CHEM 6 SPRING 2023 (Problem #1) A helium-neon laser emits an intense monochromatic light beam with...
Fantastic news! We've Found the answer you've been seeking!
Question:

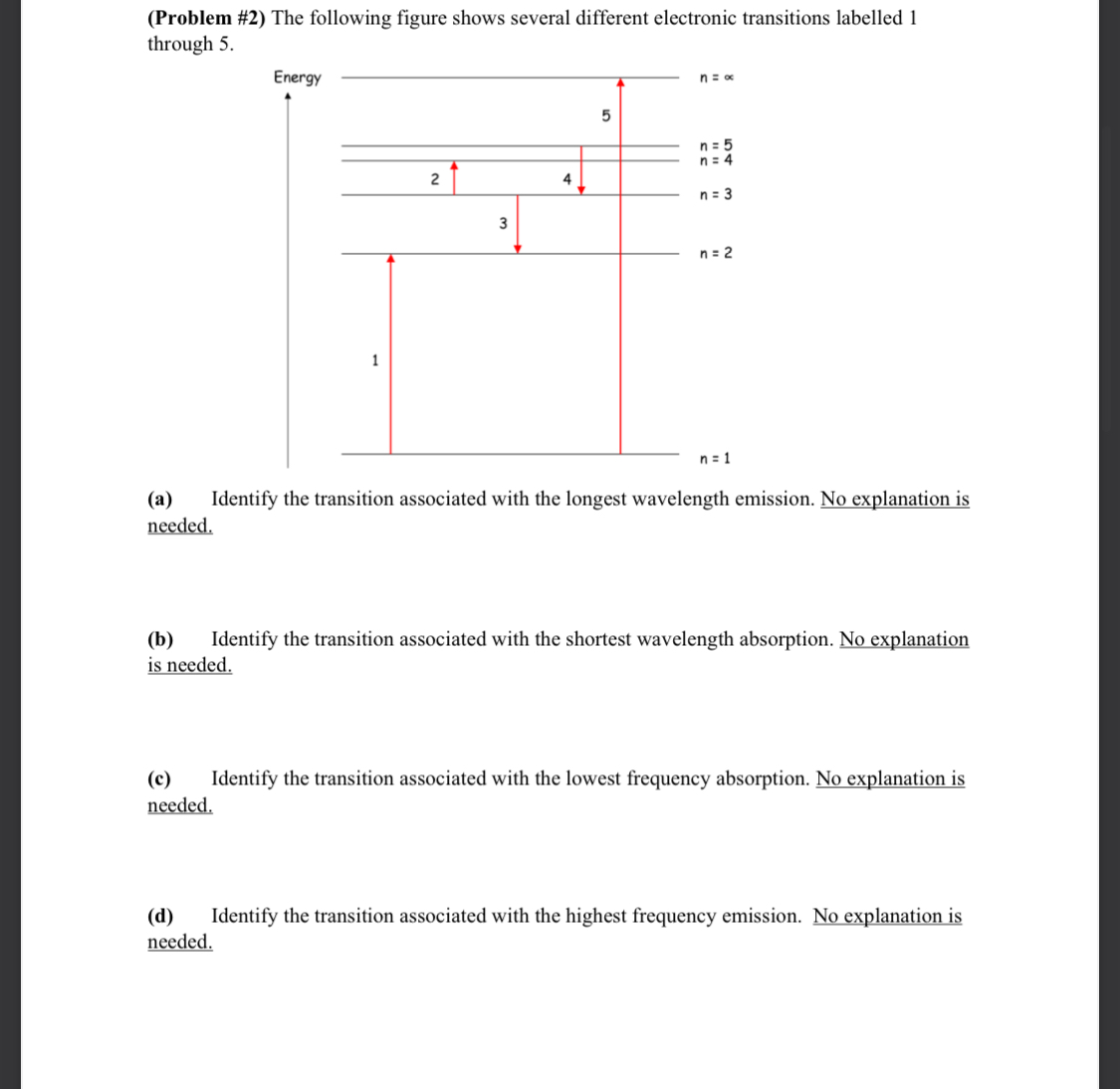








Transcribed Image Text:
CHEM 6 SPRING 2023 (Problem #1) A helium-neon laser emits an intense monochromatic light beam with a wavelength of 6328 . Calculate the angles at which the first- and second-order diffractions will be observed using a diffraction grating with 13,400 lines/inch. Make sure to take care with units in this problem. (Problem #2) The following figure shows several different electronic transitions labelled 1. through 5. Energy 1 2 3 n = x 5 n = 5 n = 4 4 n = 3 n = 2 n = 1 (a) needed. Identify the transition associated with the longest wavelength emission. No explanation is (b) Identify the transition associated with the shortest wavelength absorption. No explanation is needed. (c) needed. Identify the transition associated with the lowest frequency absorption. No explanation is (d) needed. Identify the transition associated with the highest frequency emission. No explanation is CHEM 6 SPRING 2023 (Problem #3) (a) An electron in a one-dimensional box requires the absorption of a photon with a wavelength of 8080 nm to excite it from the n = 2 to the n = 3 energy level. Calculate the length of this box. Show your work and express your final answer in nanometers. (b) An electron in a 10.0 nm one-dimensional box is excited from the ground state into a higher energy state by absorbing a photon of electromagnetic radiation with a wavelength of 1.374 105 m. Determine the final energy state (i.e., quantum number) for this electronic transition. (c) For a particle in the n = 3 state of a one-dimensional box of length L, what is the total probability of finding the particle between x = 0 and x = L/6. Show your work and rationalize your approach to receive full credit. (Problem #4) The orange-yellow light of sodium vapor lamps is emitted when the valence electron of sodium makes a transition from an excited state back to the ground state of the atom. The light emitted is produced by transitions from two different excited states. One of these transitions emits a photon with a wavelength of 589.00 nm, while the other transition emits a photon with a wavelength of 589.59 nm. (a) Draw an energy level diagram showing the relative energies of the ground state and the two excited states -- this diagram need not be to scale. (b) On your diagram, label and show which change of energy level -- i.e., which transition produces the photon with wavelength 589.00 nm. (c) On your diagram, label and show which change of energy level -- i.e., which transition produces the photon with wavelength 589.59 nm. (d) Calculate the energy of each excited state relative to the ground state. Express your answer in kJ mol-1. (e) Calculate the energy difference between the two excited states. Express your answer in kJ mol-1. (Problem #5) Using the Heisenberg uncertainty principle, calculate the uncertainty in position, Ax, for: (a) an electron with Avx = 0.100 m/s. How does your answer compare with the size of a hydrogen atom? (b) a baseball (mass = 145 g) with Avx = 0.100 m/s. How does your answer compare with the size of a baseball? (Problem #6) (a) When electromagnetic radiation with wavelength = 2000 is incident on a clean tungsten plate in a vacuum, the maximum kinetic energy observed amongst the electrons ejected is 1.64 eV. Calculate the threshold wavelength above which it will not be possible to eject electrons from tungsten metal. Show your work and express your answer in nm. (b) The maximum kinetic energy observed amongst electrons ejected from potassium metal by electromagnetic radiation with frequency v = 1.63 1015 s-1 is 7.2 x 10-19 J. Calculate the wavelength associated with the motion of an ejected electron that has this maximum kinetic energy. Show your method clearly and express your answer in nm. CHEM 6 SPRING 2023 (Problem #1) A helium-neon laser emits an intense monochromatic light beam with a wavelength of 6328 . Calculate the angles at which the first- and second-order diffractions will be observed using a diffraction grating with 13,400 lines/inch. Make sure to take care with units in this problem. (Problem #2) The following figure shows several different electronic transitions labelled 1. through 5. Energy 1 2 3 n = x 5 n = 5 n = 4 4 n = 3 n = 2 n = 1 (a) needed. Identify the transition associated with the longest wavelength emission. No explanation is (b) Identify the transition associated with the shortest wavelength absorption. No explanation is needed. (c) needed. Identify the transition associated with the lowest frequency absorption. No explanation is (d) needed. Identify the transition associated with the highest frequency emission. No explanation is CHEM 6 SPRING 2023 (Problem #3) (a) An electron in a one-dimensional box requires the absorption of a photon with a wavelength of 8080 nm to excite it from the n = 2 to the n = 3 energy level. Calculate the length of this box. Show your work and express your final answer in nanometers. (b) An electron in a 10.0 nm one-dimensional box is excited from the ground state into a higher energy state by absorbing a photon of electromagnetic radiation with a wavelength of 1.374 105 m. Determine the final energy state (i.e., quantum number) for this electronic transition. (c) For a particle in the n = 3 state of a one-dimensional box of length L, what is the total probability of finding the particle between x = 0 and x = L/6. Show your work and rationalize your approach to receive full credit. (Problem #4) The orange-yellow light of sodium vapor lamps is emitted when the valence electron of sodium makes a transition from an excited state back to the ground state of the atom. The light emitted is produced by transitions from two different excited states. One of these transitions emits a photon with a wavelength of 589.00 nm, while the other transition emits a photon with a wavelength of 589.59 nm. (a) Draw an energy level diagram showing the relative energies of the ground state and the two excited states -- this diagram need not be to scale. (b) On your diagram, label and show which change of energy level -- i.e., which transition produces the photon with wavelength 589.00 nm. (c) On your diagram, label and show which change of energy level -- i.e., which transition produces the photon with wavelength 589.59 nm. (d) Calculate the energy of each excited state relative to the ground state. Express your answer in kJ mol-1. (e) Calculate the energy difference between the two excited states. Express your answer in kJ mol-1. (Problem #5) Using the Heisenberg uncertainty principle, calculate the uncertainty in position, Ax, for: (a) an electron with Avx = 0.100 m/s. How does your answer compare with the size of a hydrogen atom? (b) a baseball (mass = 145 g) with Avx = 0.100 m/s. How does your answer compare with the size of a baseball? (Problem #6) (a) When electromagnetic radiation with wavelength = 2000 is incident on a clean tungsten plate in a vacuum, the maximum kinetic energy observed amongst the electrons ejected is 1.64 eV. Calculate the threshold wavelength above which it will not be possible to eject electrons from tungsten metal. Show your work and express your answer in nm. (b) The maximum kinetic energy observed amongst electrons ejected from potassium metal by electromagnetic radiation with frequency v = 1.63 1015 s-1 is 7.2 x 10-19 J. Calculate the wavelength associated with the motion of an ejected electron that has this maximum kinetic energy. Show your method clearly and express your answer in nm.
Expert Answer:

Related Book For 

Physics for Scientists and Engineers A Strategic Approach with Modern Physics
ISBN: 978-0133942651
4th edition
Authors: Randall D. Knight
Posted Date:
Students also viewed these physics questions
-
ered Suppose you hold the following stocks in your portfolio, thier Beta and their corresponding returns are as follows: Stocks Invested amount Beta Return Brooks Bro. $863 1.4 6% Samsung $817 2 8%...
-
Question 2 of 4 A bond that has a face value of $2,000 and coupon rate of 3.20% payable semi-annually was redeemable on July 1, 2021. Calculate the purchase price of the bond on February 10, 2015...
-
The following are sample report output (NOTE: the numbers shown below are not the actual aggregate values. You can write simple SQL queries to verify the actual aggregate values). Report #1: CUSTOMER...
-
Delmott sells a snowboard, Xpert that is popular with snowboard enthusiasts. Below is information relating to Delmotts purchases of Xpert snowboards during September. During the same month, 102 Xpert...
-
Suppose an environmental impact study shows that the coral reef near Port Douglas, Australia, can sustain 20 scuba diving tours per week. Discuss the pros and cons of setting a quota of 20 dive tours...
-
A 0.67-mH inductor and a 130- resistor are placed in series with a 24-V battery. (a) How long will it take for the current to reach 67% of its maximum value? (b) What is the maximum energy stored in...
-
Aggregate Effects on Exchange Rates. Assume that the United States invests heavily in government and corporate securities of Country K. In addition, residents of Country K invest heavily in the...
-
Soybean oil is to be pumped from a storage tank to a processing vessel. The distance is 148 m and included in the pipeline are six right-angle bends, two gate valves and one globe valve. If the...
-
GEOMETRY Word problem i empty gas tank of umped into the tank
-
QUESTION FOUR (a) Given a Total Revenue (TR) as TR-100000q-10q2 and Total Cost (TC) as TC= 10000+20q+q Determine: (i) Profit maximizing output.(3marks) (ii) Maximum profit.(2mark) (iii) Proof that...
-
Economic Theory uses a lot of simplification to get to its key analytical points. One type of simplification is an assumption that economic actors are sensitive to changes in the price and supply of...
-
Instructions Balances at 3 1 January 2 0 0 9 : Debtors control account.............................$ 3 2 , 4 0 0 Creditors control account...........................$ 2 5 , 2 0 0...
-
Using a truth table determine which of the following statements are equivalent to the statement "You don't go to the party and we don't go to the movie, or, you go to the party." Select the correct...
-
Amos Company reported the following liabilities on December 31, 2020: Loans payable, due October 1, 2021 4,000,000 Trade notes payable 450,000 Mortgage payable (P500,000 of 4,000,000 which is due on...
-
During the current year, LL Partnership reported the following items: Sales Utilities and rent 480,000 (80,000) Salaries to employees (200,000) Guaranteed payment partner Antonio (20,000) Interest...
-
Explain how the law in Balfour v Balfour (1919) and Merritt v Merrit (1970) has been affected by the High Court decision in Ermogenous v Greek Orthodox Community of SA Inc (2002). What was the...
-
Tell whether the angles or sides are corresponding angles, corresponding sides, or neither. AC and JK
-
What sort of cash organisation is generally in place for highly decentralised groups?
-
Is the risk of bankruptcy of a subsidiary an obstacle to cash pooling for a group which balances its accounts daily?
-
What is the main argument against full cash pooling for a group?

Study smarter with the SolutionInn App


