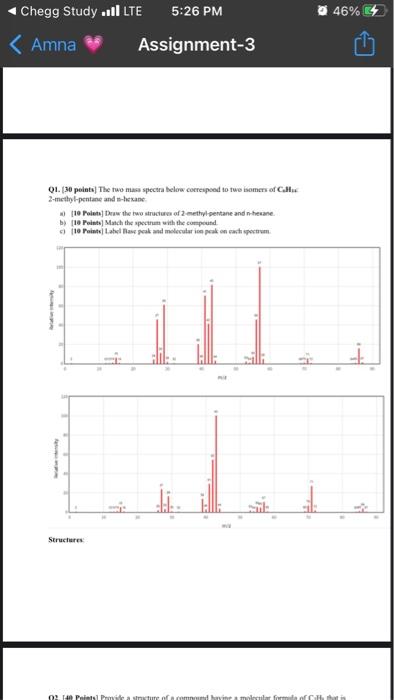
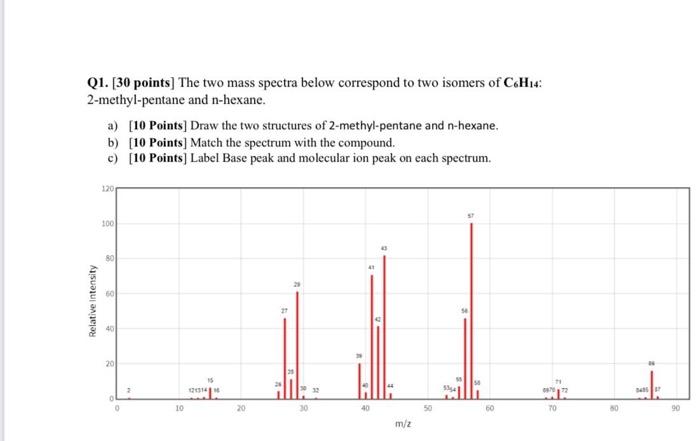
Chegg Study .ILTE Amna 5:26 PM Assignment-3 Q1. [30 points) The two mass spectra below correspond...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
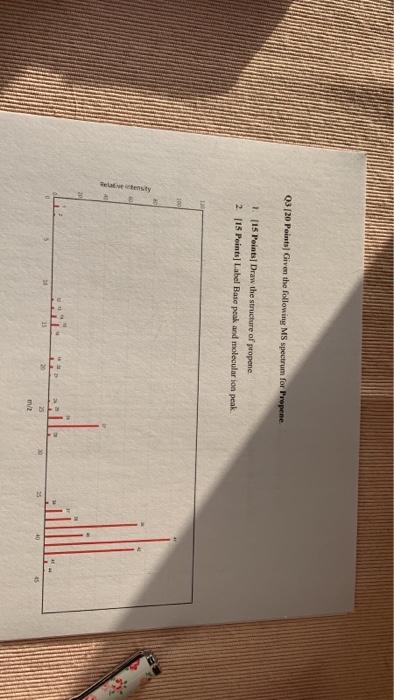
Chegg Study .■ILTE Amna 5:26 PM Assignment-3 Q1. [30 points) The two mass spectra below correspond to two isomers of C 2-methyl-pentime and n-hexane a) [19 Points] Draw the two structures of 2-methyl-pentane and n-hexane b) [10 Points] Match the spectrum with the compound c) (10 Points) Label Base peak and molecular ion peak on each spectrum Structures ا 46% 4 02 140 Points Provide a structure of a compound havine a molecular formida of Call that is Relative intensity Q3 (20 Points] Given the following MS spectrum for Propene. 1. [15 Points] Draw the structure of propene 2 [15 Points] Label Base peak and molecular ion peak 100 25 25 m/2 Q1. [30 points] The two mass spectra below correspond to two isomers of C6H14: 2-methyl-pentane and n-hexane. Relative Intensity a) [10 Points] Draw the two structures of 2-methyl-pentane and n-hexane. b) [10 Points] Match the spectrum with the compound. c) [10 Points] Label Base peak and molecular ion peak on each spectrum. 120 100 20 O 10 121314 15 20 27 30 41 40 43 m/z 50 15 534 60 11 067072 TO 245 St 90 Chegg Study .■ILTE Amna 5:26 PM Assignment-3 Q1. [30 points) The two mass spectra below correspond to two isomers of C 2-methyl-pentime and n-hexane a) [19 Points] Draw the two structures of 2-methyl-pentane and n-hexane b) [10 Points] Match the spectrum with the compound c) (10 Points) Label Base peak and molecular ion peak on each spectrum Structures ا 46% 4 02 140 Points Provide a structure of a compound havine a molecular formida of Call that is Relative intensity Q3 (20 Points] Given the following MS spectrum for Propene. 1. [15 Points] Draw the structure of propene 2 [15 Points] Label Base peak and molecular ion peak 100 25 25 m/2 Q1. [30 points] The two mass spectra below correspond to two isomers of C6H14: 2-methyl-pentane and n-hexane. Relative Intensity a) [10 Points] Draw the two structures of 2-methyl-pentane and n-hexane. b) [10 Points] Match the spectrum with the compound. c) [10 Points] Label Base peak and molecular ion peak on each spectrum. 120 100 20 O 10 121314 15 20 27 30 41 40 43 m/z 50 15 534 60 11 067072 TO 245 St 90
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The 1H NMR spectra of three isomers with molecular formula C4H9Br are shown here. Which isomer produces which spectrum? a. b. c. (ppm) -frequency o (ppm) frequency 5 2 (ppm) frequency
-
The 1H NMR spectra of three isomers with molecular formula C7H14O are shown here. Which isomer produces which spectrum? a. b. c. QUESTION CONTINUE TO NEXT PAGE 0 (ppm) frequency 6 (ppm) frequency 8...
-
The mass spectrum of unknown compound A shows a molecular ion at m/z 116 and prominent peaks at m/z 87 and m/z 101. Its UV spectrum shows no maximum above 200 nm. The IR and NMR spectra of A follow....
-
Fox rented a house from Chiodini, but before he moved in, he noticed exposed electrical wiring in the basement, so he refused to move in. City inspectors confirmed a code violation and Chiodini fixed...
-
Texas International Speedway, Inc. (TIS) filed a registration statement for $4,398,900 in securities with the proceeds to be used to construct an automobile racetrack called the Texas International...
-
Using the MINITAB output and information in Exercise 9.79, determine the values of the following terms: a. Point estimate b. Confidence coefficient c. Standard error of the mean d. Maximum error of...
-
3. Gibson Greetings, Inc., had a plant in Berea, Kentucky, where the workers belonged to the International Brotherhood of Firemen & Oilers. The old CBA expired, and the parties negotiated a new one,...
-
At December 31, 2010, Fell Corporation had a deferred tax liability of $680,000, resulting from future taxable amounts of $2,000,000 and an enacted tax rate of 34%. In May 2011, a new income tax act...
-
Luke, Lando and Leia decide to start a partnership called LLL Consulting on March 1, 2020. Each of them contribute a number of items to the partnership, which are listed below. Luke contributed...
-
Frito-Lay Inc. manufactures convenience foods, including potato chips and corn chips. Production of corn chips occurs in four departments: Cleaning, Mixing, Cooking, and Drying and Packaging....
-
An Ivy League University Endowment Fund believes the stock price for Colleen's Cement Company (CCC) will increase substantially over the next 12 months. The current price is $72.00 per share. The...
-
True or false: An employees work environment has no influence on whether he or she will engage in substance abuse. a. True b. False
-
Workplace bullies have been known to possess high levels of . a. self-confidence b. aggression c. sleep deprivation d. social problems
-
A heuristic framework for helping managers cope with theft is called . a. crime stopper b. audit check c. steal d. honesty
-
In the near future, will organizations have at their disposal tests that can precisely identify job candidates with a high propensity to misbehave? Explain.
-
True or false: Communication, conflict resolution, and problem-solving skills will help a person be a better team member. a. True b. False
-
Widmer Company had gross wages of $200,000 during the week ended June 17. The amount of wages subject to social security tax was $180,000, while the amount of wages subject to federal and state...
-
A 6-lb shell moving with a velocity ?? v0k explodes at point D into three fragments which hit the vertical wall at the points indicated. Fragments A, B, and C hit the wall 0.010 s, 0.018 s, and 0.012...
-
Draw the NMR spectra you expect for the following compounds. (a) (b) (c) (d) C(CH33 CH3O CH3 - Cl C-OCH CH - CH3
-
Show how you would accomplish the following synthetic conversions. (a) but-1-ene butan-2-ol (b) but-1-ene butan-1-ol (c) 2-bromo-2,4-dimethylpentane 2,4-dimethylpentan-3-ol
-
Propose a mechanism for the reaction of benzyl bromide with ethanol to give benzyl ethyl ether (shown above).
-
Clarkson Inc. purchased \(10 \%\) of the 10,000 shares of common stock in Nashville Inc. for \(\$ 40,000\) in January 2020. Shelton Inc. purchased 35\% of the 10,000 shares of common stock in...
-
On December 31, 2020, Raven Company's portfolio of equity securities was valued at \(\$ 1,800\). The original cost of the investments in the portfolio was \(\$ 1,600\). Raven does not have...
-
On January 1, 2020, Evergreen Inc. purchased 3,750 of the 15,000 outstanding shares of common stock of Nature Net Inc. obtaining significant influence of the company. The shares were purchased for...

Study smarter with the SolutionInn App


