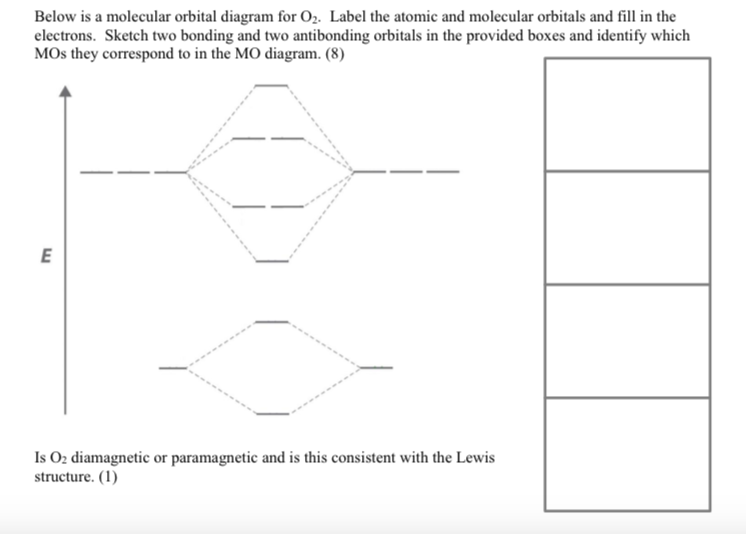
Below is a molecular orbital diagram for O. Label the atomic and molecular orbitals and fill...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Below is a molecular orbital diagram for O₂. Label the atomic and molecular orbitals and fill in the electrons. Sketch two bonding and two antibonding orbitals in the provided boxes and identify which MOs they correspond to in the MO diagram. (8) E Is O₂ diamagnetic or paramagnetic and is this consistent with the Lewis structure. (1) Below is a molecular orbital diagram for O₂. Label the atomic and molecular orbitals and fill in the electrons. Sketch two bonding and two antibonding orbitals in the provided boxes and identify which MOs they correspond to in the MO diagram. (8) E Is O₂ diamagnetic or paramagnetic and is this consistent with the Lewis structure. (1)
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
i. Construct Molecular Orbital diagram for C 2 -2 ii. Write the electron configuration for C 2 -2 iii. Identify the magnetism for C 2 -2 iv. State the HOMO and LUMO for C 2 -2 v. Calculate the bond...
-
Draw the complete molecular orbital diagram for Dichlorobis(ethylenediamine)cobalt(III), considering only the -bonding.
-
Sketch the molecular orbital and label its type (Ï or p; bonding or antibonding) that would be formed when the following atomic orbitals overlap. Explain your labels. a. b. d.
-
What is opportunity cost? explain a time in your life when you experienced opportunity cost. Why did you make the decision? What was another alternative that you could have made? Discuss how scarcity...
-
How do internal auditors deter or detect financial fraud?
-
All accounts have normal balances. What is the normal balance of each of these accounts? a. Cash b. Sales c. Notes payable d. Inventory e. Retained earnings f. Salary expense g. Equipment h. Unearned
-
7. On June 30, John N. Willis signed a demand promissory note for $1,620 to the Camelot Country Club in Carrollton, Texas. The note stated that it was being given in payment for a membership in the...
-
The standards for one case of liquid weed killer are: Direct materials . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3 lbs. @ $ 6.00/lb. Direct labor . . . . . . . . . . . . . ....
-
Given are five observations for two variables, x and y. 1 xi 8 14 16 19 Yi 58 56 45 21 10 The estimated regression equation for these data is = 68.64 - 2.64x. a. Compute SSE, SST, and SSR. SSE (to 2...
-
An automated car wash called Jet Express outside a large Texas city on the beltway is deciding whether to build and install one dedicated vehicle wash line or have two parallel wash lines. There are...
-
The prices for the White Swan Corporation for the first quarter of the last year are given below. Find the holding period return (percentage return) for February. End of the month Stock price January...
-
Analyze the information provided to reconcile the shareholders' beginning and ending bases in their stock to consequences of JJC's corporate distributions. (Decreases should be entered with a minus...
-
Expeditionary Advanced Base Operations employs mobile, low-signature, persistent naval expeditionary forces within a contested maritime area for what purposes? Question 3Answer a. To conduct...
-
The stockholders equity section of the balance sheet for Mann Equipment Co . at December 3 1 , Year 2 , is as follows. Stockholders Equity Paid - in capital Preferred stock, ? par value, 5 %...
-
Describe how the proposed change in procedure and practice impacted relevant stakeholders. Describe the behaviors of the team during implementation of an innovation team in an early childcare...
-
Hospitality in the Big City1. How does this case illustrate the importance of 2. How important is it to defuse unpleasantness even 3.Describe any similar experiences you may havehad where people...
-
Accounting 1C Long Term Project- Master Budget You need to prepare a Master Budget for the The company has an exclusive right to sell PowerPulses and sales have been brisk. The Master Budget will be...
-
Write the expression in radical notation. Then evaluate the expression when the result is an integer. 23 -1/2
-
Account for the following observations: (a) H3PO3 is a diprotic acid. (b) Nitric acid is a strong acid, whereas phosphoric acid is weak. (c) Phosphate rock is ineffective as a phosphate fertilizer....
-
The osmotic pressure of a saturated solution of strontium sulfate at 25c is 21 torr. What is the solubility product of this salt at25?
-
Ortho-dichlorobenzene, C6H4Cl2, is obtained when two of the adjacent hydrogen atoms in benzene are replaced with Cl atoms. A skeleton of the molecule is shown here. (a) Complete a Lewis structure for...
-
What roles are played by the CICA, the CGAAC, and the SMAC for their members?
-
The following questions deal with audits by public accounting firms. Select the best response for each question. a. Which of the following best describes why an independent auditor is asked to...
-
Explain what is meant by determining the degree of correspondence between information and established criteria. What are the information and established criteria for the audit of Jones Ltd.'s tax...

Study smarter with the SolutionInn App



