Below is the reaction you will be performing today: To perform the synthesis you will be using
Question:
Below is the reaction you will be performing today:
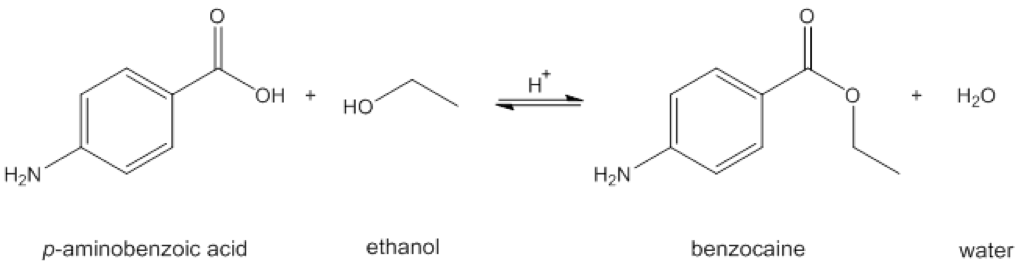
To perform the synthesis you will be using the following reagents:
| p-aminobenzoic acid (PABA) | ethanol |
| MR = 137.14 g/mol | MR = 47.07 g/mol |
| 0.80 g | 10 mL (density: 0.789 g/mL) |
Calculate (to 2 significant figures):
No. of moles of PABA: Answer = _____ mol
Mass of ethanol: Answer = _____ grams
No. of moles of ethanol: Answer = _____ mol
Determine the molar ratio of PABA : ethanol being used today (round to the nearest integer):
| PABA | : | ethanol |
| Ratio = nPABA / nPABA |
| Ratio = nethanol / nPABA |
| Ratio = 1 | : | Answer = ______ |
Out of PABA and ethanol, which reagent will be used in excess?
a) PABA
b) Ethanol
c) Neither is in excess – they will be used in their stoichiometric ratios

Essentials Of Business Statistics Communicating With Numbers
ISBN: 9780078020544
1st Edition
Authors: Sanjiv Jaggia, Alison Kelly




