Below is some background information about vapor liquid equilibrium of mixtures - you can read if...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
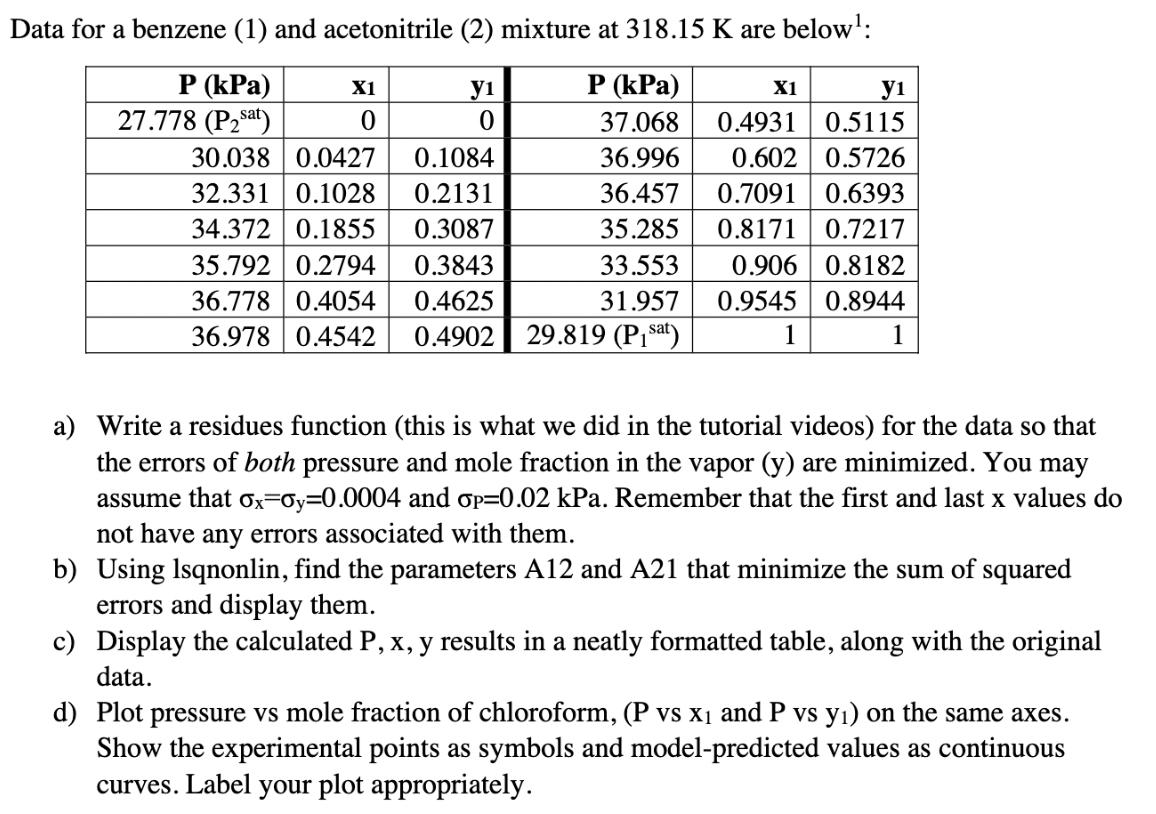
Below is some background information about vapor liquid equilibrium of mixtures - you can read if you'd like, but it's not required to do the problem. This is a topic that will show up in 311 and in separation processes. I've put a ** next to the key equations/information for this problem. Vapor-liquid equilibrium of ideal mixtures can be modeled with Raoult's Law: Psat (T) Yi = P where y; is the mole fraction of component i in the vapor, X is the mole fraction of component i in the liquid, Pisat is the vapor pressure of the pure component at the temperature T, and P is the pressure. However, mixtures are not always ideal. To correct for "realness," we can use a modified Raoult's law: Psat (T)Yi ** Yi = -xi P where Yi is the activity coefficient of component i in the mixture. Using material balances, we can rewrite the second equation as follows; ** P = Psat (T)Y1x1 + Pat (T)Y2x2 This allows us to calculate the equilibrium pressure (or bubble point pressure), which then allows us to calculate yi. The activity coefficients can be modeled by a number of different equations: one of which is the 2-parameter van Laar model: Aij ** In Yi = Aijxi] + Ajixj **i and j are the component numbers - if i=1, then j=2 (and vice versa). This equation needs to be used twice - once for y and once for y2. Aij's can be found from experimental data and can be assumed constant within a range of T and P. They are specific to the components in the mixture. Data for a benzene (1) and acetonitrile (2) mixture at 318.15 K are below: P (kPa) X1 y1 P (kPa) X1 y1 27.778 (Psat) 0 0 37.068 0.4931 0.5115 30.038 0.0427 0.1084 36.996 0.602 0.5726 32.331 0.1028 0.2131 34.372 0.1855 0.3087 35.792 0.2794 0.3843 36.457 0.7091 0.6393 35.285 0.8171 0.7217 36.778 0.4054 0.4625 36.978 0.4542 0.4902 33.553 31.957 29.819 (Pisa) 0.906 0.8182 0.9545 0.8944 1 1 1 a) Write a residues function (this is what we did in the tutorial videos) for the data so that the errors of both pressure and mole fraction in the vapor (y) are minimized. You may assume that ox-oy=0.0004 and p=0.02 kPa. Remember that the first and last x values do not have any errors associated with them. b) Using Isqnonlin, find the parameters A12 and A21 that minimize the sum of squared errors and display them. c) Display the calculated P, x, y results in a neatly formatted table, along with the original data. d) Plot pressure vs mole fraction of chloroform, (P vs X1 and P vs y) on the same axes. Show the experimental points as symbols and model-predicted values as continuous curves. Label your plot appropriately. Below is some background information about vapor liquid equilibrium of mixtures - you can read if you'd like, but it's not required to do the problem. This is a topic that will show up in 311 and in separation processes. I've put a ** next to the key equations/information for this problem. Vapor-liquid equilibrium of ideal mixtures can be modeled with Raoult's Law: Psat (T) Yi = P where y; is the mole fraction of component i in the vapor, X is the mole fraction of component i in the liquid, Pisat is the vapor pressure of the pure component at the temperature T, and P is the pressure. However, mixtures are not always ideal. To correct for "realness," we can use a modified Raoult's law: Psat (T)Yi ** Yi = -xi P where Yi is the activity coefficient of component i in the mixture. Using material balances, we can rewrite the second equation as follows; ** P = Psat (T)Y1x1 + Pat (T)Y2x2 This allows us to calculate the equilibrium pressure (or bubble point pressure), which then allows us to calculate yi. The activity coefficients can be modeled by a number of different equations: one of which is the 2-parameter van Laar model: Aij ** In Yi = Aijxi] + Ajixj **i and j are the component numbers - if i=1, then j=2 (and vice versa). This equation needs to be used twice - once for y and once for y2. Aij's can be found from experimental data and can be assumed constant within a range of T and P. They are specific to the components in the mixture. Data for a benzene (1) and acetonitrile (2) mixture at 318.15 K are below: P (kPa) X1 y1 P (kPa) X1 y1 27.778 (Psat) 0 0 37.068 0.4931 0.5115 30.038 0.0427 0.1084 36.996 0.602 0.5726 32.331 0.1028 0.2131 34.372 0.1855 0.3087 35.792 0.2794 0.3843 36.457 0.7091 0.6393 35.285 0.8171 0.7217 36.778 0.4054 0.4625 36.978 0.4542 0.4902 33.553 31.957 29.819 (Pisa) 0.906 0.8182 0.9545 0.8944 1 1 1 a) Write a residues function (this is what we did in the tutorial videos) for the data so that the errors of both pressure and mole fraction in the vapor (y) are minimized. You may assume that ox-oy=0.0004 and p=0.02 kPa. Remember that the first and last x values do not have any errors associated with them. b) Using Isqnonlin, find the parameters A12 and A21 that minimize the sum of squared errors and display them. c) Display the calculated P, x, y results in a neatly formatted table, along with the original data. d) Plot pressure vs mole fraction of chloroform, (P vs X1 and P vs y) on the same axes. Show the experimental points as symbols and model-predicted values as continuous curves. Label your plot appropriately.
Expert Answer:

Related Book For 

Fundamentals of Cost Accounting
ISBN: 978-1259565403
5th edition
Authors: William Lanen, Shannon Anderson, Michael Maher
Posted Date:
Students also viewed these chemical engineering questions
-
THIRD AVENUE SOFTWARE HEALTH-CARE APP PROJECT This case is new for the ninth edition of Information Technology Project Management . The case provides an opportunity to apply agile and Scrum...
-
List three specific parts of the Case Guide, Objectives and Strategy Section (See below) that you had the most difficulty understanding. Describe your current understanding of these parts. Provide...
-
Suppose in a given area there are three power plants, each of which emits SO 2 with different intensities. The abatement cost functions for each firm j are: (a) Set up the conditions for the socially...
-
The following information is available for Lock-Safe Company, which produces special-order security products and uses a job order cost accounting system. Compute the following amounts for the month...
-
Fabrication Holdings Ltd. (FH) has been a client of KFP Partners for many years. You are an audit senior and have been assigned to the FH audit for the first time for the financial year end, December...
-
Write a code to test a Gaussian pseudorandom number generator. If you do not have a canned generator available, write a generator based on the Box-Muller algorithm in Appendix I. Apply the following...
-
For the following test marketing project at week 6: a. Ignore the far right % Complete column and using the 5050 percent completion rule for PV and EV, calculate the cost, schedule, and time...
-
Consider that your cache is a direct mapped memory. It can hold up to 256 byte of data in 32-bit MIPS architecture. (a) Assuming that your block numbers, validity bits, and space allocated for tags...
-
Imogen runs the Manor Park Hotel in Powys, Wales. She purchased the 50-bedroom hotel 10 years ago. She is wondering if the business is trading as well as it could be and would like a report stating...
-
Assume that market penetration is 0.30 and that your company's penetration share is 0.87. Calculate brand penetration. Round your answer to 2 decimal places.
-
A certain apple bruises if a net force greater than 9 . 5 N is exerted on it . Would a 0 . 1 3 k g apple be likely to bruise if it falls 1 . 8 m and stops after sinking 0 . 0 5 m into the grass?...
-
A projectile launcher fires a marble of mass 2 5 grams perfectly vertical. The launcher uses a spring with a constant, k , of 6 0 . 0 Newtons / meter . If the spring is depressed 1 5 centimeters and...
-
A car is initially travelling at 3 0 m / s , which is above the speed limit . The driver sees a speed limit trap ahead and applies the brakes for 5 seconds, causing the car to slow down by 2 m / s...
-
Two wires carrying equal and opposite currents are twisted together in the construction of a circuit. Why does this technique reduce stray magnetic fields? Please explain for dummies.
-
A magnet of mass 5 . 0 1 kg is suspended from the ceiling by a cord as shown. A large magnet is somewhere off to the right, pulling on the small hanging magnet with a constant force of F = 8 0 . 4 N...
-
List the factors responsible for determining the forward and backward feed arrangement in evaporator system.
-
Using a graphing utility, graph y = cot -1 x.
-
What is a critical success factor?
-
Refer to Exercise. In Exercise, Larcker Manufacturings cost accountant has provided you with the following information for January operations: Direct materials . . . . . . . . . . . . . . . . . . . ....
-
As a marketing manager for an airline, would you sell a seat to a passenger who walked up to the gate at the last minute at the variable (marginal) cost? Why or why not? Do the costs from the...
-
Scientists propose that the first macromolecules in protobionts were a. DNA molecules. b. RNA molecules. c. proteins. d. all of the above.
-
Which of the following function(s) is/are carried out by piRITS or piRISC? a. Inhibits transcription of TEs b. Causes the degradation of TE RNA c. Causes chromosome breakage d. Both a and b are...
-
In the CRISPR-Cas system, what does tracrRNA bind to? a. crRNA and Cas1 protein b. crRNA and Cas2 protein c. crRNA and Cas9 protein d. Cas1 and Cas2 proteins

Study smarter with the SolutionInn App


