI units, calculate the speed of the wave in m/s with a single...
-
All of the following assets can be depreciated, except: (a) A bulldozer (b) A copper mine (c) A surgical robot (d) A conveyor belt
-
What defines career success to you?
-
Why are some people satisfied with what is identified as a midcareer plateau while others at the same point experience career burnout?
-
Describe the current trends in union membership and union organizing.
-
Baggai Enterprises has an ROA of 10 percent, retains 30 percent of earnings, and has an equity multiplier of 1.25. Mondale Enterprises also has an ROA of 10 percent, but it retains two-thirds of...
-
International Business Machines (NYSE: IBM), which currently pays a dividend of \($3.40\) per share, has been the subject of two other examples in this reading. In one example, an analyst estimated...
-
An analyst is preparing a forecast of dividends for Hoshino Distributors for the next five years. He uses a spreadsheet model with the following assumptions: Sales are $100 million in Year 1. They...

Study smarter with the SolutionInn App


