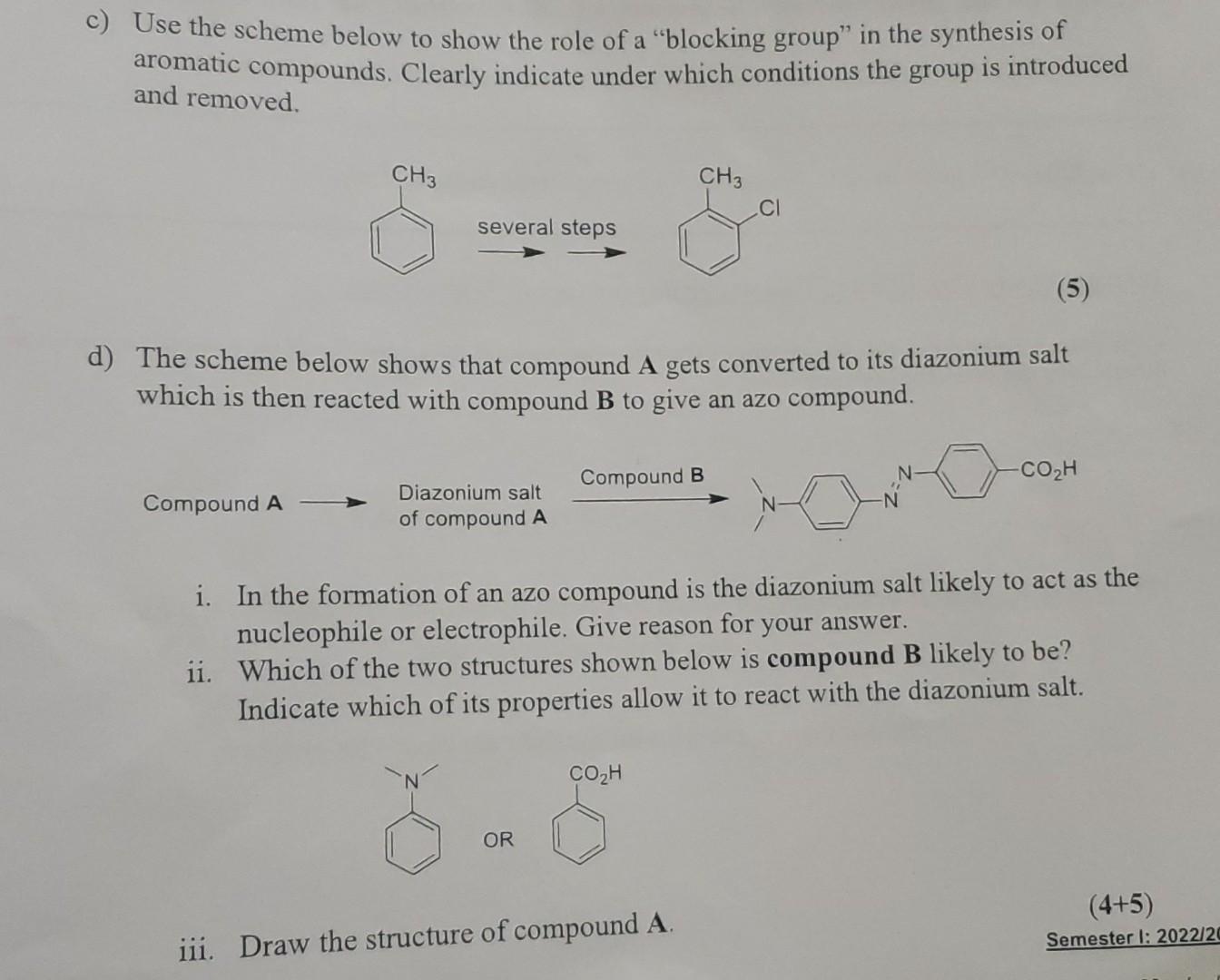
c) Use the scheme below to show the role of a blocking group in the synthesis...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
c) Use the scheme below to show the role of a "blocking group" in the synthesis of aromatic compounds. Clearly indicate under which conditions the group is introduced and removed. CH3 Compound A several steps (5) d) The scheme below shows that compound A gets converted to its diazonium salt which is then reacted with compound to give an azo compound. Diazonium salt of compound A OR CH3 Compound B CO₂H CI i. In the formation of an azo compound is the diazonium salt likely to act as the nucleophile or electrophile. Give reason for your answer. ii. Which of the two structures shown below is compound B likely to be? Indicate which of its properties allow it to react with the diazonium salt. iii. Draw the structure of compound A. COzH (4+5) Semester 1: 2022/20 c) Use the scheme below to show the role of a "blocking group" in the synthesis of aromatic compounds. Clearly indicate under which conditions the group is introduced and removed. CH3 Compound A several steps (5) d) The scheme below shows that compound A gets converted to its diazonium salt which is then reacted with compound to give an azo compound. Diazonium salt of compound A OR CH3 Compound B CO₂H CI i. In the formation of an azo compound is the diazonium salt likely to act as the nucleophile or electrophile. Give reason for your answer. ii. Which of the two structures shown below is compound B likely to be? Indicate which of its properties allow it to react with the diazonium salt. iii. Draw the structure of compound A. COzH (4+5) Semester 1: 2022/20
Expert Answer:
Answer rating: 100% (QA)
The given reaction is The methyl group on the benzene ring is orthop... View the full answer

Related Book For 

Organic Chemistry
ISBN: 978-1118133576
11th edition
Authors: Graham Solomons, Craig Fryhle, Scott Snyder
Posted Date:
Students also viewed these chemical engineering questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
A woman flies from Phoenix to Denver (a distance of 800 mi) at a rate 40 mph faster than on the return trip. If the total time of the trip is 9 hrs, what was her rate going to Denver, and what was...
-
Bank Management Printers, Inc., produces luxury checkbooks with three checks and stubs per page. Each checkbook is designed for an individual customer and is ordered through the customer's bank. The...
-
Many have suggested that vitamin D could be more appropriately called a hormone than a vitamin. Is this correct?
-
The file colleges contains the following data for the sample of 102 private colleges: year founded, tuition and fees (not including room and board), and the percentage of undergraduates who obtained...
-
Review the February 26 and March 25 transactions for Success Systems (SP 5) from Chapter 5. Required 1. Assume that Lyn Addie is an unmarried employee. Her $ 1,000 of wages are subject to no...
-
Discuss how men and women differ in their efforts to end their lives. How do marriage, age, and ethnicity influence these differences? 3. Discuss suicide prevention strategies.
-
Calculator Calculating Payroll Taxes Expense and Preparing Journal Entry Selected information from the payroll register of Ebeling's Dairy for the week ended July 7, 20--, is shown below. The SUTA...
-
A 10 MVA, 22 kV star connected alternator has armature resistance of 0.5 ohm. The machine is supplying 90 A current at zero pf leading when the terminal voltage is 25 kV. Under this condition, if the...
-
Evaluate the following postfix expression: 112 4/37*-5+
-
1. What is a special law? 2. LAW 54 -DO YOU THINK THE LAW FOR THE PREVENTION OF DOMESTIC VIOLENCE IS A DETERRENt OR PREVENT DOMESTIC ABUSE?WHY? 3. STALKING . Look up the Puerto Rico stalking law...
-
V 3. Find poles and zeros. Draw ROC. Find inverse z transform of the following signal X(z) = (z +0.1) (z-1.5z+0.5)
-
A tennis ball with a mass of 150 g is initially travelling at 15 m/s [E] when it is struck by a racket. The ball now is moving at 20 m/s [W]. If the time of contact between the racket and ball is...
-
A truck for hauling coal has an estimated net cost of $80000 and is expected to give service for 230000 kilometres. Its salvage value will be $4000 and depreciation will be charged at a rate of 33...
-
Presented below are the components in determining cost of goods sold. Determine the missing amounts. Beginning Inventory Cost of Goods Available for Sale Purchases $70,000 $90,000 $40,000 $100,000 tA...
-
Suppose that the electrical potential at the point (x, y, z) is E(x, y, z) = x + y - 2z. What is the direction of the acceleration at the point (1,3,2)?
-
Rewrite each of the following using bond-line formulas: (a) (b) (c) (CH3)3CCH2CH2CH2OH (d) (e) CH2==CHCH2CH2CH==CHCH3 (f) CH3CH2CH2CCH3 CH3CHCH2CH2CHCH2CH3 CH3 CH3 CHsCH2CHCH2COH CH3 CH2 HC H2
-
Using decarboxylation reactions outline a synthesis of each of the following from appropriate starting materials: (a) 2-Hexanone (b) 2-Methylbutanoic acid (c) Cyclohexanone (d) Pentanoic acid
-
Use the curved-arrow notation to write the reaction that would take place between dimethylamine (CH3)2NH and boron trifluoride. Identify the Lewis acid, Lewis base, nucleophile, and electrophile and...
-
Coherent states are potentially an interesting basis to consider in which to express states on the Hilbert space. Are they a good basis, satisfying qualities that we desire of a basis on the Hilbert...
-
In this chapter, we had only expressed eigenstates of the harmonic oscillator Hamiltonian through repeated action of the raising operator, \(\hat{a}^{\dagger}\). This gives us a concrete algorithm...
-
A simple model for radioactive decay of an unstable nuclear isotope is as follows.3 Consider the potential illustrated in Fig. 7.9, in which there is a hard, infinite barrier at the spatial origin, a...

Study smarter with the SolutionInn App


