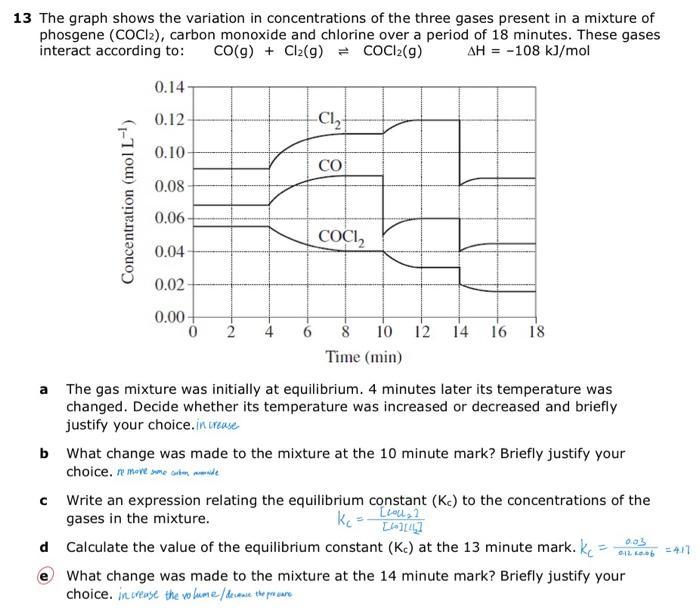
13 The graph shows the variation in concentrations of the three gases present in a mixture...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
13 The graph shows the variation in concentrations of the three gases present in a mixture of phosgene (COCI2), carbon monoxide and chlorine over a period of 18 minutes. These gases interact according to: CO(g) + Cl₂(g) = COCl2(g) AH = -108 kJ/mol Concentration (mol L-¹) 0.14- 0.12- 0.10 0.08- 0.06- 0.04- 0.02- 0.00+ 0 2 -C₁₂ CO COCI₂2 10 12 14 16 18 Time (min) a The gas mixture was initially at equilibrium. 4 minutes later its temperature was changed. Decide whether its temperature was increased or decreased and briefly justify your choice.increase b What change was made to the mixture at the 10 minute mark? Briefly justify your choice. remove me tende с Write an expression relating the equilibrium constant (Kc) to the concentrations of the gases in the mixture. kc [lol₂] [4][44] d Calculate the value of the equilibrium constant (Kc) at the 13 minute mark. k 0.03 012 60.06 What change was made to the mixture at the 14 minute mark? Briefly justify your choice. increase the volume / decause the procure = 4.17 13 The graph shows the variation in concentrations of the three gases present in a mixture of phosgene (COCI2), carbon monoxide and chlorine over a period of 18 minutes. These gases interact according to: CO(g) + Cl₂(g) = COCl2(g) AH = -108 kJ/mol Concentration (mol L-¹) 0.14- 0.12- 0.10 0.08- 0.06- 0.04- 0.02- 0.00+ 0 2 -C₁₂ CO COCI₂2 10 12 14 16 18 Time (min) a The gas mixture was initially at equilibrium. 4 minutes later its temperature was changed. Decide whether its temperature was increased or decreased and briefly justify your choice.increase b What change was made to the mixture at the 10 minute mark? Briefly justify your choice. remove me tende с Write an expression relating the equilibrium constant (Kc) to the concentrations of the gases in the mixture. kc [lol₂] [4][44] d Calculate the value of the equilibrium constant (Kc) at the 13 minute mark. k 0.03 012 60.06 What change was made to the mixture at the 14 minute mark? Briefly justify your choice. increase the volume / decause the procure = 4.17
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
When a firm needs to raise money via a bond issue, one of the quickest ways is through a______. This activity involves the purpose of a large block of securities by a large institutional investor...
-
Financial markets allow savers and borrowers to meet differing consumption preferences if Select one a. a limited number of financial intermediaries exist b. riskier borrowers are able to access...
-
The information in the data table above represents two different trials for an experiment to study the rate of the reaction between NO (g) and H 2(g) , as represented by the balanced equation above...
-
The following auditors report was drafted by a staff accountant of Turner & Turner, CPAs, at the completion of the audit of the financial statements of Lyon Computers, Inc. (a non-public company) for...
-
The following events occurred at VG Consulting during the most recent month. a. VG provided consulting services and billed clients $ 2,000. b. VG collected $ 1,500 for consulting services performed...
-
Describe how a cash-flow budget is constructed.
-
Question: When Shiloh interviewed for a sales job at a medical supply company, the interviewer promised that she could work exclusively selling medical devices and would not have to be involved in...
-
Selected comparative financial statements of Korbin Company follow. Required 1. Compute each years current ratio. (Round ratio amounts to one decimal.) 2. Express the income statement data in common-...
-
Bobcat Software has a dividend yield of 5% and a current growth rate of 12% per year. Growth is expected to decline linearly for 8 years until the company hits its mature growth rate of 4% per year....
-
Nathan is married with two children and has AGI of $405,000. He also has the following AMT adjustments and preferences: Itemized deductions: medical after floor:$5,375 regular tax taxes: $34,354...
-
Find a Cartesian equation for the curve. 2 cos(20) = 64 Identify the curve. O circle O hyperbola Oline Olimaon O ellipse
-
\(n=800, p_{i}=0.7\) Find the expected frequency for the values of \(n\) and \(p_{i}\).
-
Why do you think that a single best strategy for selecting auditees has not been agreed upon?
-
Claim: \(\mu_{1}>\mu_{2} ; \alpha=0.10\). Assume \(\sigma_{1}^{2} eq \sigma_{2}^{2}\) Sample statistics: \(\bar{x}_{1}=520, s_{1}=25, n_{1}=7\) and \[ \bar{x}_{2}=500, s_{2}=55, n_{2}=6 \] Test the...
-
Outline and discuss briefly eight factors that help to define an organizations strategy for selecting auditees to be included in the budget period.
-
Identify the four steps in selecting auditees for a budget period.
-
. 1. The following table shows the relationship between Good X, Good Y and consumers' income. Price of good Quantity Quantity Consumers' X (RM) demanded for demanded for income per good X (unit)...
-
A local politician is concerned that a program for the homeless in her city is discriminating against blacks and other minorities. The following data were taken from a random sample of black and...
-
A certain reaction is spontaneous at 72C. If the enthalpy change for the reaction is 19 kJ/mol, what is the minimum value of S (in J/K ? mol) for the reaction?
-
A compound has the empirical formula SF4. At 20C, 0.100 g of the gaseous compound occupies a volume of 22.1 mL and exerts a pressure of 1.02 atm. What is the molecular formula of the gas?
-
Ethanol, C2H5OH, and dimethyl ether, CH3OCH3, are structural isomers. Compare their melting points, boiling points, and solubilities in water.
-
What three conditions are normally present when misappropriation occurs?
-
What is money laundering? How do you catch it?
-
List the things that should not be done in a fraud investigation.

Study smarter with the SolutionInn App


