B(x) represents Brownian motion Problem 5: What is the conditional distribution of (B(5), B(9), B(12)) given B(10)
Fantastic news! We've Found the answer you've been seeking!
Question:
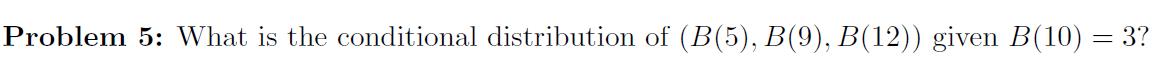
B(x) represents Brownian motion
Transcribed Image Text:
Problem 5: What is the conditional distribution of (B(5), B(9), B(12)) given B(10) = 3? Problem 5: What is the conditional distribution of (B(5), B(9), B(12)) given B(10) = 3?
Expert Answer:
Answer rating: 100% (QA)
PB5 B9 B12B103 PB5B103PB9B103PB12B103 PB5B103 PB5 B10 ... View the full answer

Posted Date:
Students also viewed these mathematics questions
-
Suppose that S1 and S2 follow geometric Brownian motion and pay continuous proportional dividends at the rates 1 and 2. Use the martingale argument to show that the value of a claim paying S1(T) if...
-
Let X(t) be a Brownian motion process with variance Var[X(t)] = t. For a constant c > 0, determine whether Y(t) = X(ct) is a Brownian motion process.
-
The phenomenon of Brownian motion is the random motion of microscopically small particles as they are buffeted by the still smaller molecules of a fluid in which they are suspended. For a particle of...
-
Brothers Willie and Billie each own a gas station for the same national chain. As brothers, they are very competitive. They held a contest to see who could do a better job forecasting sales for their...
-
A quality characteristic has a design specification (in cm.) of 0.200 0.04. If the actual process value of the quality characteristic is at the boundary of the tolerance on either side, the product...
-
a. What is financial leverage? b. What impact does positive leverage have on the rate earned on stockholders equity compared to the rate earned on total assets?
-
Jupiter, with an inertia 317.83 times that of Earth, is at an average distance of \(7.784 \times 10^{11} \mathrm{~m}\) from the Sun. At what distance from the centre of the Sun is the center of mass...
-
On October 1, 2014, Caldonia Company paid East Alabama Rentals $4,800 for a 12-month lease on warehouse space. Required a. Record the deferral and the related December 31, 2014, adjustment for...
-
7. The full symbol of an ion of an element is 39X+? Which one of the following is the number of electrons in the ion? A. 18 B. 19 C. 20 D. 39 8. The atomic numbers of elements W, X, Y and Z are 9,...
-
On January 1, 2024, LLB Industries borrowed $200,000 from Trust Bank by issuing a two-year, 10% note, with interest payable quarterly. LLB entered into a two-year interest rate swap agreement on...
-
1. Oliver is a graduate student who receives utility from the power cooling his home down to 75 (x1) and the power he uses for his own entertainment at night (x2). He has utility from consumption of...
-
Two equal charges exert equal forces on each other. What if one charge has twice the magnitude of another? How do the forces they exert on each other compare?
-
Louie E. Brown worked for the Phelps Dodge Corporation under an oral contract for approximately twenty-three years. In 2015, he was suspended from work for unauthorized possession of company...
-
Presti claims that he reached an oral agreement with Wilson by telephone in October to buy a horse for \($60,000.\) Presti asserts that he sent Wilson a bill of sale and a postdated check, which...
-
A fifteen-year-old minor was employed by Midway Toyota, Inc. On August 18, 2014, the minor, while engaged in lifting heavy objects, injured his lower back. In October 2014 he underwent surgery to...
-
A consumer has initial real wealth of 20 , current real income of 90 , and future real income of 110 . The real interest rate is \(10 \%\) per period. a. Find the consumer's PVLR. b. Write the...
-
the company decides to dispose of the machine on June 3 0 , 2 0 2 3 . 3 0 , 2 0 2 3 . The machine was purchased on January 1 , 2 0 1 8 1 , 2 0 1 8 for $ 8 2 , 0 0 0 8 2 , 0 0 0 and depreciated on a...
-
Why did management adopt the new plan even though it provides a smaller expected number of exposures than the original plan recommended by the original linear programming model?
-
How many oxygen atoms are bonded to the carbon of the carbonyl of an ester functional group? (a) none (b) one (c) two (d) three
-
Which would you expect to be more viscousa polymer made of long molecular strands or a polymer made of short molecular stands? Why? (a) long strands, because they tend to tangle among themselves (b)...
-
Explain why caprylic acid, CH 3 (CH 2 ) 6 COOH, dissolves in a 5, aqueous solution of sodium hydroxide but caprylaldehyde, CH 3 (CH 2 ) 6 CHO, does not dissolve. (a) With two oxygens, the caprylic...

Study smarter with the SolutionInn App

