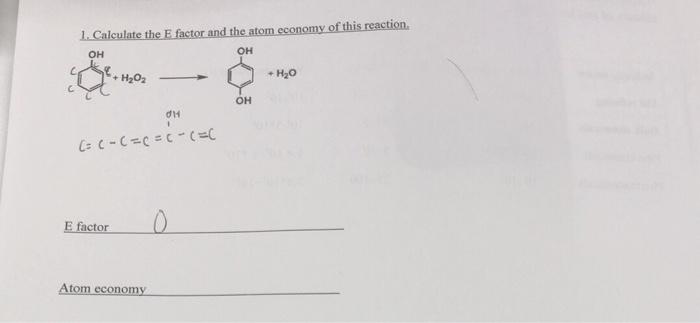
1. Calculate the E factor and the atom economy of this reaction. + H2O2...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
1. Calculate the E factor and the atom economy of this reaction. он он + H2O2 + H20 E factor Atom economy 1. Calculate the E factor and the atom economy of this reaction. он он + H2O2 + H20 E factor Atom economy
Expert Answer:
Answer rating: 100% (QA)
The Given equnation is OH OH L0 MA phenolm m2 desivad pr... View the full answer

Posted Date:
Students also viewed these chemistry questions
-
Factor A Factor B 1 2 3 4 Xj for Factor B 1 2 3 4 1 2500 2 9 10 6 9 8500 3 14 11 16 12 13250 Xi for Factor A 8333 8000 9000 7333 X8083
-
Wonder Textile has 10-year-old sewing machine with a cost price of $5,000. The rate of depreciation on the sewing machine is 10% per annum. The scrap value is $750. It has not been used in the...
-
Calculate for a hydrogen atom and a He + ion: (a) The radius of the first Bohr orbit and the velocity of an electron moving along it; (b) The kinetic energy and the binding energy of an electron in...
-
Argue whether or not you believe using a sample of students from your schools cafeteria (you recruit the next 100 people to visit the cafeteria to participate) may or may not yield biased estimates...
-
Why does the auditor have to be concerned about variable interest annuities when auditing the consolidation process?
-
A baseball bat of length L has a peculiar linear density (mass per unit length) given by = 0 (1 + x 2 /L 2 ). Find the x coordinate of the center of mass in terms of L.
-
Celestial Inc. is organized into 3 distinct divisions. The company is currently contemplating the elimination of either Division 1 or Division 2 because they are showing a pre-tax loss. An annual...
-
The Pandora Company, a U.S.-based manufacturer of furniture and appliances that offshores all of its manufacturing operations to Asia, has distribution centers at various locations on the East Coast...
-
Lou Barlow, a divisional manager for Sage Company, has an opportunity to manufacture and sell one of two new products for a five- year period. His annual pay raises are determined by his division's...
-
Criterion for Vapor-Free Flow in a Pipeline, to ensure that a pipeline is completely liquid-filled, it is necessary that p > P vap at every point. Apply this criterion to the system in Fig. 7.5-1, by...
-
You should always address who should implement a recommendation at which step of the planning process?
-
Below we present the MO diagram of ML4 molecule with a C4 point group, as well as detailed information of each MO. Here L is a o ligand that only contributes a s orbital (OL orbitals) and M is a...
-
The risk structure and the term structure of interest rate both signal following; 1. Financial Markets( Liquidity Premium Theory). 2. expectations of future economic activity.( Expectations...
-
Explain how fractional reserve banking creates money out of thin air and the potential consequences to the economy if banks over-extend themselves.. Do you think that banks should have a reserve...
-
When a market economy is operating efficiently it is also providing an equitable distribution of income and opportunity. State your opinion re: this statement. Do you agree that this is always the...
-
Assume the short run variable cost function for Japanese beer is VC=0.55 Superscript 0.8q If the fixed cost (F) is $1800 and the firm produces 550 units, determine the total cost of production (C),...
-
5 - 6 | 3 x + 4 | < - 1 3
-
If a and b are positive numbers, find the maximum value of f ( x ) = x a (9 x ) b on the interval 0 x 9.
-
Corporate annual reports include a letter from the chief executive officer (CEO). The letter highlights achievements and positive results from the past year. If necessary, it delivers news of...
-
Alpha Network Solutions has had declining net income the past four years. The company president employs you as a consultant to review the companys operations in an effort to identify the reason for...
-
Minta Perry has received year-end information, including financial statements, from her accountant. Among the information is a report that the accounts receivable turnover ratio in the prior year was...

Study smarter with the SolutionInn App

