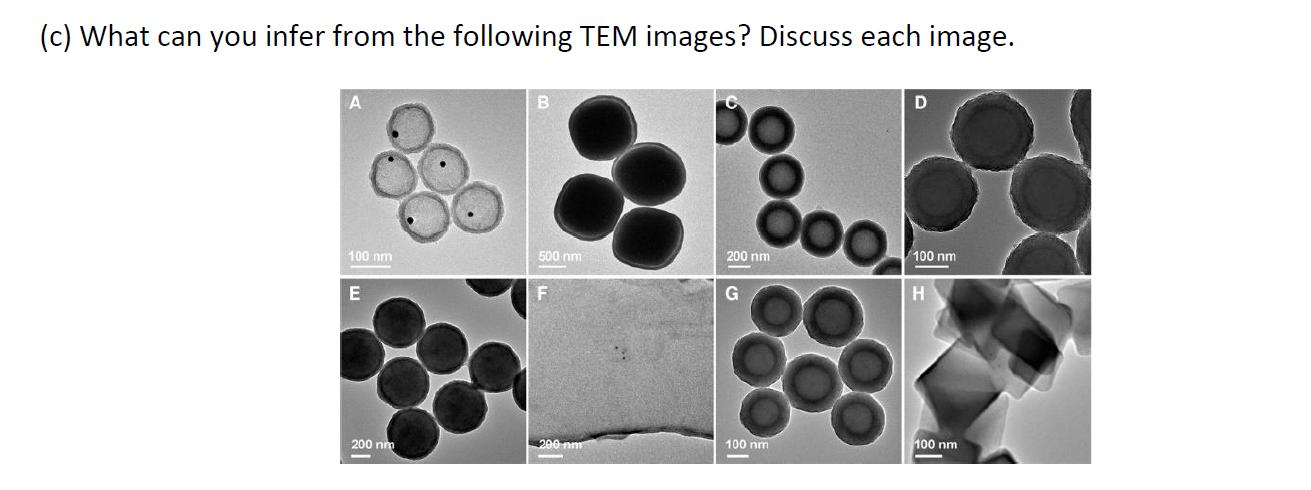
(c) What can you infer from the following TEM images? Discuss each image. 200 nm 100...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
(c) What can you infer from the following TEM images? Discuss each image. 200 nm 100 nm 100 nm 500 nm G 200 nm 100 nm 100 nm 200 nm (c) What can you infer from the following TEM images? Discuss each image. 200 nm 100 nm 100 nm 500 nm G 200 nm 100 nm 100 nm 200 nm
Expert Answer:
Answer rating: 100% (QA)
TEM stand for Transmission electron microscopy which is used for study of internal structure of samp... View the full answer

Related Book For 

Posted Date:
Students also viewed these physics questions
-
1. What can you infer about this organization and its structure based on the conversation you witness in this video clip? 2. Would you describe this as a mechanistic or organic organization? Explain...
-
This organism is an animal. What can you infer about its body structure and lifestyle (that might not be obvious from its appearance)? This animal has a deuterostome developmental pattern and a...
-
You own 100 shares of a C corporation. The corporation earns $5.00 per share before taxes. Once the corporation has paid any corporate taxes that are due it will distribute the rest of its earnings...
-
A charged isolated metal sphere of diameter 10 cm has a potential of 8000 V relative to V = 0 at infinity. Calculate the energy density in the electric field near the surface of the sphere.
-
Four Seasons Boots Ltd. had sales in November 2010 of $538,255. Ten percent of the sales were cash sales, with 70% of the credit sales collected one month after the sale, 25% collected two months...
-
BuildCo, Inc., a domestic C corporation, has used ASC 740-30 (APB 23) to avoid reporting any U.S. deferred tax expense on $100 million of the earnings of BuildCo's foreign subsidiaries. All of these...
-
The following is a general result from matrix theory: Let $\mathbf{A}$ be an $m \times n$ matrix. Suppose that the equation $\mathbf{A x}=\mathbf{p}$ can achieve no $\mathbf{p} \geq \mathbf{0}$...
-
The company reported the following information for the year: Ending work-in-process inventory . . . . . . . . . . . . . . . . . . . . . . . . . . . . $100,000 Depreciation on factory building . . . ....
-
Suppose that next year you observe a higher equilibrium price but note that the equilibrium quantity of bagels sold is unchanged. What changes to the price of a donut and the price of flour could...
-
First Trax Company manufactures snowboards. Its standard cost information follows. First Trax has the following actual results for the month of June: Number of units produced and sold...
-
Q.4. All the tulips in Zoe's garden are white. All the pansiesin Zoe's garden are yellow. All the flowers in Zoe's garden areeither white or yellow. If the first two statements are true. Thethird...
-
1. What is Stoichiometry? 2. What is a Chemical Equation? 3. Balance and then interpret the chemical equation: NaOH + CuSO, Cu(OH), +Na,SO, 4. Based on your interpretation of the chemical equation...
-
5. Imagine that a car batter costs $30 to manufacture and $45 to dispose of properly, but it can be disposed of improperly at a cost of $2 and, well, it's a car battery, so it really can't be traced...
-
Your manager has assigned you to present valuation recommendations to the senior leadership team of your organization. You are to justify how properly implemented valuation techniques may improve the...
-
If a silver dollar contains 91.0% silver by mass, how many kilograms of silver can be extracted from 4.68 kg of silver dollars? Enter your answer numerically in units of kilograms.
-
You have taken on a seven-week contract with the management consulting firm BLUEtin Consulting. This contract is covering one project cycle and all learning activities and assessment will be tied to...
-
How do materialized views enhance query performance and support complex analytics in data warehousing environments by precomputing and caching aggregated or derived data from underlying tables, and...
-
Quality Chicken grows and processes chickens. Each chicken is disassembled into five main parts. Information pertaining to production in July 2012 is: Joint cost of production in July 2012 was $50. A...
-
What responsibility should a company have to their employees quality of life and working conditions? Provide specific examples to support your answer.
-
What are potential risks and rewards of using partners as part of the Preserve brands overall strategy?
-
From an employee recruitment and retention perspective, what benefits, policies, and management practices provide Gold + Williams with a competitive advantage? Explain why.
-
The distribution of the energy of the molecules is also of importance in the kinetics of chemical reactions. The fraction of molecules with energy in the range between \(E\) and \(E+d E\) is given...
-
Show the details leading to the equation (1.52) in the text, \[D_{\mathrm{AA}}=\frac{1}{3} \bar{c} \lambda\] where \(D_{\mathrm{AA}}\) is the self-diffusion coefficient.
-
The simple formula \(C_{p}=(5 / 2) R\) (molar units) is valid only for monatomic gases. A simple extension that has been suggested is \[C_{p}=\left(5+N_{\mathrm{r}} ight) \frac{1}{2} R\] where...

Study smarter with the SolutionInn App