Calculate the heat content in a two tonne (2,000 kg) iron magnet at 300 K. How...
Fantastic news! We've Found the answer you've been seeking!
Question:
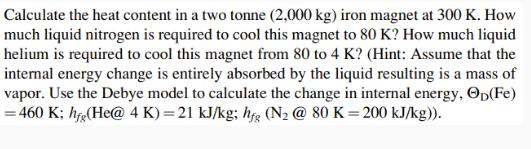
Transcribed Image Text:
Calculate the heat content in a two tonne (2,000 kg) iron magnet at 300 K. How much liquid nitrogen is required to cool this magnet to 80 K? How much liquid helium is required to cool this magnet from 80 to 4 K? (Hint: Assume that the internal energy change is entirely absorbed by the liquid resulting is a mass of vapor. Use the Debye model to calculate the change in internal energy, (Fe) = 460 K; hfg(He@ 4 K)=21 kJ/kg; hfg (N₂ @ 80 K=200 kJ/kg)). Calculate the heat content in a two tonne (2,000 kg) iron magnet at 300 K. How much liquid nitrogen is required to cool this magnet to 80 K? How much liquid helium is required to cool this magnet from 80 to 4 K? (Hint: Assume that the internal energy change is entirely absorbed by the liquid resulting is a mass of vapor. Use the Debye model to calculate the change in internal energy, (Fe) = 460 K; hfg(He@ 4 K)=21 kJ/kg; hfg (N₂ @ 80 K=200 kJ/kg)).
Expert Answer:
Answer rating: 100% (QA)
Mass of iron magnet 2000 kg Initial temperature 300 K Final temperature for liquid nitrogen cooling ... View the full answer

Related Book For 

Fundamentals of Thermodynamics
ISBN: 978-0471152323
6th edition
Authors: Richard E. Sonntag, Claus Borgnakke, Gordon J. Van Wylen
Posted Date:
Students also viewed these finance questions
-
O The region is D: x 0, y 0, x + y 4. Choose the sketch of the region. A -2 D D 4 x x Integrate f(x, y) = 4xy over D using polar coordinates. (Use symbolic notation and fractions where needed.) 14....
-
Helium in a steel tank is at 250 kPa, 300 K with a volume of 0.1 m3. It is used to fill a balloon. When the tank pressure drops to 150 kPa the flow of helium stops by itself, if all the helium still...
-
In a physics lab, a student accidentally drops a 25.0-g brass washer into an open dewar of liquid nitrogen at 77.2 K. How much liquid nitrogen boils away as the washer cools from 293 K to 77.2 K? The...
-
Someone offered the investment options to Hendry on January 1, 2023: 1. Hendry has to save up to 5 times the initial deposit of US $ 150,000/year. 2. The savings cannot be taken for 20 years until...
-
Let s > 0. (a) Find the solution m(x, e) to the boundary value problem - u" + e2 u = 1, u(0) = u(l) = 0. (b) Show that as -> 0+, the solution u(x. s) -> u.(x) converges uniformly to the solution to...
-
How would you summarize the differences between accounting and finance
-
We have \(100 \mathrm{~kg} / \mathrm{h}\) of a feed that is \(60 \mathrm{wt} \%\) methylcyclohexane (A) and \(40 \mathrm{wt} \% \mathrm{n}\)-heptane (D) and \(50 \mathrm{~kg} / \mathrm{h}\) of a feed...
-
The Salza Technology Corporation successfully increased its "top line" sales from $375,000 in 2009 to $450,000 in 2010. Net income also increased as did the venture's total assets. You have been...
-
Required information Problem 9-24A (Algo) Computation of net pay and payroll expense LO 9-5 [The following information applies to the questions displayed below.] The following information is...
-
Although the company has already established medical and retirement plan benefits, Toni Prevosti wants to consider other benefits to attract employees. As the company's accountant, you have been...
-
During the course of the year, Mr. Winston made use of his personal vehicle for business purposes and kept a logbook to keep track of the distance he traveled for work. On January 15, 2021, he put...
-
A ship sailing in the Gulf Stream is heading 25.5 west of north at a speed of 4.05 m/s relative to the water. Its velocity relative to the Earth is 4.60 m/s 5.30 west of north. What is the velocity...
-
What are sequence numbers and why are they used? Describe an attack that uses sequence numbers? Why do you think the he numbering system is desired for use by technologists and hackers alike?
-
A gas has volume of 4 . 0 0 \ times 1 0 4 m 3 mol 1 , temperature of 2 8 8 K and pressure of 4 . 0 MPa. Justify whether the gas behaves as an ideal / perfect gas.
-
West Partners manufactures metal fixtures. Each fitting requires both steel and an alloy that can withstand extreme temperatures. The following data apply to the production of the fittings for year...
-
What is classful addressing? Why it is not used anymore? Discuss class B address in brief.
-
T wo tennis balls of identical mass of 3 kg roll towards each other thee tennis ball on the left is moving at 8 m / s and the one on the right is moving at - 8 m / s .what is the velocity ?
-
What is your opinion of advertising awards, such as the Cannes Lions, that are based solely on creativity? If you were a marketer looking for an agency, would you take these creative awards into...
-
Do Problem 11.26 with ammonia as the working fluid. A supply of geothermal hot water is to be used as the energy source in an ideal Rankine cycle, with R-134a as the cycle working fluid. Saturated...
-
Consider an ideal Stirling-cycle engine in which the state at the beginning of the isothermal compression process is 100 kPa, 25C, the compression ratio is 6, and the maximum temperature in the cycle...
-
Ethane is burned with 150% theoretical air in a gas turbine combustor. The products exiting consist of a mixture of CO2, H2O, O2, N2, and NO in chemical equilibrium at 1800 K, 1 MPa. Determine the...
-
Suppose that in the final simplex system for a dual maximum problem of a given minimum problem, there is a non-basic variable in the objective row with coefficient zero. Recall that this indicates...
-
A woman operating her own business is trying to plan her weekly sales activity schedule to produce the most valuable sales results in the least possible time. She can make personal visits, do phone...
-
Suppose that in the final simplex system for a dual maximum problem of a given minimum problem, there is a degenerate basic slack variable \(x_{j}\). In the equation to which \(x_{j}\) belongs is...

Study smarter with the SolutionInn App


