The vapor pressure of a pure liquid was measured at several temperatures and is given in...
Fantastic news! We've Found the answer you've been seeking!
Question:
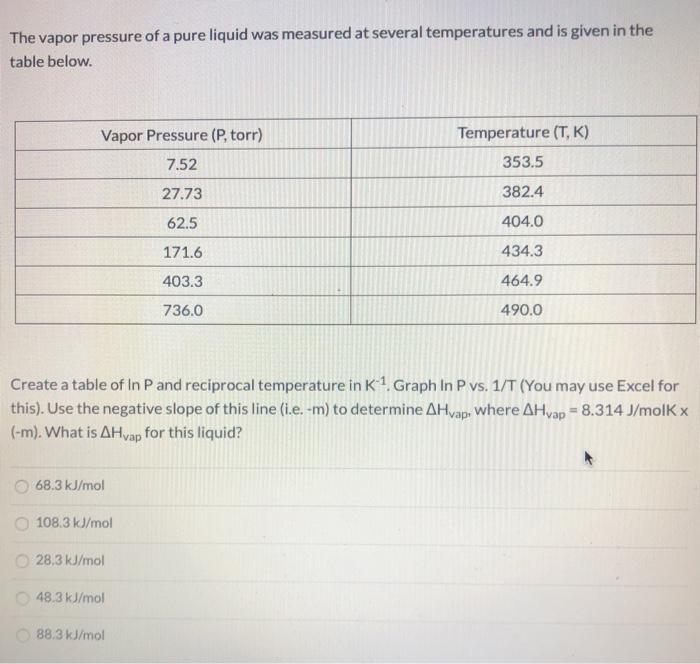
Transcribed Image Text:
The vapor pressure of a pure liquid was measured at several temperatures and is given in the table below. Vapor Pressure (P, torr) Temperature (T, K) 7.52 353.5 27.73 382.4 62.5 404.0 171.6 434.3 403.3 464.9 736.0 490.0 Create a table of In P and reciprocal temperature in K1. Graph In P vs. 1/T (You may use Excel for this). Use the negative slope of this line (i.e. -m) to determine AHvap, Where AHvap = 8.314 J/molK x (-m). What is AHvap for this liquid? 68.3 kJ/mol 108.3 kJ/mol 28,3 kJ/mol O 48.3 kJ/mol O 88.3 kJ/mol The vapor pressure of a pure liquid was measured at several temperatures and is given in the table below. Vapor Pressure (P, torr) Temperature (T, K) 7.52 353.5 27.73 382.4 62.5 404.0 171.6 434.3 403.3 464.9 736.0 490.0 Create a table of In P and reciprocal temperature in K1. Graph In P vs. 1/T (You may use Excel for this). Use the negative slope of this line (i.e. -m) to determine AHvap, Where AHvap = 8.314 J/molK x (-m). What is AHvap for this liquid? 68.3 kJ/mol 108.3 kJ/mol 28,3 kJ/mol O 48.3 kJ/mol O 88.3 kJ/mol
Expert Answer:
Answer rating: 100% (QA)
Select a middle data and Apply Clausius Clapeyron equation ... View the full answer

Related Book For 

Practicing Statistics Guided Investigations for the Second Course
ISBN: 978-0321586018
1st edition
Authors: Shonda Kuiper, Jeff Sklar
Posted Date:
Students also viewed these accounting questions
-
In Section 11.5 we defined the vapor pressure of a liquid in terms of an equilibrium. (a) Write the equation representing the equilibrium between liquid water and water vapor and the corresponding...
-
The data given in the table below are the midterm scores in a course for a sample of 10 students and the scores of student evaluations of the instructor. (In the instructor evaluation scores, 1 is...
-
The following table gives the vapor pressure of hexafluorobenzene (C6F6) as a function of temperature: (a) By plotting these data in a suitable fashion, determine whether the Clausius-Clapeyron...
-
X-Tech Inc. produces specialized bolts for the aerospace industry. The operating cost of producing a single bolt is $2. The company currently sells the bolts for $6/unit. Each time the company...
-
Cost for inventory purposes should be determined by the inventory cost flow method most clearly reflecting periodic income. Required: 1. Describe the fundamental cost flow assumptions for the average...
-
Jon Hampton, president and CEO of Plastics, Inc., is living a life-style beyond his means. He has several short positions to cover with his stock brokers. His creditors are becoming especially...
-
How is the contribution margin per unit used?
-
A company president remarked, The operations of our company are such that we can take advantage of only a minor amount of financial leverage. Explain the likely reasoning the company president had in...
-
A tank contains 2000 liters (L) of a solution consisting of 165 kg of salt dissolved in water. Pure water is pumped into the tank at the rate of 10 L/s, and the mixture-kept uniform by stirring-is...
-
Suppose the daily demand curve for flounder at Cape May is given by QD = 1,600 - 600P, where QD is demand in pounds per day and P is price per pound. a. If fishing boats land 1,000 pounds one day,...
-
Using Java Programming 1. Create a program to count and store the word frequency in a document. a. Ignore punctuation and case. Hyphenated words count separately. b. Use a java.util. HashMap to store...
-
A hedge fund manager believes that the US central bank has raised interest rates to such a degree that a severe recession is on the horizon. The manager therefore anticipates that the central bank...
-
Semi-Annually Compounded Zero Yields Time(Year) 0.5 1 1.5 2 2.5 3 Zero Yield 1.2% 1.6% 2.0% 2.4% 2.8% 3.2%A mortgage loan is a "bond" paying the same dollar amount of cash flows. Suppose a mortgage...
-
At the start of the year, a client s portfolio is worth $250,000. During the year, the client deposits an additional $20,000, while at the end of the year the portfolio is worth $285,000. What is the...
-
What might the reaction of workers be if you implemented shorter work schedules? How would you change the culture in your organization to be more ethical and humane?
-
This week's thread deals with the legal concept of caveatemptor , or, as it is commonly known, "let the buyer beware." Caveatemptorplaces the burden on the buyer to find out any problems that may...
-
Find three small business websites. Analyze each website in terms of its trustworthiness. Discuss why you would or would not trust each site. Be specific. one page)
-
What does non-recourse financing mean?
-
Use software to calculate the eigenvectors of R.
-
In 2009, three Grinnell students (Allie Greenberg, Hannah Lytle, and Phillip Brogdon) conducted an analysis to estimate the probability of winning the Academy Award for Best Picture. The Academy...
-
State the null and alternative hypotheses for this study. Conduct an ANOVA to analyze the Flower data. Check the normality and equal variance assumptions. State your conclusions based on the p-...
-
Nikron Corporation issued 20,000 shares of \(\$ 0.50\) par value common stock during the year for \(\$ 20\) each. Nikron also repurchased treasury stock for \(\$ 15,000\). Net income for the year was...
-
What is debt finance, and how does it differ from equity finance?
-
What is a preference share and why might a company favour this form of finance?

Study smarter with the SolutionInn App


