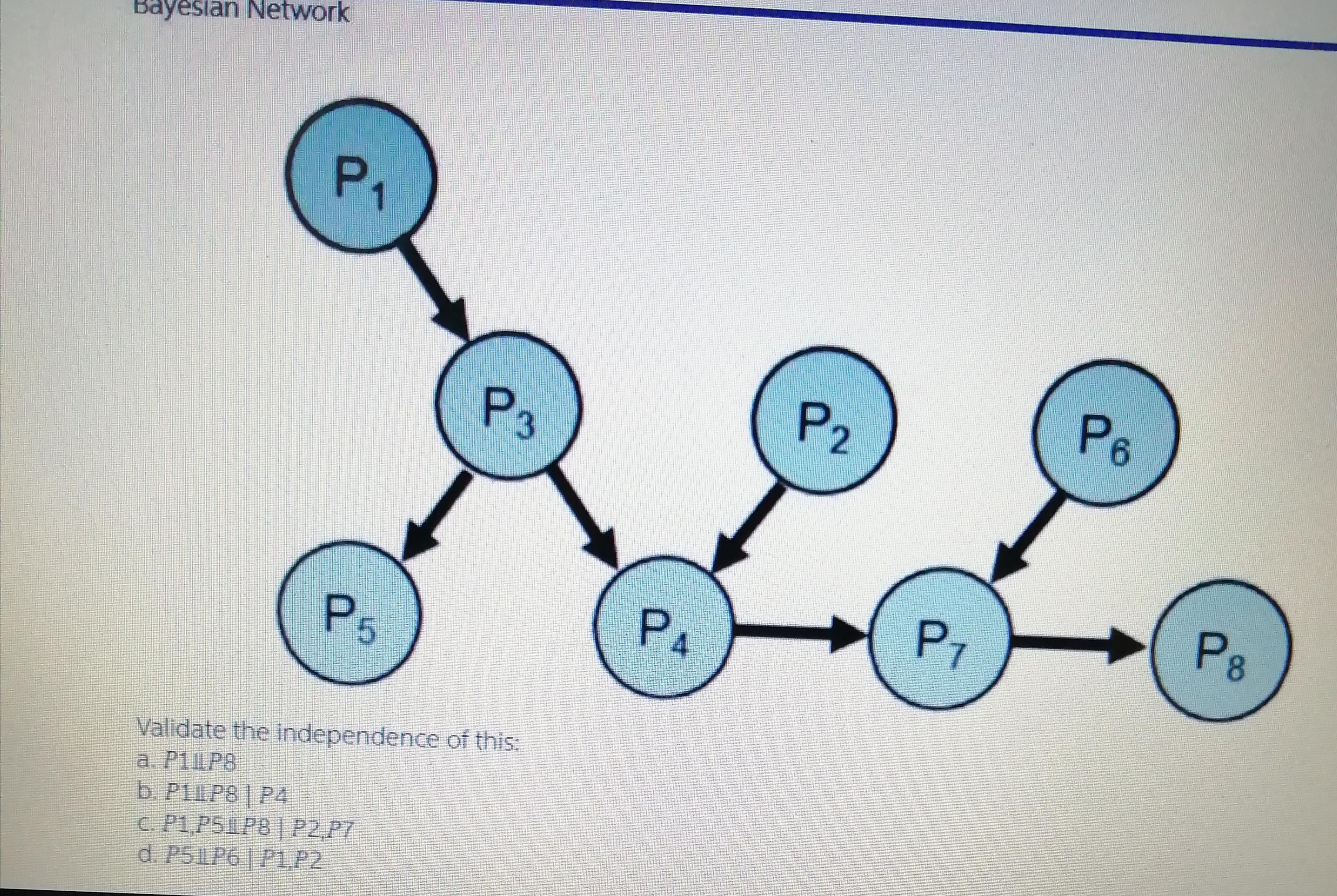
Bayesian Network P1 PB P3 P2 P8 P7 PA P5 Validate the independence of this: a....
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Bayesian Network P1 PB P3 P2 P8 P7 PA P5 Validate the independence of this: a. P1LP8 b. P1LP8 P4 C. P1,P5LP8| P2,P7 d. P51LP6|P1P2 Bayesian Network P1 PB P3 P2 P8 P7 PA P5 Validate the independence of this: a. P1LP8 b. P1LP8 P4 C. P1,P5LP8| P2,P7 d. P51LP6|P1P2
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these algorithms questions
-
A $30,000 new car loan is taken out with the terms 6% APR for 48 months. How much are monthly payments on this loan? O A. $915.92 O B. $775.01 O C. $704.55 OD. $845.46
-
A and B react to produce C according to the following chemical equation: A+ B C Amounts of A and B are added to an equilibrium reaction mixture of A, B, and C such that when equilibrium is again...
-
The figure shows two circles C and D of radius 1 that touch at P.T is a common tangent line; C1 is the circle that touches C, D, and T; C2 is the circle that touches C, D, and C1; C3 is the circle...
-
Is informing the consumer about any toxic substances used in the manufacture of a product sufficient to produce an efficient level of toxic substance use for that product? Why or why not?
-
Prove that (- a)v = - (ax) in Theorem 3 by first computing (- a)v + av. Then do it using (4) of Theorem 3 and axiom S4.
-
Use the intermediate value theorem to show that each polynomial function has a real zero between the numbers given. (x) = x 4 - 2x 3 - 2x 2 - 18x + 5; 3.7 and 3.8
-
Reconsider Parts (a) through (f) of Problem 2. For each "true" statement, develop a mathematical proof based on the time value of money factor equations from Table 2.6 in Chapter 2. Data from problem...
-
Rattigan Companys income statement contained the condensed information below. Rattigans balance sheet contained the comparative data at December 31, shown below. Accounts payable pertain to operating...
-
The unemployment rate among workers under 25 in a populous state went from 8.9% to 6.7% in one year. Assume an average of 1 comma 340 comma 500 workers and estimate the decrease in the number...
-
DeBauge Realtors, Inc., is a realty firm owned by Jeff and Kristi DeBauge. The DeBauge family owns 100% of the corporations stock. The following summarized data (in thousands) are taken from the...
-
Two members of a gym are competing to see who lost the most weight in the first 5 months of the year. Make a program to create two separate arrays for the two members. The program should ask for the...
-
John is setting up his timetable for second semester. There are 7 courses that John is interested in taking, but there is room for only 4 courses in his timetable. How many different groups of 4...
-
Gamma Ltd has a reconciliation problem. The bank reconciliation officer is unable to reconcile the cash at bank value recorded in the general ledger account with the cash at bank balance disclosed on...
-
contract proposal for a newly drafted first-round draft pick The agent will seek to construct a contract that will benefit his or her client the best. This contract proposal that will meet the needs...
-
Send an email to "All Staff" outlining the details of the upcoming Staff Appreciation Night. Here are a few details to consider..... - Message will be sent the middle of next month. - Acknowledge...
-
Can proprietary companies engage in crowd-sourced funding? If so, what compliance obligations are imposed by the Corporations Act?
-
Shelby says that her cat's weight has a z-score (or standard score) of -2. The average weight of a cat is 8.9 pounds, with a standard deviation of 1.5 pounds. T 15. How much does his cat weigh? 6.9...
-
Shreemaya Hotel in !adore was facing a problem of low demand for its rooms due to off season. The Managing Director (MD) of the hotel, Mrs. Sakina was very worried. She called upon the Marketing...
-
Consider the following setup, which shows identical containers connected by a tube with a valve that is presently closed. The container on the left has 1.0 mol of H2 gas; the container on the right...
-
A hypothetical element A has the following properties: First ionization energy: 850 kJ/mol Second ionization energy: 1700 kJ/mol Third ionization energy: 13,999 kJ/mol a. If you were to react element...
-
Calculate the energy released when 5.00 kg of uranium-235 undergoes the following fission process. The masses of and nuclei are 135.8401 amu and 95.8629 amu, respectively. Other masses are given in...
-
Go to http://management.about.com/lr/project_time_ management/174690/1/ and click on Project Management on the left side of the screen. When you consider several of the articles on time management in...
-
Consider the following information that you have compiled regarding the steps needed to complete a project. You have identified all relevant steps and have made some determinations regarding...
-
Key in a search with the prompt projects in in which you select a country of interest (e.g., projects in Finland). Many of the projects generated by such a search are government-sponsored...

Study smarter with the SolutionInn App


