Carbon monoxide is burned at 200 C under atmospheric pressure with dry air at 500 C in
Fantastic news! We've Found the answer you've been seeking!
Question:
Carbon monoxide is burned at 200 °C under atmospheric pressure with dry air at 500 °C in excess of 90% of that theoretically necessary. The combustion products leave the reaction chamber at 1000 °C. Calculate the heat released in the reaction chamber in kilocalories per kg mol of CO burned, assuming complete combustion.
DATA:
Chemical reaction:
CO (g) + ½ O2 (g) = CO2 (g)
Energy balance equation:
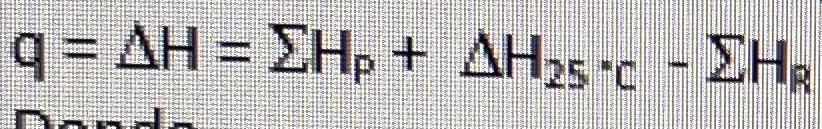
where,
P = products = CO2, O2, N2
R = reactants = CO, Air
Calculation base 1.0 kg mol
Normal heat of reaction = ΔH25°C=-67636 kcal
Heat capacity of substances
| SUBSTANCE | AVERAGE MOLAR HEAT CAPACITY between 25 -1000°C |
| CO | 7.017 |
| Air | 7.225 |
| CO2 | 11.92 |
| O2 | 7.941 |
| N2 | 7.507 |
Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:




