Naturally occurring lead exists as four stable isotopes: 204X with a mass of 203.973 amu (1.4806);...
Fantastic news! We've Found the answer you've been seeking!
Question:

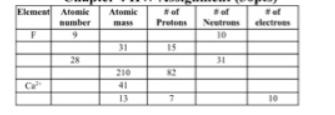
Transcribed Image Text:
Naturally occurring lead exists as four stable isotopes: 204X with a mass of 203.973 amu (1.4806); 206X 205.974 amu (23.60%); 207X 206.9759 amu (22.60%); and 20X 207.9766 amu (52.30%). Calculate the average atomic mass of X. 68.9257 amu is the mass of 60.4% of the atoms of an element with only two naturally occurring isotopes. The atomic mass of the other isotope is 70.9249 amu. Determine the average atomic mass of the element? Identify the element? Give the nuclear composition and isotopic notation for a, an atom containing 27 protons, 32 neutrons, and 27 electrons b. an atom containing 15 protons, 16 neutrons, and 15 electrons c. an atom containing 110 neutrons, 74 clectrons, and 74 protons d. an atom containing 92 electrons, 143 neutrons, and 92 protons Ekment of of ef Atomie Atomic Rumber Protons Neutrons electrees mass 10 15 28 31 210 Ca 13 10 Naturally occurring lead exists as four stable isotopes: 204X with a mass of 203.973 amu (1.4806); 206X 205.974 amu (23.60%); 207X 206.9759 amu (22.60%); and 20X 207.9766 amu (52.30%). Calculate the average atomic mass of X. 68.9257 amu is the mass of 60.4% of the atoms of an element with only two naturally occurring isotopes. The atomic mass of the other isotope is 70.9249 amu. Determine the average atomic mass of the element? Identify the element? Give the nuclear composition and isotopic notation for a, an atom containing 27 protons, 32 neutrons, and 27 electrons b. an atom containing 15 protons, 16 neutrons, and 15 electrons c. an atom containing 110 neutrons, 74 clectrons, and 74 protons d. an atom containing 92 electrons, 143 neutrons, and 92 protons Ekment of of ef Atomie Atomic Rumber Protons Neutrons electrees mass 10 15 28 31 210 Ca 13 10
Expert Answer:
Answer rating: 100% (QA)
Q1 Isotope abundance Atomic mass amu X204 1480 203973 X206 2360 205974 X207 ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Protons and neutrons (together called nucleons) are held together in the nucleus of an atom by a force called the strong force. At very small separations, the strong force between two nucleons is...
-
Symbol Atomic # Protons Neutrons Atomic Mass Na 11 11 24 - 11 = 13 24 24-11: 24 Na 13
-
Protons with a kinetic energy of 2.00 MeV scatter off gold nuclei in a foil target. Each gold nucleus contains 79 protons. If both the incoming protons and the gold nuclei can be treated as point...
-
Solve the inequalities and show the solution sets on the real line. -2x > 4
-
Uncle Vern has just come into some money $100,000 and is thinking about putting this away into some investment accounts for a while. A: Vern is a simple guy so he goes to the bank and asks them...
-
Different types and capacities of crawler hoes are being considered for use in a major excavation on a pipe-laying project. Several supervisors who served on similar projects in the past have...
-
Tyler Company reported the following costs on its financial statements (in thousands): REQUIRED: Using the reserve disclosure for Tyler Company in problem 13 and the data presented in this problem,...
-
Reynolds.com uses the allowance method of accounting for bad debts. The company produced the following aging of the accounts receivable at year-end. Instructions (a) Calculate the total estimated bad...
-
$400,000 for a new technology.The lender charges them 36% annually with monthly compounding.The agreement calls for no payment until the end of the first month of the 5th year with equal monthly...
-
Mr G is an accountant. Mr. G is 47 years old and is married to Claire who is 45 years old and blind. She has Net Income For Tax Purposes in 2020 of $9,000, all of which is interest on investments she...
-
Contrast the general penetration mechanisms utilized by animal viruses to that utilized by large bacterial viruses.
-
Is it appropriate to write and discuss the civil rights movement as a singular noun? Why or why not? Use at least three of the primary source documents to construct your answer to this question.
-
An employer faces a minimum wage control where it cannot pay its workers less than $10.25 per hour. The employer knows that workers value the jobs and are willing to work even for much less. The...
-
The Federal Reserve Board, under Jerome Powell, is keeping interest rates at relatively low levels (now near zero).What are the benefits and consequences of taking such action?Include in your answer...
-
Find three examples of companies that were not the pioneers, but entered later to eventually overtake the pioneer as the market leader.In his opinion, why did the failed pioneers fail?
-
(a) What is the equivalent resistance of four resistors connected in series with a 17.0-V battery if each resistor has a value of 24.0 ? (b) Determine the current flowing through each of the four...
-
1. The balance sheet reports the net value ( book value) of the equipment. The book value of the equipment is______________. Select one or more: a. The original cost of the asset b. The original cost...
-
Factor and simplify, if possible. Check your result using a graphing calculator. 3 cot 2 + 6 cot + 3
-
An electrochemical cell is made by placing an iron electrode in 1.00 L of 0.15 M FeSO4 solution and a copper electrode in 1.00 L of 0.036 M CuSO4 solution. a. What is the initial voltage of this cell...
-
An antacid tablet weighing 0.853 g contained calcium carbonate as the active ingredient, in addition to an inert binder. When an acid solution weighing 56.519 g was added to the tablet, carbon...
-
Write the electron configuration and the Lewis symbol for Ca2+ and for S2.
-
Please list possible definitions of the term embedded system!
-
How would you define the term cyber-physical system (CPS)? Do you see any difference between the terms embedded systems and cyber-physical systems?
-
What is the Internet of Things (IoT)?

Study smarter with the SolutionInn App


