Calculate the solubility at 25 C of PbCrO, in pure water and in a 0.0160 M...
Fantastic news! We've Found the answer you've been seeking!
Question:
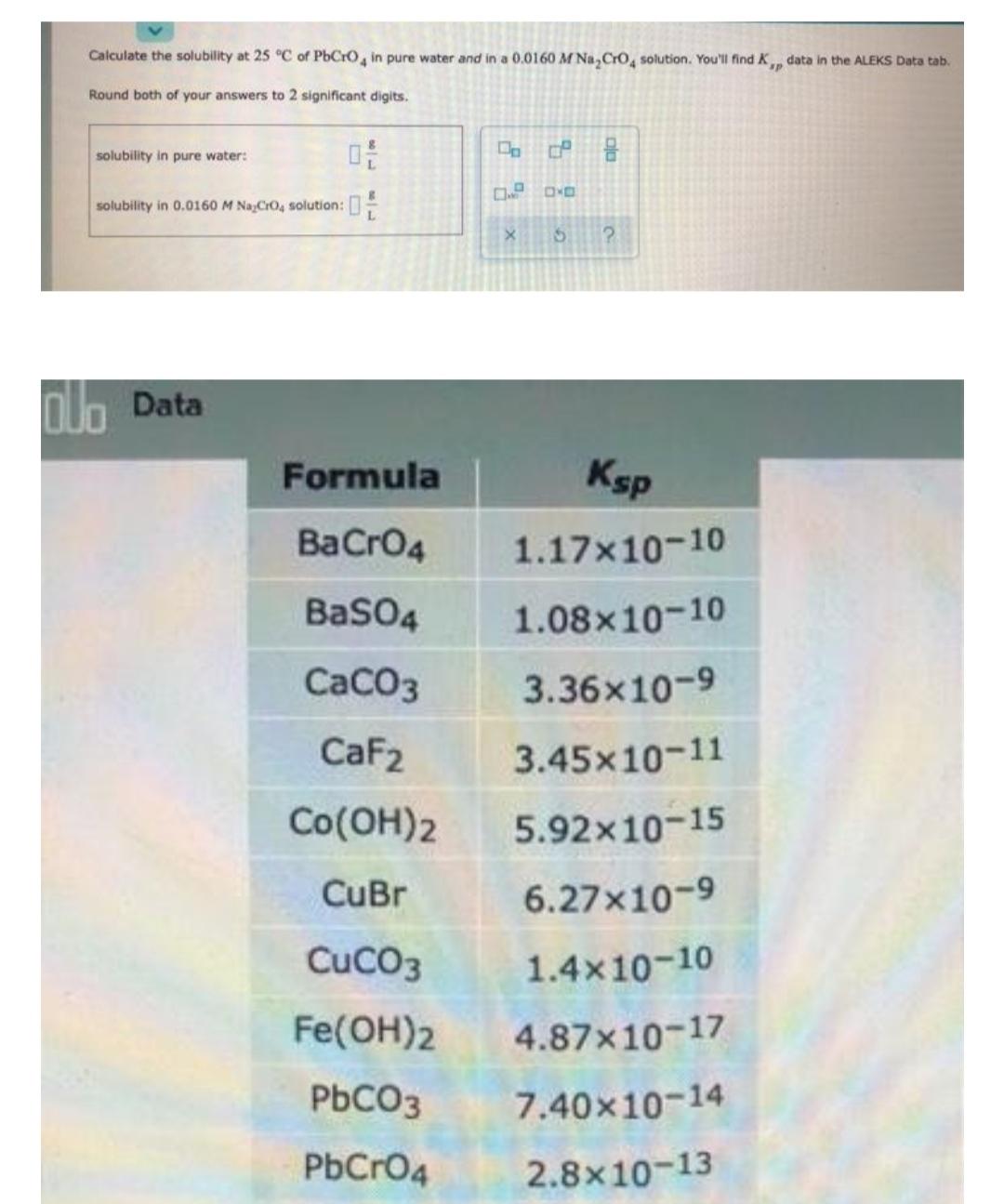

Transcribed Image Text:
Calculate the solubility at 25 °C of PbCrO, in pure water and in a 0.0160 M Na, Cro, solution. You'll find K data in the ALEKS Data tab. Round both of your answers to 2 significant digits. solubility in pure water: D OO solubility in 0.0160 M NaCrO4 solution:: olo Data Formula Ksp BaCrO4 1.17x10-10 BasO4 1.08x10-10 CaCO3 3.36x10-9 CaF2 3.45x10-11 Co(OH)2 5.92x10-15 CuBr 6.27x10-9 CuCO3 1.4x10-10 Fe(OH)2 4.87x10-17 PBCO3 7.40x10-14 PbCrO4 2.8x10-13 Co(OH)2 5.92x10-15 CuBr 6.27x10-9 CuCO3 1.4x10-10 Fe(OH)2 4.87x10-17 PBCO3 7.40x10-14 PbCrO4 2.8x10-13 PBF2 3.3x10-8 Mg(OH)2 5.61x10-12 Ni(OH)2 5.48x10-16 AgBrO3 5.38x10-5 Ag2CO3 8.46x10-12 AgCI 1.77x10-10 Ag2CrO4 1.12x10-12 SrCO3 5.60x10-10 ZnCO3 1.46x10-10 Zn(OH)2 3.0x10-17 AgBr 5.35x10-13 AuCI 1.77x10-10 Calculate the solubility at 25 °C of PbCrO, in pure water and in a 0.0160 M Na, Cro, solution. You'll find K data in the ALEKS Data tab. Round both of your answers to 2 significant digits. solubility in pure water: D OO solubility in 0.0160 M NaCrO4 solution:: olo Data Formula Ksp BaCrO4 1.17x10-10 BasO4 1.08x10-10 CaCO3 3.36x10-9 CaF2 3.45x10-11 Co(OH)2 5.92x10-15 CuBr 6.27x10-9 CuCO3 1.4x10-10 Fe(OH)2 4.87x10-17 PBCO3 7.40x10-14 PbCrO4 2.8x10-13 Co(OH)2 5.92x10-15 CuBr 6.27x10-9 CuCO3 1.4x10-10 Fe(OH)2 4.87x10-17 PBCO3 7.40x10-14 PbCrO4 2.8x10-13 PBF2 3.3x10-8 Mg(OH)2 5.61x10-12 Ni(OH)2 5.48x10-16 AgBrO3 5.38x10-5 Ag2CO3 8.46x10-12 AgCI 1.77x10-10 Ag2CrO4 1.12x10-12 SrCO3 5.60x10-10 ZnCO3 1.46x10-10 Zn(OH)2 3.0x10-17 AgBr 5.35x10-13 AuCI 1.77x10-10
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
The pH at 25 C of an aqueous solution of the sodium salt of -hydroxyquinoline (NaC10H7O) is 11.12. Calculate the concentration of C10H7O- in this solution, in moles per liter. Ka for HC10H7O is equal...
-
What is the pH at 25 C of water saturated with CO 2 at a partial pressure of 1.10 atm? The Henry's law constant for CO 2 at 25 C is 3.1 10 2 mol/L-atm. The CO 2 is an acidic oxide, reading with H 2...
-
Calculate the solubility of Ag,CO3 (a) in pure water and (b) in a solution in which [CO,]= 0.203 M. %3D Solubility in pure water = M Solubility in 0.203 M Co,2 - M %3!
-
Cooper Movie Studio Corp. makes movies and is interested in lowering its operating costs for the following year, while maintaining the high quality and appeal of its movies. Cooper's management is...
-
Citric acid, H3C6H5O7, occurs in plants. Lemons contain 5% to 8% citric acid by mass. The acid is added to beverages and candy. An aqueous solution is 0.688 m citric acid. The density is 1.049 g/mL....
-
How are leases to be accounted for by lessees according to AASB 16/IFRS 16?
-
The Haaland formula for the friction factor is \[ f=\frac{0.3086}{\left\{\log \left[6.9 / \operatorname{Re}+(\varepsilon / 3.7 D)^{1.11} ight] ight\}^{2}} \] Compare this equation for \(f\) for...
-
The following are the statements of financial position of two companies at 31 October 2018, the end of their most recent financial years: The following additional information is available: 1). On 1...
-
A block slides down a ramp that has a coefficient of kinetic friction of 0.2. The ramp makes an angle of 15 degrees with the horizontal. What is the total force that the block feels if its mass is 3...
-
Perform the test in Problem 8.44 using both the raw and ln scale, and report a p-value. Are there any advantages to using the raw or the ln scale? Renal Disease Ten patients with advanced diabetic...
-
2. Construct a natural cubic spline to approximate f(x) 0,0.25, 0.5, 0.75 and 1.0. Integrate the spline the derivatives of the spline to approximate f'(0.5) and f"(0.5). Compare to the analytic...
-
Transactions for C Amden are shown below. Which journal would they be written in? Transaction C Amden purchased goods on credit from C Unwin Goods sold by C Amden to T Coachhouse; C Amden expected to...
-
Prepare the general journal to transfer a gross profit of $17 000 to the profit and loss account.
-
On 25 June 2022 F Stool withdrew inventory for private use at a cost of $825 ($750 + $75 GST). On 29 June $1000 was taken by F Stool from the business bank account for private use. Prepare the...
-
Drawing of goods by D Bonita totalled $385 ($350 + $35 GST). Prepare two general journals showing the entry where perpetual inventory applies and then where periodic inventory applies.
-
On 14 July 2022, P Chaing used the debit card to pay the ABC Concert Department for two tickets to the Sydney Symphony Orchestra. This payment was not related to the business at all. Show the payment...
-
Best Audits is a medium sized auditing firm with branches all over Zimbabwe. You are an audit manager at Best Audits, currently doing your articles. Your fianc is a finance director at Quilts...
-
Calculate Total Contribution Margin for the same items. Total Revenue Total Variable Costs Total Contribution Margin $50.00 a. $116.00 $329.70 b. $275.00 $14,796.00 $7,440.00 c. $40,931.25 d....
-
An unknown salt is either NaF,NaCl, or NaOCl. When 0.050 mol of the salt is dissolved in water to form 0.500 L of solution, the pH of the solution is 8.08.What is the identity of the salt?
-
Calculate the percentage by mass of oxygen in the following compounds: (a) Morphine, C17H19NO3 (b) Codeine, C18H21NO3 (c) Cocaine, C17H21NO4 (d) Tetracycline, C22H24N2O8 (e) Digitoxin, C41H64O13 (f)...
-
Describe how a cholesteric liquid crystal phase differs from a nematic phase?
-
Describe which characteristics of HR metrics and workforce analytics are likely to result in greater return on investment and organizational impact.
-
Why are information security and privacy important considerations in the design, development, and maintenance of an HRIS?
-
List and discuss the major information security and privacy threats to organizations.

Study smarter with the SolutionInn App


