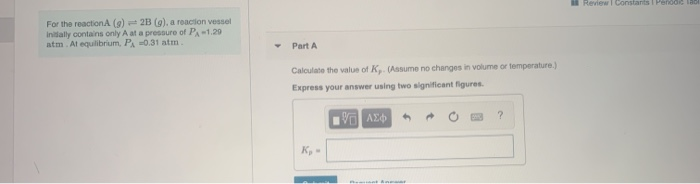
For the reactionA (g) 2B (g), a reaction vessel Initially contains only A at a pressure...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
For the reactionA (g) 2B (g), a reaction vessel Initially contains only A at a pressure of PA-1.29 atm. At equilibrium, PA -0.31 atm. Part A Calculate the value of K,. (Assume no changes in volume or temperature.) Express your answer using two significant figures. Η ΑΣΦ K₂ - Remiant 4 → ? Review 1 Constants 1 Penodic Tabl For the reactionA (g) 2B (g), a reaction vessel Initially contains only A at a pressure of PA-1.29 atm. At equilibrium, PA -0.31 atm. Part A Calculate the value of K,. (Assume no changes in volume or temperature.) Express your answer using two significant figures. Η ΑΣΦ K₂ - Remiant 4 → ? Review 1 Constants 1 Penodic Tabl
Expert Answer:
Answer rating: 100% (QA)
The initial pressure of gas A PA 129 atm The pressure of gas A at ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Using your financial calculator, calculate the value of the following bonds: Par Value $1,000 51,000 $1,000 Interest Rate 5% 59% 5% Required Rate of Return 5% 996 4% Years to Maturity 10 10 10
-
Using the following data, calculate the value of Ksp for Ba(NO3)2, one of the least soluble of the common nitrate salts. Species Baa) NO ia Ba(NOs2s)- -561 kJ/mol -109 kJ/mol -797 k/mol
-
Using information from Table 15.1, calculate the value of DG' for the following reaction. Glucose- + -phosphate Glucose-6-phosphate
-
Give a recursive algorithm for finding the reversal of a bit string. (See the definition of the reversal of a bit string in the preamble of Exercise 34 in Section 5.3.)
-
Dr. R. Chad Halversen performed a laparotomy surgery on Tina Archuleta at St. Mark's Hospital, a Utah facility. Less than two days after being discharged from St. Mark's, Archuleta was admitted to a...
-
Amazon.com, Inc.s financial statements are presented in Appendix D. Financial statements for Wal-Mart Stores, Inc. are presented in Appendix E. Instructions for accessing and using the complete...
-
North Shore Architectural Stone, Inc., a company that installs limestone in residential and commercial buildings, agreed to supply and install limestone for a property owned by Joseph Vitacco. North...
-
Springsteen Co. had the following activity in its most recent year of operations. (a) Pension expense exceeds amount funded. (b) Redemption of bonds payable. (c) Sale of building at book value. (d)...
-
Following are the results from two different simple regression analyses, estimating the costs of the purchasing department using number of purchase orders and number of vendors as potential cost...
-
From 10 men and 8 women in a pool of potential jurors, 12 are chosen at random to constitute a jury. Suppose that you observe the number of men who are chosen for the jury. Let A be the event that at...
-
If interest rates increase from 8% to 9%, we would expect to see A. an increase in the demand for money. B. a decrease in the demand for money. C. an increase in the quantity of money demanded. D. a...
-
A company is evaluating a project that requires an initial investment of $80,000 pesos. In the first year, it will generate profits of $30,000, and these earnings will increase by 10% each year. The...
-
A heat pump absorbs 10 kW of heat from outside environment at 250 K while absorbing 15 kW of work. It delivers the heat to a room that must be kept warm at 300 K. The Coefficient of performance (COP)...
-
Check your eLMS for all the topics discussed from prelim to final period. Create a proposal on how you would like to showcase any art as your best practice. The art you choose should be related to...
-
Christian Pawl holds the opinion that the Black Diamond Skylight three-person three-season tent is the most amazing tent on the market. Pawl is so convinced of the tent's versatility that he would...
-
You are preparing for your role as an international marketing manager and want to get a deeper understanding of high context cultures. Explain what a high-context culture is, what are its...
-
A treasurer for a large multinational firm based in the UK. The firm is attempting to raise 100 million euros for 6 years to expand operations in Spain. With the credit crunch, borrowing rates have...
-
Reconsider Prob. 1474. In order to drain the tank faster, a pump is installed near the tank exit as in Fig. P1475. Determine how much pump power input is necessary to establish an average water...
-
What is the energy (in joules) evolved when 1 mol of helium-4 nuclei is produced from protons and neutrons? How many liters of ethane, C2H6(g), at 25°C and 725 mmHg are needed to evolve the same...
-
You have two piles of different unknown radioactive substances: pile A with a mass of 200 g, and pile B with a mass of 100 g. Would it be possible for these two piles to have the same rate of...
-
Ethane consists of molecules with two atoms of carbon and six atoms of hydrogen. Write the molecular formula for ethane.
-
The microfinance concept has been a blessing for many people in developing countries. Its success there causes some to wonder if it can spur growth in areas of developed nations that need...
-
An institution that many people know little about and some governments find worrisome is offshore financial centers. They operate with little oversight, few regulations, and often little taxation....
-
What is the appeal of the eurocurrency market?

Study smarter with the SolutionInn App


