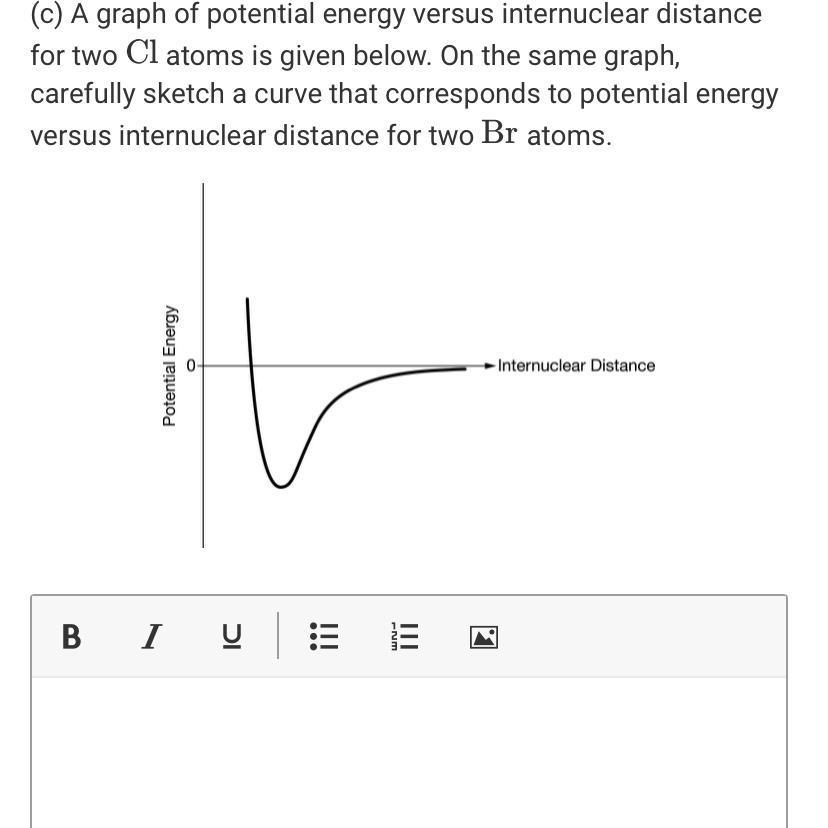
(c) A graph of potential energy versus internuclear distance for two Cl atoms is given below....
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
(c) A graph of potential energy versus internuclear distance for two Cl atoms is given below. On the same graph, carefully sketch a curve that corresponds to potential energy versus internuclear distance for two Br atoms. Internuclear Distance Potential Energy B I IC ... ||| -Nm ||| (c) A graph of potential energy versus internuclear distance for two Cl atoms is given below. On the same graph, carefully sketch a curve that corresponds to potential energy versus internuclear distance for two Br atoms. Internuclear Distance Potential Energy B I IC ... ||| -Nm |||
Expert Answer:
Answer rating: 100% (QA)
QA graph of Potential energy Answergraph is sketched fo... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Make a graph of potential energy versus angle of bond rotation for propane, and assign values to the energy maxima.
-
Consider a perfectly competitive market for wheat in Dallas. There are 60 firms in the industry, each of which has the cost curves shown on the following graph: Hint Use the black point (plus symbol)...
-
The potential energy of two atoms in a diatomic molecule is approximated by V(r) = a/r12 - b/r2, where r is the spacing between atoms and a and b are positive constants. (a) Find the force F( r) on...
-
Roasters Limited is a coffee-blending firm. It produces a special blend of coffee known as "Utopia Blend" by mixing two grades of coffee "AB" and "QP" as follows: Material AB QP Standard mix ratio AB...
-
One of your friends is a financial analyst for a major stock brokerage firm. Recently she indicated to you that she had read an article in a weekly business magazine that alluded to the political...
-
Suppose a firm makes purchases of $3.65 million per year under terms of 2/10, net 30, and takes discounts. a. What is the average amount of accounts payable net of discounts? (Assume the $3.65...
-
The Basel Accords have had a great impact on the banking industry. Describe these and explain their significance to the ordinary person.
-
Go to Case 3.1, Cruz v. Fagor America, Inc. Read the excerpt and answer the following questions. (a) Issue: On what preliminary step to litigation does the issue in this case focus? (b) Rule of Law:...
-
May Corp. has estimated that total depreciation expense for the year ending December 31, 2022, will amount to $400,000 and that 2022 year-end bonuses to employees will total $800,000. In May's...
-
You want to park your bicycle in a bicycle parking area where bike racks are aligned in a row. There are already N bikes parked there (each bike is attached to exactly one rack, but a rack can have...
-
A steel rod with a diameter of 24 mm is provided with a screw thread on each end and also nuts having a pitch of 1,0 mm. A pipe of the same material which is 1,2 m long with an internal diameter of...
-
1. Keanu is 47 years old, and the amplitude of accommodation in his left eye is about one standard deviation above average for his age. From now on, we concentrate exclusively on his left eye. When...
-
A pilot performs a vertical maneuver around a circle with a Radius R. When the airplane is at the lowest point of the circle that pilot's apparent weight is 6 mg. What is the acceleration of the...
-
Solve for x. 765 = 5x3
-
Required rate of return is 12%, what level of annual cash savings? Grouper's Lawn Service needs to purchase a new lawnmower costing $8,696 to replace an old lawnmower that cannot be repaired. The new...
-
Using circular motion to approximate elliptical orbits, how can you describe the motion of planets, moons (natural satellites) and satellites (artificial satellites) using mathematical...
-
Employee's Rates 7.65% on first $118,500 of income .1.45% on income above $118,500 2016 FICA Tax Rates Matching Rates Paid by the Employer 7.65% on first $118,500 paid in wages 1.45% on wages above...
-
d. The characteristic equation of a control system is given by s+2s+8s+12s+20s+16+16=0. Determine the number of the roots of the equation which lie on the imaginary axis of s-plane
-
Propose a mechanism to account for the formation of 3, 5-dimethylpyrazole from hydrazine and 2, 4-pcntancdionc. Look carefully to see what has happened to each carbonyl carbon in going from starting...
-
Aldehydes and ketones undergo acid-catalyzed reaction with alcohols to yield hemiacetals, compounds that have one alcohol-like oxygen and one ether-like oxygen bonded to the same carbon. Further...
-
Name the following cycloalkanes:
-
Sarah Saxon and Jane Rolf are partners in a business called J & S Service. The partnership's work sheet for the year ended December 31 of the current year is provided in the Working Papers....
-
Donald Winn and Judy Reed agreed to liquidate their partnership on June 30 of the current year. On that date, after financial statements were prepared and closing entries were posted, the general...
-
Theresa Doran and Roy Eden are partners in a business called D & E Sales. The partnership's work sheet for the year ended December 31 of the current year is provided in the Working Papers. ...

Study smarter with the SolutionInn App


