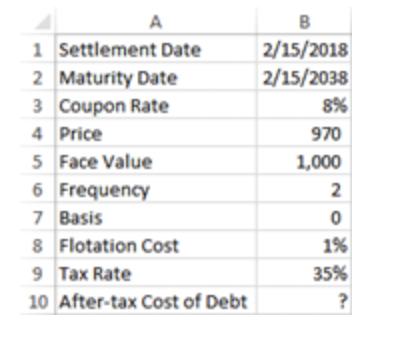
Calculate the after-tax cost of debt? A 1 Settlement Date 2 Maturity Date 3 Coupon Rate 4
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
A 1 Settlement Date 2 Maturity Date 3 Coupon Rate 4 Price 5 Face Value 6 Frequency 7 Basis 8 Flotation Cost 9 Tax Rate 10 After-tax Cost of Debt B 2/15/2018 2/15/2038 8% 970 1,000 2 0 1% 35% ? A 1 Settlement Date 2 Maturity Date 3 Coupon Rate 4 Price 5 Face Value 6 Frequency 7 Basis 8 Flotation Cost 9 Tax Rate 10 After-tax Cost of Debt B 2/15/2018 2/15/2038 8% 970 1,000 2 0 1% 35% ?
Expert Answer:
Answer rating: 100% (QA)
Flotation Cost Price Flotation Cost Rate Flotation Cost 970 001 970 Net Price Price Flotat... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these finance questions
-
The hazard rate of a device is h(t) = 1/ Vt. Find the following: %3D a) Probability density function b) Reliability function c) MTTF d) Variance
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
An investment will pay 24k in 6 years. If the interest rate is 10% with daily compounding what is the value of this contract today. If the quoted rate is 9.75% per year what is the present value of...
-
Given the following information, prepare a production report with materials added at the beginning and ending work in process inventory being 20% complete with regard to conversion costs. Costs to...
-
A 750-cm-long guitar string of mass 2.10g is near a tube that is open at one end and also 75 cm long. How much tension should be in the string if it is to produce resonance (in its fundamental mode)...
-
The gas space above the water in a closed storage tank contains nitrogen at 25C, 100 kPa. Total tank volume is 4 m3, and there is 500 kg of water at 25C. Additional 500 kg water is...
-
Locate an expert in the area of products liability who resides in California. Are there websites that provide a directory of experts in California who can testify in civil litigation cases?
-
Kim Yi has prepared the following list of statements about accounts. 1. An account is an accounting record of either a specific asset or a specific liability. 2. An account shows only increases, not...
-
Question 1: Capital Budgeting with a Spreadsheet MitKees Enterprises is evaluating a project with the following characteristics: Fixed capital investment is $2,000,000. The project has an expected...
-
Crane Library, a nonprofit organization, presented the following statement of financial position and statement of activities for its fiscal year ended February 28, 2024. Assets Current Assets Cash...
-
Sylvestor Systems borrows $177,000 cash on May 15 by signing a 30-day, 5%, $177,000 note. 1. On what date does this note mature? 2-a. Prepare the entry to record issuance of the note. 2-b. First,...
-
[Language-Science Kits] The president of Kool Science, Inc., which makes educational science kits for children, calls the president of Language Fun, Inc., which makes foreign language learning...
-
Explain why an understanding of the law of demand and the law of supply is important to being an effective manager. Identify the supply factors that are most important in determining the market...
-
How do technology cycles intersect with broader socio-economic transformations, such as the transition to a knowledge-based economy, the rise of digital platforms, and the Fourth Industrial...
-
Explain in detail Johnson & Johnson's competitive rivalry. Who are their rivals and what can they do differently?
-
Imperial Jewelers manufactures and sells a gold bracelet for $410.00. The company's accounting system says that the unit product cost for this bracelet is $260.00 as shown below: Direct materials...
-
PF Focus Inc (hereafter, PFI) is considering the impact that an improvement to the environmental management of their production processes. PFI already has an uncertified program in place....
-
What mass of H2 will be produced when 122 g of Zn are reacted? Zn(s) + 2HCl(aq) ( ZnCl2(aq) + H2(g)
-
Ulysses and Penelope are married and file separate returns for 2012. Penelope itemizes her deductions on her return. Ulysses' adjusted gross income was $17,400, his itemized deductions were $2,250,...
-
Sophie is a single taxpayer. For the first payroll period in October 2012, she is paid wages of $3,250 monthly. Sophie claims three allowances on her Form W-4. a. Use the percentage method to...
-
Laura is a single taxpayer living in New Jersey with adjusted gross income for the 2012 tax year of $35,550. Laura's employer withheld $3,300 in state income tax from her salary. In April of 2012,...
-
Covenant Trucking Company uses the units-of-production (UOP) depreciation method because UOP best measures wear and tear on the trucks. Consider these facts about one Mack truck in the company's...
-
PepsiCo, Inc., has acquired several other companies. Assume that PepsiCo purchased Kettle Chips Co. for \(\$ 8\) million cash. The book value of Kettle Chips' assets is \(\$ 12\) million (market...
-
The board of directors of Park Place Porsche is having a quarterly meeting. Accounting policies are on the agenda, and depreciation is being discussed. A new board member, an attorney, has some...

Study smarter with the SolutionInn App


